PHYSE 0421 Solid State Physics Period V spring





![Paramagnetism of a magnetic gas Example Partition junction [E(7. 167, 169)] Helmholtz free energy Paramagnetism of a magnetic gas Example Partition junction [E(7. 167, 169)] Helmholtz free energy](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-6.jpg)
![Paramagnetism of a magnetic gas Curie law (Elliott 7. 2. 4. 2) [E(7. 174)] Paramagnetism of a magnetic gas Curie law (Elliott 7. 2. 4. 2) [E(7. 174)]](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-7.jpg)



![Paramagnetism of Valence Electrons in Metals Total density Pauli paramagnetic susceptibility [E(7. 188)] ~[E(7. Paramagnetism of Valence Electrons in Metals Total density Pauli paramagnetic susceptibility [E(7. 188)] ~[E(7.](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-13.jpg)

![Exchange Interaction: H 2 –molecule, singlet-triplet energy splitting Large Rab [E(7. 208)] <0 Coulomb Exchange Interaction: H 2 –molecule, singlet-triplet energy splitting Large Rab [E(7. 208)] <0 Coulomb](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-21.jpg)
![Heisenberg Spin Hamiltonian Generalization to solids Proof Exercise Parametric form [E(7. 217)] Solids Exchange Heisenberg Spin Hamiltonian Generalization to solids Proof Exercise Parametric form [E(7. 217)] Solids Exchange](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-22.jpg)



- Slides: 26

PHYS-E 0421 Solid State Physics Period V, spring 2018 Prof. Martti Puska Dr. Hannu-Pekka. Komsa MSc. Maria Fedina MSc. Arsalan Hashemi Lecture 12, Wednesday 2. 5. 2018 Dielectric Properties of Solids Magnetism Superconductivity

Magnetic properties • Response of materials to an external magnetic field – – Magnetic quantities, magnetism is quantum mechanics (home work) Quantum mechanical description Atomic diamagnetism, paramagnetism (lecture work) TODAY Response of free electron gas • Spontaneous magnetism Ferromagnetism and antiferromagnetism – – – – Exchange interaction, H 2 molecule, Heisenberg spin Hamiltonian Mean-field approximation for ferromagnetism of magnetic moments Spin waves (low-energy excitations) Free electron gas Stoner model for ferromagnetism of itinerant electrons Antiferromagnetism Domain structure 2

Diamagnetism and Paramagnetism, Atomic (Ionic) Structures Atom in many-electron state Ground state Hund’s rules Total angular momentum operator Perturbation energy in magnetic field [AM(31. 20)] (I) (II) (III) 3 (III) Per one atom

Atomic (ionic) diamagnetism, summary Diamagnetism: • Quantum mechanics: Closed shells, no permanent magnetic moments (Cu, Hg, Xe) • • Quantum mechanics: Magnetic moments remain until B 0 is removed Non-degenerate ground state well below excited states no temperature dependence • Quantum mechanics: cm, d ~10 -5, increases with the atom size 4

Paramagnetism (Elliott 7. 2. 4 ; A-M pp. 650 -659) 1 st-order correction to ground state Ions with open shells Degeneracy when B 0=0 If, and only if (Exercise) Degenerate ground state and permanent magnetic moment when B 0=0 Landé factor Wigner-Eckart theorem for vector operators [AM(31. 40)] (See AM Appendix P) Pertubation lifts the degeneracy [E(7. 163), AM(31. 39)] Direction of magnetic moment is quantized. Lowest states: [E(7. 165)] 5
![Paramagnetism of a magnetic gas Example Partition junction E7 167 169 Helmholtz free energy Paramagnetism of a magnetic gas Example Partition junction [E(7. 167, 169)] Helmholtz free energy](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-6.jpg)
Paramagnetism of a magnetic gas Example Partition junction [E(7. 167, 169)] Helmholtz free energy Curie law [E(7. 174)] 6 (Elliott 7. 2. 4. 2)
![Paramagnetism of a magnetic gas Curie law Elliott 7 2 4 2 E7 174 Paramagnetism of a magnetic gas Curie law (Elliott 7. 2. 4. 2) [E(7. 174)]](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-7.jpg)
Paramagnetism of a magnetic gas Curie law (Elliott 7. 2. 4. 2) [E(7. 174)] C=? Magnetic moment Landé factor Susceptibility Magnetic moment squared Cf. Exercise 2/6: Thermal energy - At room temperature - Rare earth ions in insulators (open 4 f shell): Model quantitatively OK. - Transition metal ions in insulators (open 3 d shell): Including crystal-field quenching (L=0), model quantitatively OK. 7

