PHY 430 Lecture 1 Physical Quantities Units Base

PHY 430 – Lecture 1 Physical Quantities & Units Base & Derived Quantities & SI Units Dimensional Analysis Unit Conversion 1

Base quantity & Derived quantity Base quantity Ø Derived quantity Ø A quantity which is not Ø A quantity which is a a combination of other combination of two or physical quantities. more physical Ø Must be defined in quantities. terms of a standard. Ø Units for derived Ø Units for base quantities are derived quantities are base units Ø 2

3

1 meter Ø The meter is the length of the path traveled by light in vacuum during a time interval of 1/299, 792, 458 of a second. 4

1 kilogram Ø A particular platinumiridium cylinder kept at the International Bureau of Weights and Measures near Paris. 5

1 second Ø One second is defined as the time required for 9, 192, 631, 770 periods of radiation emitted by cesium atoms. 6
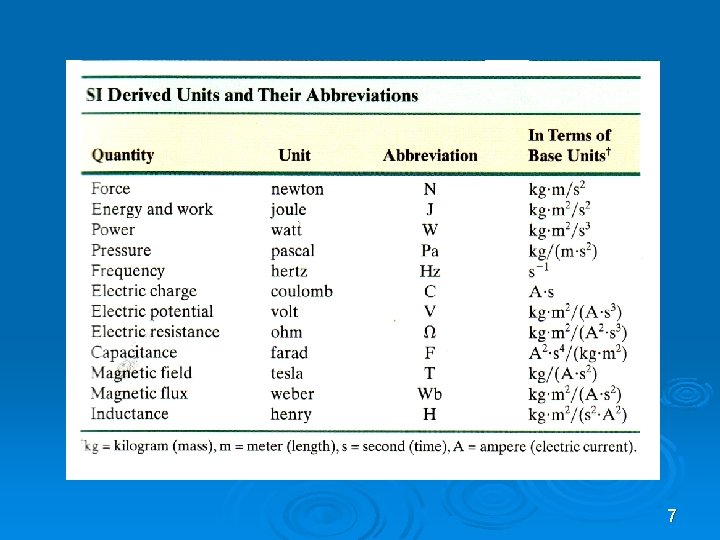
7
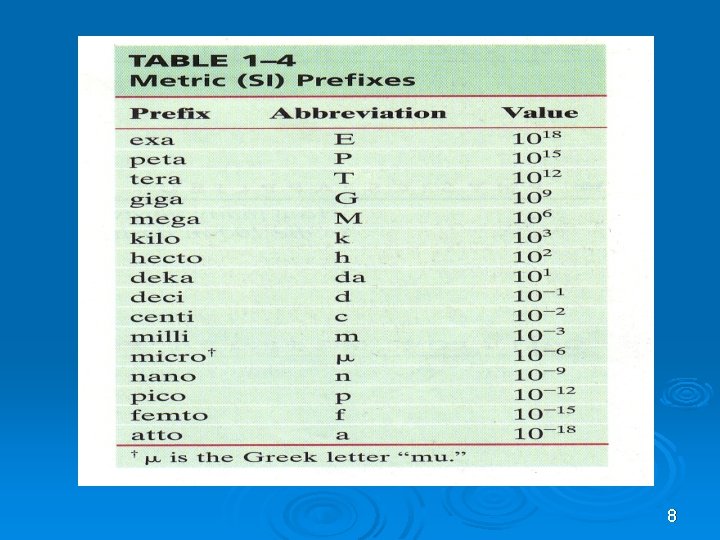
8
9

Effective technique of Unit Conversion 1. Write the unit in traditional form and not the index form. eg. Remember or look up the conversion factor. 3. Multiply the initial quantity with the proper conversion factor so that only the required units are left, the others cancelled off. 2. 10

Conversion factor to remember Ø 1 km = 1000 m Ø 1 m = 100 cm Ø 1 cm = 10 mm Ø 1 kg = 1000 g Ø 1 h = 60 min Ø 1 min = 60 s Ø 1 h = 3600 s Ø Remember the values for the prefixes. Ø The rest – look up the conversion table 11

Unit Conversion Ø Convert Ø (a) 110 km/h to m/s Ø (b) 340 m/s to km/h Ø (c) 300 m 2 to cm 2 Ø (d) 20 m 3 to cm 3 Ø (e) 1000 kg/m 3 to g/cm 3 Ø (f) 15 nm to m Ø (g) 25 A to A Ø (h) 240 MW to k. W 12

Dimensional Analysis Ø Dimensions of a Ø Add & subtract quantity are base quantities only if quantities or base they have the units that make up same dimensions the quantity Ø [LHS] = [RHS] Ø Dimensional Analysis is a useful technique to check if a relationship is incorrect 13
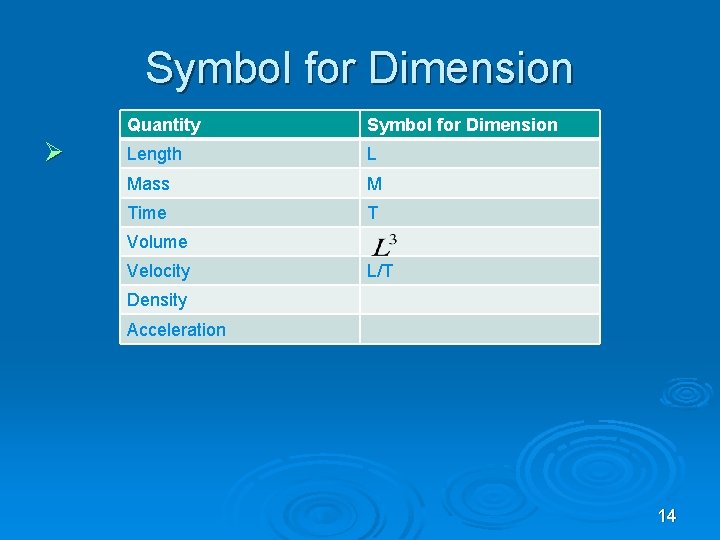
Symbol for Dimension Ø Quantity Symbol for Dimension Length L Mass M Time T Volume Velocity L/T Density Acceleration 14

Eg. 1 Dimensional Analysis Ø Show that v = u + at is dimensionally correct. 15

Significant Figures (sf) Rule Examples 1. All non zero digits in a number are sf 1. (i) 3421 : 4 sf (ii) 62. 5 : 3 sf 2. Zero in between two non zero digits are sf 2. (i) 503 : 3 sf (ii) 1. 006 : 4 sf 3. For any whole number, zero at the end of a number can be a sf or not a sf. It depends on the of precision of the reading. 3. (i) 63 000 : 2 sf if the precision is to the nearest thousand (ii) 63 000 : 3 sf if the precision is to the nearest hundred 16

Significant Figures (sf) Rule 4. For a decimal number less than 1, zero placed before any non-zero digit is not a sf. 5. For a decimal number, zero placed after a nonzero digit is a sf. Examples 4. (i) 0. 0028 : 2 sf (ii) 0. 0902 : 3 sf 5. (i) 7. 40 : 3 sf (ii) 0. 020 : 2 sf 17

Significant Figures : multiplication & division Ø The final results of multiplication or division should have only as many digits as the number with the least number of significant figures used in the calculation. Ø Eg. 11. 3 x 6. 8 = 76. 84 = 77 ( round off to 2 sf) 18
- Slides: 18