PHYS 172 Modern Mechanics Lecture 24 The Boltzmann

PHYS 172: Modern Mechanics Lecture 24 – The Boltzmann Distribution Summer 2012 Read 12. 7

Summary: Foundations Einstein Model of Solids # microstates (N oscillators, q quanta) Fundamental assumption of statistical mechanics Over time, an isolated system in a given macrostate (total energy) is equally likely to be found in any of its microstates (microscopic distribution of energy).

Summary: Entropy and Temperature If the initial state is not the most probable, energy is exchanged until the most probable distribution is reached.
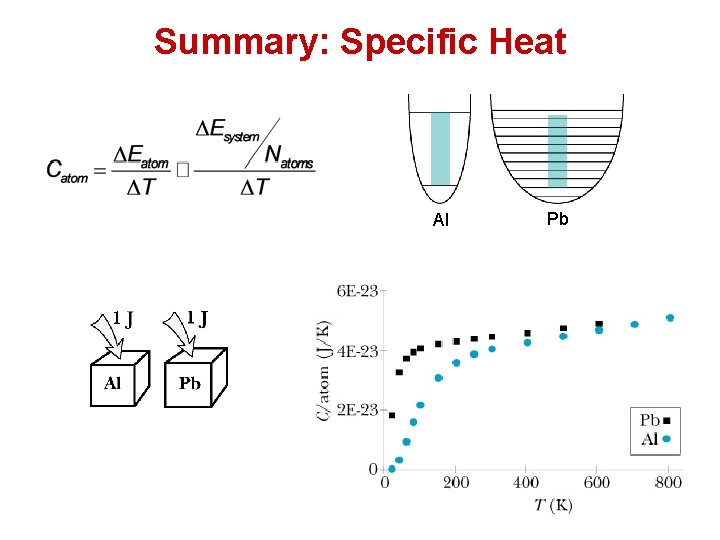
Summary: Specific Heat Al Pb

Today: The Boltzmann Distribution Derivation: Choose Your System Wisely Many Systems or Many Observations? Simple Applications: Probabilities for Atomic Excitations Block of Lead Biological Physics
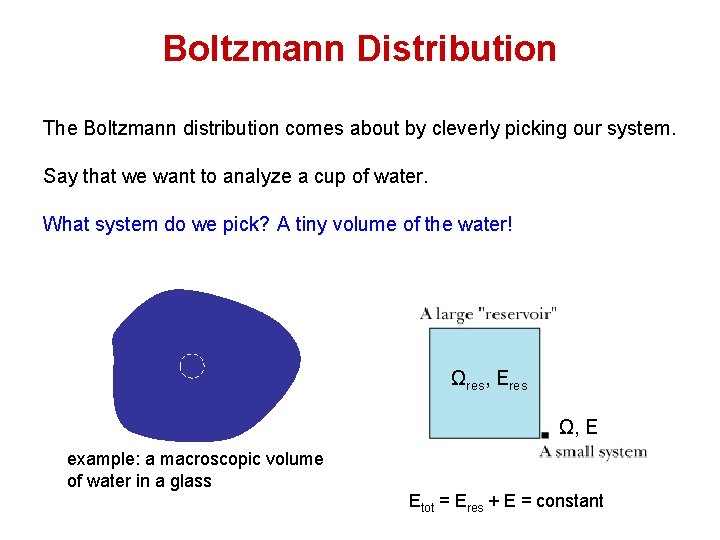
Boltzmann Distribution The Boltzmann distribution comes about by cleverly picking our system. Say that we want to analyze a cup of water. What system do we pick? A tiny volume of the water! Ωres, Eres Ω, E example: a macroscopic volume of water in a glass Etot = Eres + E = constant

CLICKER QUESTION For a macrostate of 4 quanta distributed among 2 atoms (6 oscillators), what is the probability that the first atom has 1 quanta? A) 1/2 B) 3/30 D) 3/126 E) 60/126 atom 1 (3 oscillators) atom 2 (3 oscillators) C) 30/126 q 1 q 2 Ω 1 Ω 2 0 4 1 15 15 1 3 3 10 30 2 2 6 6 36 3 1 10 3 30 4 0 15 1 15 Total = 126