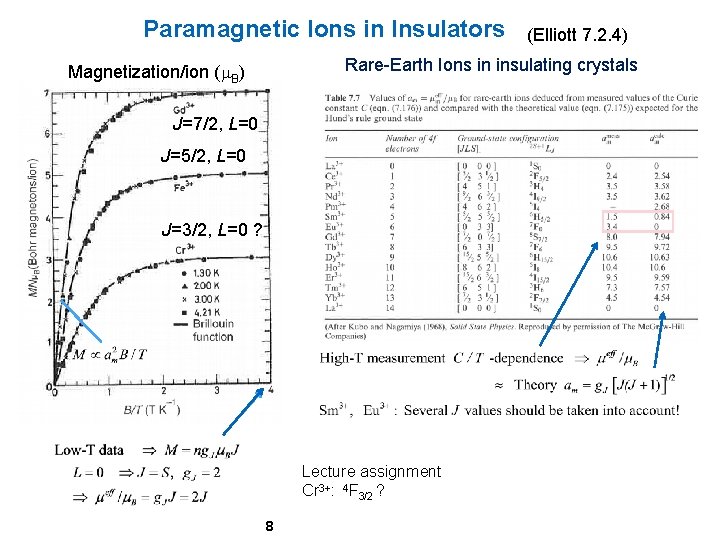
Paramagnetic Ions in Insulators (Elliott 7. 2. 4) Rare-Earth Ions in insulating crystals Magnetization/ion (m. B) J=7/2, L=0 J=5/2, L=0 J=3/2, L=0 ? Lecture assignment Cr 3+: 4 F 3/2 ? 8

Paramagnetic Ions in Insulators 3 d Transition Metal Ions in insulating crystals (Elliott 7. 2. 4) Crystal-Field Quenching d-electron – crystal-field interaction point symmetry < spherical L not a good quantum number (not a constant of motion) Lz averages to zero m. L is quenched, m. L = 0 S commutates with crystal field, m. S > 0 Where is the difference between 4 f and 3 d ions 4 f Ion radius: Large LS-interaction: Large Crystal-field effect: Small 9 3 d Small Large

Atomic (ionic) paramagnetism, summary • Quantum mechanics: Open shells (3 d, and 4 f), orbital and spin angular momenta permanent magnetic moments • • Quantum mechanics: Degenerate ground state when B 0 = 0 Zeeman splitting lifts the degeneracy • Statistical physics (Maxwell-Boltzmann) FH M cm, p ~ 1/T Curie law • Classical physics: Finite T causes disorder and decreases M • At room temperature: QM + MB: cm, p ~ 500 cm, d 10

