Photosynthesis UNIT 1 BIOINORGANIC CHEMISTRY Prof Sartaj Tabassum

Photosynthesis UNIT -1 BIOINORGANIC CHEMISTRY Prof. Sartaj Tabassum Department of Chemistry Aligarh Muslim University tsartaj 62@yahoo. com Mobile: 9358255791
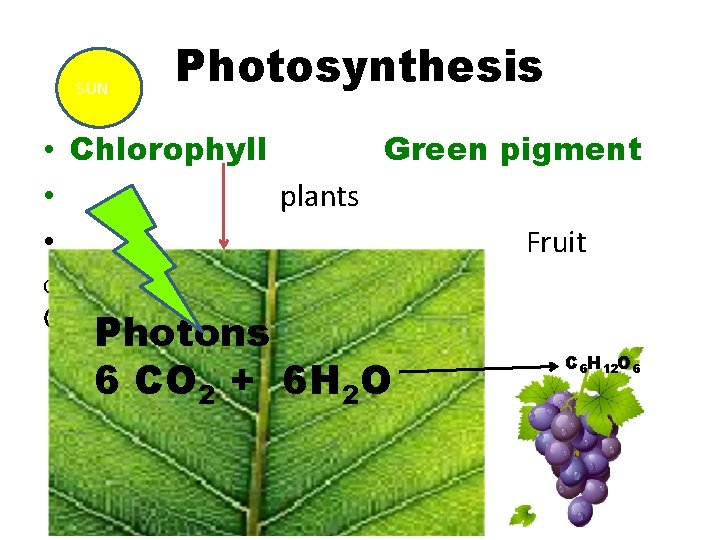
SUN Photosynthesis • Chlorophyll Green pigment • plants • Fruit Glucose GLUCOSE Photons 6 CO 2 + 6 H 2 O C 6 H 12 O 6


Chlorophyll pigment have Mg++ in the center of the biomolecule harvest energy (Photons) by absorbing blue light 420 nm and red 660 nm