Recall the Pb Nanoparticle (3 atoms) from Ch 11, p 392 -393: A q Ω ln Ω S 4 465 6. 20 5 1287 6 3003 B C q T(K) 6. 20 k. B 4 -5 58. 9 7. 16 k. B 5 62. 5 8. 01 k. B 5 -6 66. 6 What if we wanted to know what is happening on one of the three atoms? For example, with q=6, how often is atom A found with q. A=2?
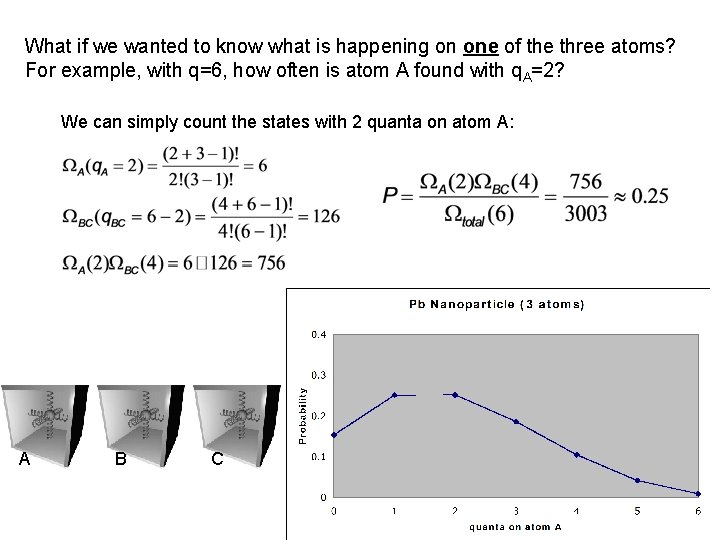
What if we wanted to know what is happening on one of the three atoms? For example, with q=6, how often is atom A found with q. A=2? We can simply count the states with 2 quanta on atom A: A B C
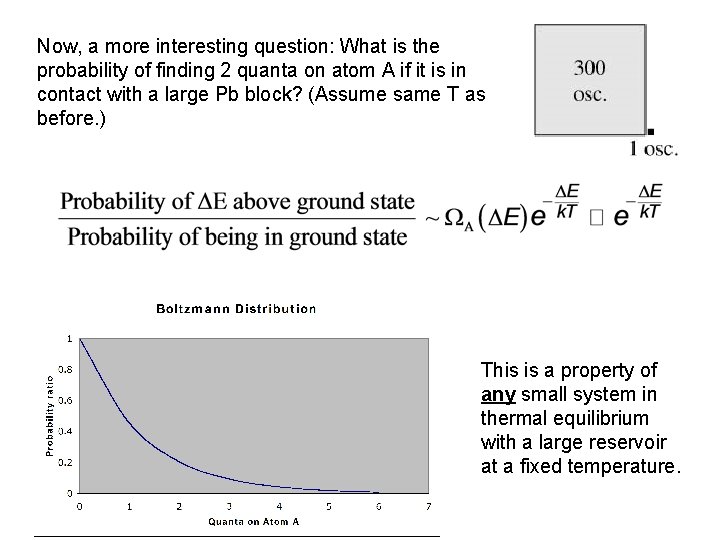
Now, a more interesting question: What is the probability of finding 2 quanta on atom A if it is in contact with a large Pb block? (Assume same T as before. ) This is a property of any small system in thermal equilibrium with a large reservoir at a fixed temperature.
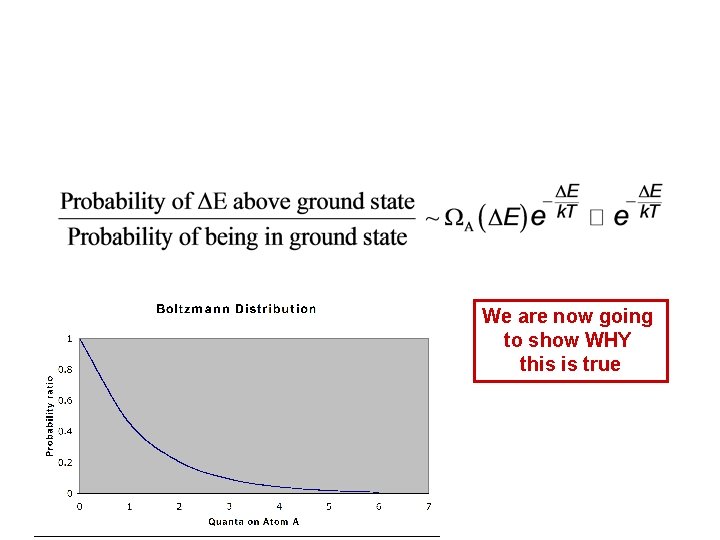
We are now going to show WHY this is true