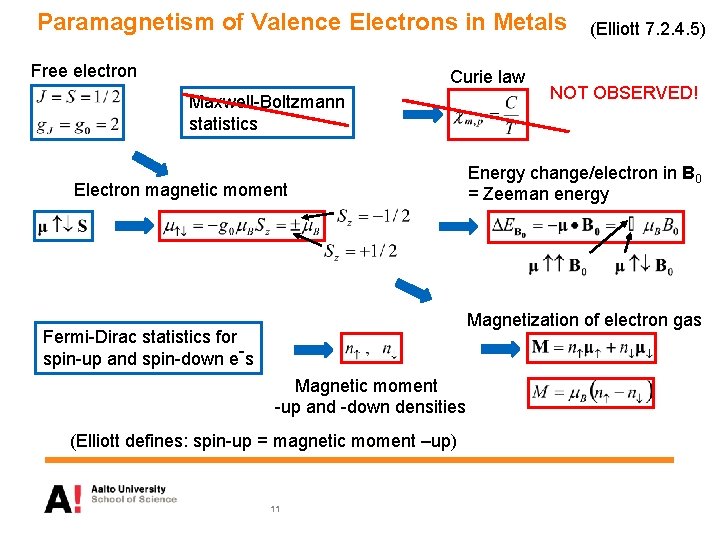
Paramagnetism of Valence Electrons in Metals Free electron Curie law Maxwell-Boltzmann statistics Electron magnetic moment (Elliott 7. 2. 4. 5) NOT OBSERVED! Energy change/electron in B 0 = Zeeman energy Magnetization of electron gas Fermi-Dirac statistics for spin-up and spin-down e-s Magnetic moment -up and -down densities (Elliott defines: spin-up = magnetic moment –up) 11

Paramagnetism of Valence Electrons in Metals Zeeman energy B 0=0 (Elliott 7. 2. 4. 5) Density Of States (DOS) B 0>0 Taylor [E(7. 186, 187)] 12
![Paramagnetism of Valence Electrons in Metals Total density Pauli paramagnetic susceptibility E7 188 E7 Paramagnetism of Valence Electrons in Metals Total density Pauli paramagnetic susceptibility [E(7. 188)] ~[E(7.](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-13.jpg)
Paramagnetism of Valence Electrons in Metals Total density Pauli paramagnetic susceptibility [E(7. 188)] ~[E(7. 189)] 13

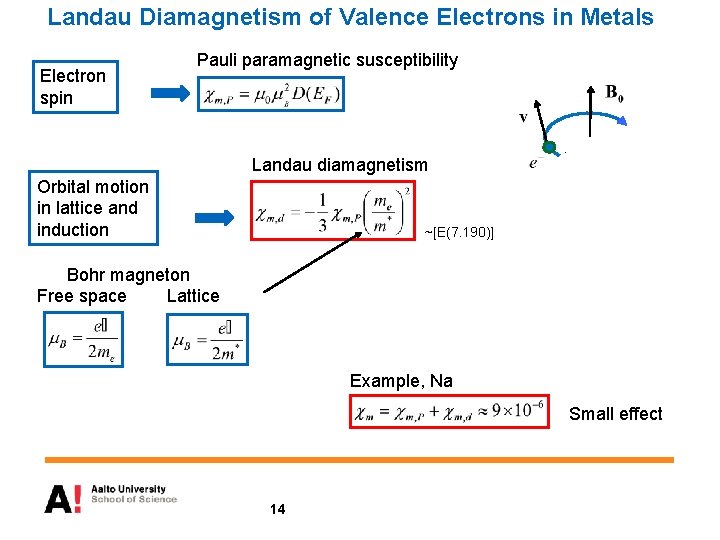
Landau Diamagnetism of Valence Electrons in Metals Electron spin Pauli paramagnetic susceptibility Landau diamagnetism Orbital motion in lattice and induction ~[E(7. 190)] Bohr magneton Free space Lattice Example, Na Small effect 14

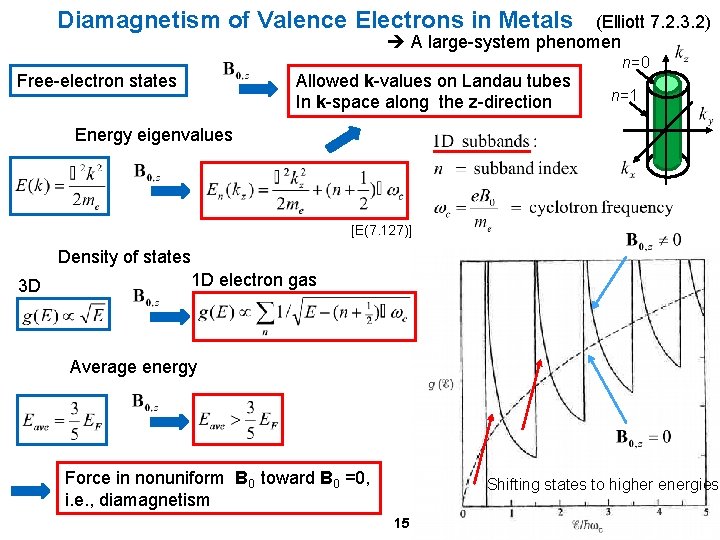
Diamagnetism of Valence Electrons in Metals (Elliott 7. 2. 3. 2) A large-system phenomen Free-electron states Allowed k-values on Landau tubes In k-space along the z-direction n=0 n=1 Energy eigenvalues [E(7. 127)] Density of states 1 D electron gas 3 D Average energy Force in nonuniform B 0 toward B 0 =0, i. e. , diamagnetism Shifting states to higher energies 15

Response of Free Electrons to an External Magnetic Field, Summary Pauli paramagnetism & Landau diamagnetism D(EF) Temperature independent Magnetic field Landau tubes in k-space (Cf. 1 D-DOS) Kinetic energy increases Diamagnetism 16

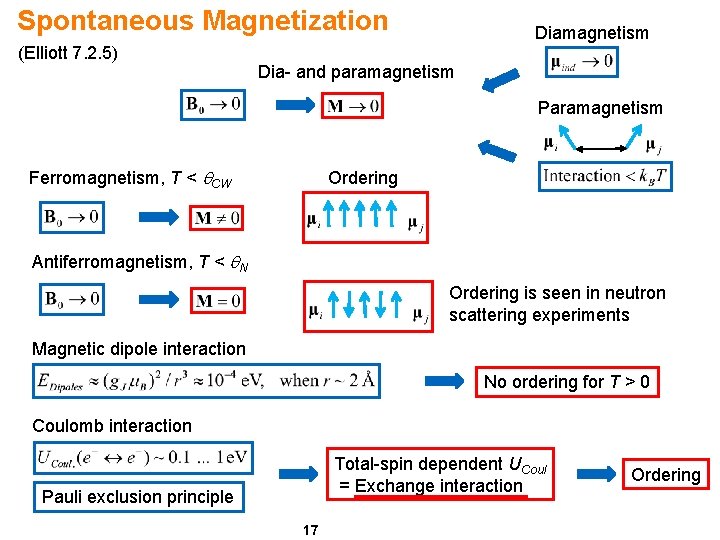
Spontaneous Magnetization (Elliott 7. 2. 5) Diamagnetism Dia- and paramagnetism Paramagnetism Ferromagnetism, T < q. CW Ordering Antiferromagnetism, T < q. N Ordering is seen in neutron scattering experiments Magnetic dipole interaction No ordering for T > 0 Coulomb interaction Total-spin dependent UCoul = Exchange interaction Pauli exclusion principle 17 Ordering

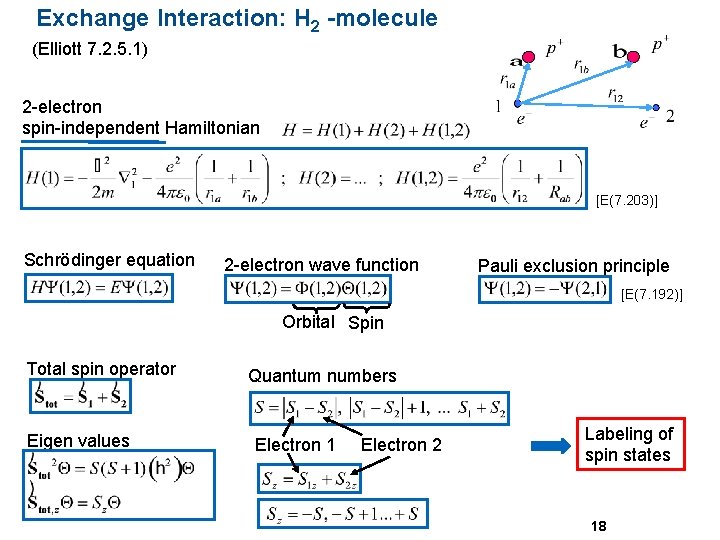
Exchange Interaction: H 2 -molecule (Elliott 7. 2. 5. 1) 2 -electron spin-independent Hamiltonian [E(7. 203)] Schrödinger equation 2 -electron wave function Pauli exclusion principle [E(7. 192)] Orbital Spin Total spin operator Eigen values Quantum numbers Electron 1 Electron 2 Labeling of spin states 18