VIEWER WARNING! The following slides may contain Derivations or Derivation Byproducts. Do not operate heavy machinery while viewing the following slides. Also, do not go swimming for at least one hour after viewing these slides. Derivations and Derivation Byproducts are known to the State of Indiana to have the following possible side effects: Furrowed Brows, Increased Logical Abilities, and in rare cases, Brain Explosions.
Probabilities “Reservoir” A ginormous system with a constant temperature tiny system WARNING! This slide may contain Derivations or Derivation Byproducts.
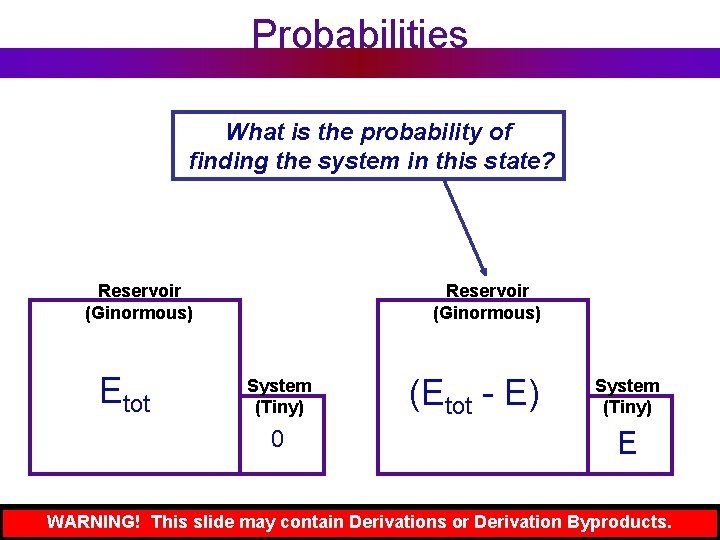
Probabilities What is the probability of finding the system in this state? Reservoir (Ginormous) Etot Reservoir (Ginormous) System (Tiny) 0 (Etot - E) System (Tiny) E WARNING! This slide may contain Derivations or Derivation Byproducts.
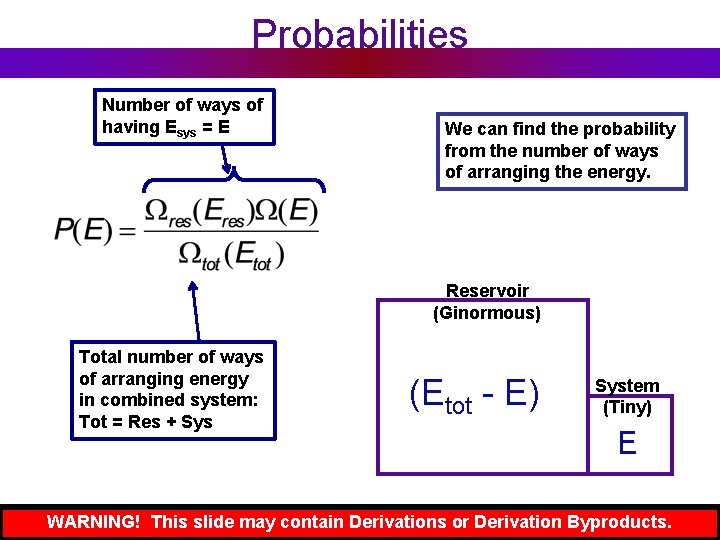
Probabilities Number of ways of having Esys = E We can find the probability from the number of ways of arranging the energy. Reservoir (Ginormous) Total number of ways of arranging energy in combined system: Tot = Res + Sys (Etot - E) System (Tiny) E WARNING! This slide may contain Derivations or Derivation Byproducts.

Now Do Some Math. . . We can find the probability from the number of ways of arranging the energy. WARNING! This slide may contain Derivations or Derivation Byproducts.

More Math. . . Use a Taylor Expansion for the Ginormous Reservoir: (E << Eres ) Spot The Temperature! WARNING! This slide may contain Derivations or Derivation Byproducts.

More Math. . . Use a Taylor Expansion for the Ginormous Reservoir: (E << Eres ) Spot The Temperature! WARNING! This slide may contain Derivations or Derivation Byproducts.

More Math. . . Exponentiate Everything. . . BOLTZMANN FACTOR Very very important! WARNING! This slide may contain Derivations or Derivation Byproducts.
VIEWER WARNING! The previous slides did contain Derivations and Derivation Byproducts. So, you should not go swimming for at least one hour after class. The Derivation is now OVER. We now return you to your regularly scheduled physics learning experience. Also known as lecture.

Boltzmann Distribution The probability of finding energy E in a small system in contact with a large reservoir is We just showed WHY this is true The exponential part, e-E/k. T, is called the “Boltzmann factor. ” Ω(E) is the number of microstates in the small system at energy E. In many circumstances, Ω(E) changes so slowly compared to e -E/k. T that it is essentially constant: Ωres, Eres Ω, E

Boltzmann Distribution The Boltzmann distribution comes about by cleverly picking our system. Say that we want to analyze a cup of water. What system do we pick? A tiny volume of the water! Example: a macroscopic volume of water in a glass of water Ωres, Eres Ω, E Etot = Eres + E = constant

Application: Atomic Excitations How likely is an atom to be in 1 st excited, compared to odds of being in ground state? NOTE: k. T at room temp = 1/40 e. V. For the above atom, odds of being in first excited state are Typical atomic energy gaps are big compared to room temp. A room-temperature box of neon doesn’t glow! (Unless you add energy).

Application: Block of Lead T=300 K at room temperature BOLTZMANN FACTOR Very very important! Think of adding q quanta of energy to one lead atom. . . At room temperature, the Boltzmann factors for exciting vibrations are: Most probable state Watch out! These are relative probabilities

Application: Block of Lead T=300 K at room temperature Think of adding q quanta of vibrational energy to one lead atom. . . The actual probabilities use a normalization factor “Z” (partition function). Most likely to find the vibrations are excited. On average, q=5.

Today: The Boltzmann Distribution Derivation: Choose Your System Wisely Many Systems or Many Observations? Simple Applications: Probabilities for Atomic Excitations Block of Lead Biological Physics

Next Time: Boltzmann Applications Speed Distribution in a Gas Energy Equipartition and Specific Heat Pressure and the Ideal Gas Law
- Slides: 27