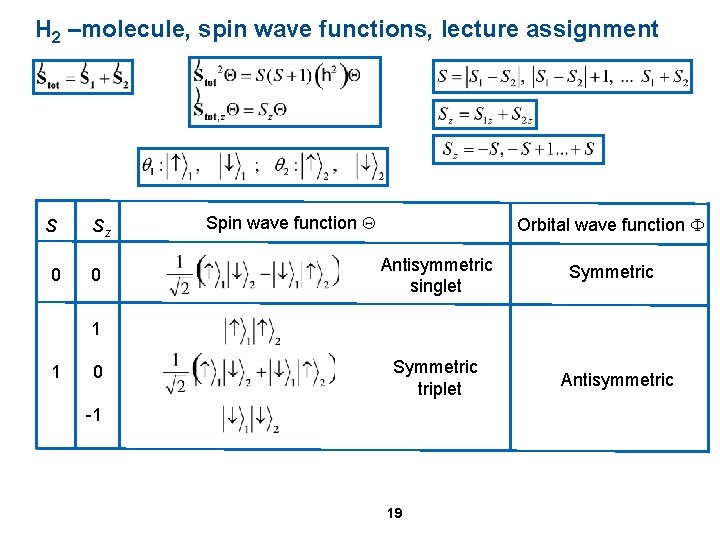
H 2 –molecule, spin wave functions, lecture assignment S Sz ? 0 1 ? 0 Spin wave function Q ? ? ? Orbital wave function F Antisymmetric singlet Symmetric triplet -1 19 Symmetric Antisymmetric

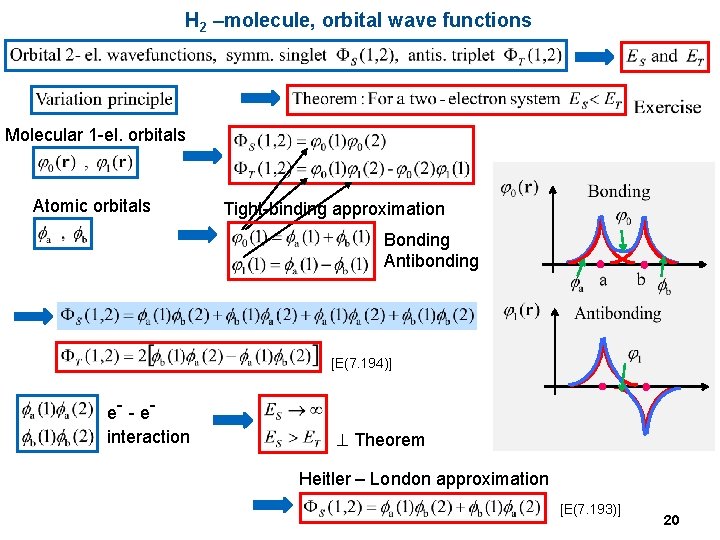
H 2 –molecule, orbital wave functions Molecular 1 -el. orbitals Atomic orbitals Tight-binding approximation Bonding Antibonding [E(7. 194)] e- - e interaction ^ Theorem Heitler – London approximation [E(7. 193)] 20
![Exchange Interaction H 2 molecule singlettriplet energy splitting Large Rab E7 208 0 Coulomb Exchange Interaction: H 2 –molecule, singlet-triplet energy splitting Large Rab [E(7. 208)] <0 Coulomb](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-21.jpg)
Exchange Interaction: H 2 –molecule, singlet-triplet energy splitting Large Rab [E(7. 208)] <0 Coulomb interaction Exchange integral Antiferromagnetic coupling EXCHANGE INTERACTION - Spin ordering, although H is spin independent - Strong as a Coulomb interaction - Easy to generalize to solids 21
![Heisenberg Spin Hamiltonian Generalization to solids Proof Exercise Parametric form E7 217 Solids Exchange Heisenberg Spin Hamiltonian Generalization to solids Proof Exercise Parametric form [E(7. 217)] Solids Exchange](https://slidetodoc.com/presentation_image/5c5cebcea3e894e2082c6911d9c8e821/image-22.jpg)
Heisenberg Spin Hamiltonian Generalization to solids Proof Exercise Parametric form [E(7. 217)] Solids Exchange constant J 22

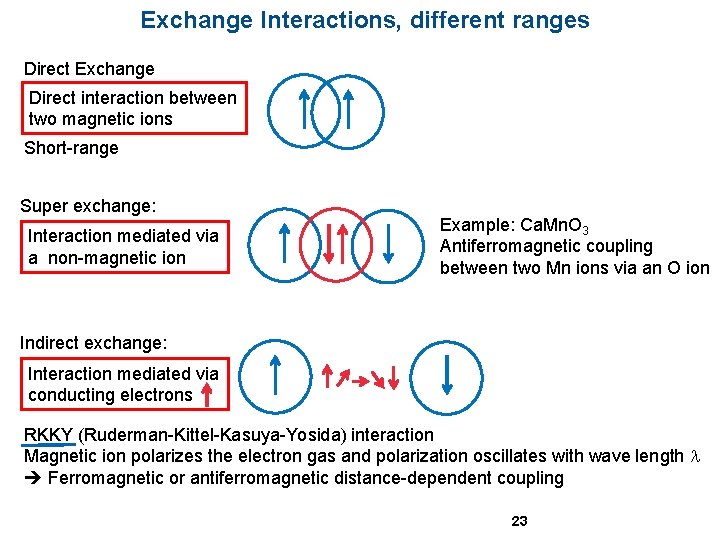
Exchange Interactions, different ranges Direct Exchange Direct interaction between two magnetic ions Short-range Super exchange: Interaction mediated via a non-magnetic ion Example: Ca. Mn. O 3 Antiferromagnetic coupling between two Mn ions via an O ion Indirect exchange: Interaction mediated via conducting electrons RKKY (Ruderman-Kittel-Kasuya-Yosida) interaction Magnetic ion polarizes the electron gas and polarization oscillates with wave length l Ferromagnetic or antiferromagnetic distance-dependent coupling 23

Exchange Interaction Spontaneous Magnetization Summary and Preface Pauli exclusion principle, spin independent Hamiltonian, strong Coulombic interaction Spontaneous magnetization Heisenberg spin Hamiltonian bulk systems, mean-field theory of temperaturedependent magnetization and critical temperature Free electron gas Itinerant (band) magnetism, Stoner model equivalent to meanfield theory 24

Magnetic properties • Response of materials to an external magnetic field – – Magnetic quantities, magnetism is quantum mechanics (home work) Quantum mechanical description Atomic diamagnetism, paramagnetism (lecture work) Response of free electron gas • Spontaneous magnetism Ferromagnetism and antiferromagnetism – – – – Exchange interaction, H 2 molecule, Heisenberg spin Hamiltonian Mean-field approximation for ferromagnetism of magnetic moments Spin waves (low-energy excitations) The next lecture Free electron gas Stoner model for ferromagnetism of itinerant electrons Antiferromagnetism Domain structure 25

Paramagnetism of Valence Electrons in Metals, Summary NOT OBSERVED! (Zeeman energy) 26