• During the fall, the green chlorophyll pigments are greatly reduced revealing the other pigments. Carotenoids are pigments that are either red or yellow.
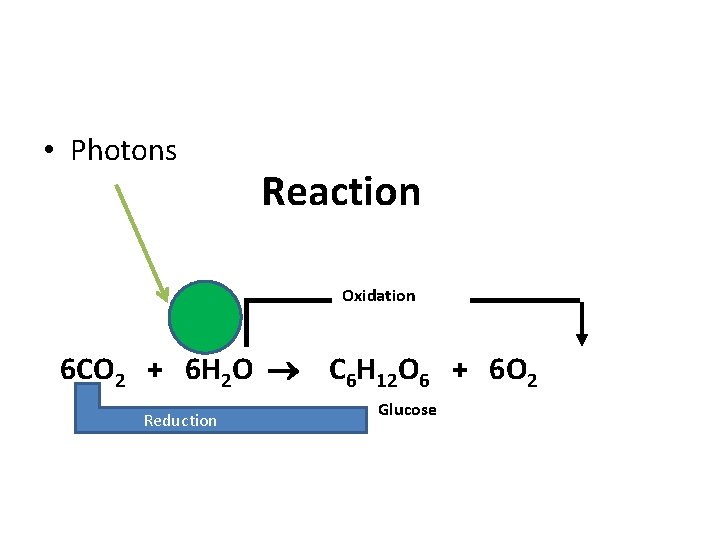
• Photons Reaction Oxidation 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 Reduction Glucose
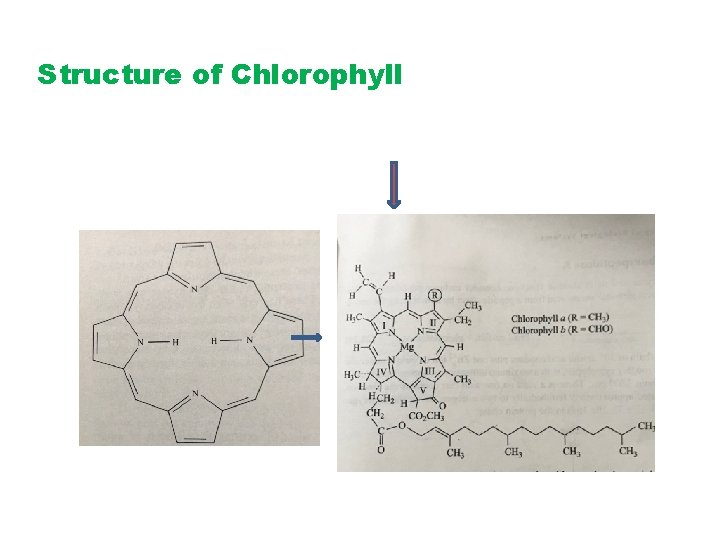
Structure of Chlorophyll
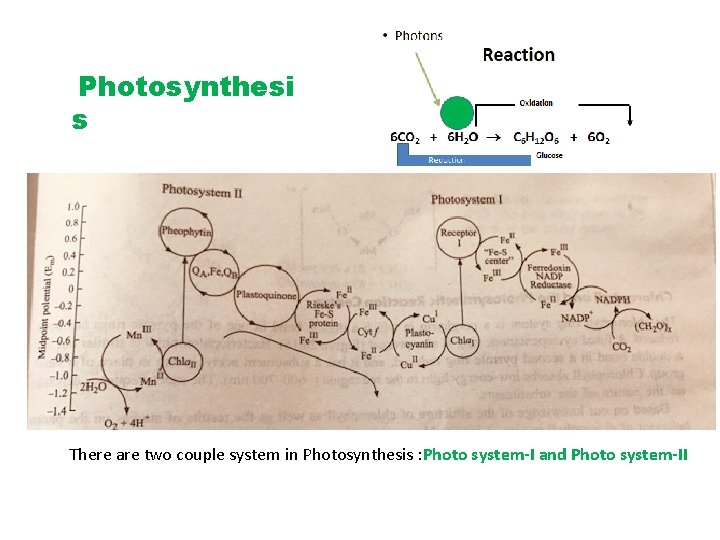
Photosynthesi s There are two couple system in Photosynthesis : Photo system-I and Photo system-II
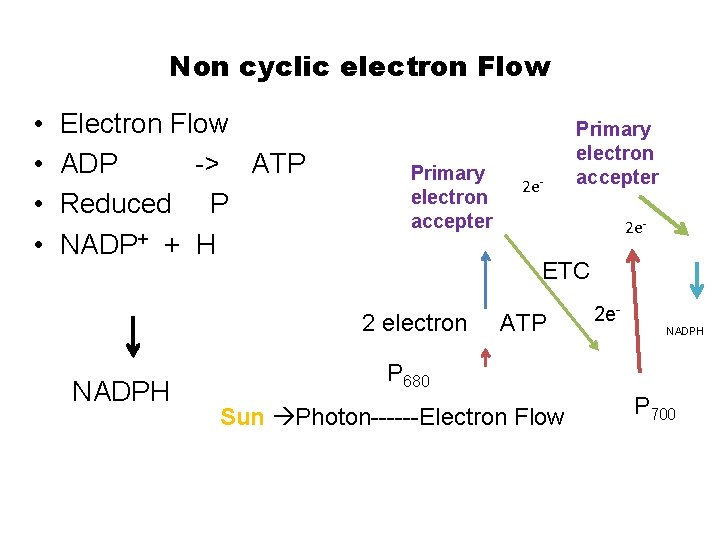
Non cyclic electron Flow • • Electron Flow ADP -> ATP Reduced P NADP+ + H Primary electron accepter 2 e- ETC 2 electron NADPH 2 e- Primary electron accepter ATP 2 e. NADPH P 680 Sun Photon------Electron Flow P 700

Photon Light harvesting by clusters antenna like rod of Chlorophyll • PS-I and PS-II have two different type of Chlorophyll trapped energy of the photon. • Product of PS-I is reduced carbon • PS-II primary product energy in the form of ATP with oxygen chemical by product • In addition of the chlorophyll, PS-I and PS-II have other pigment P 680 and P 700
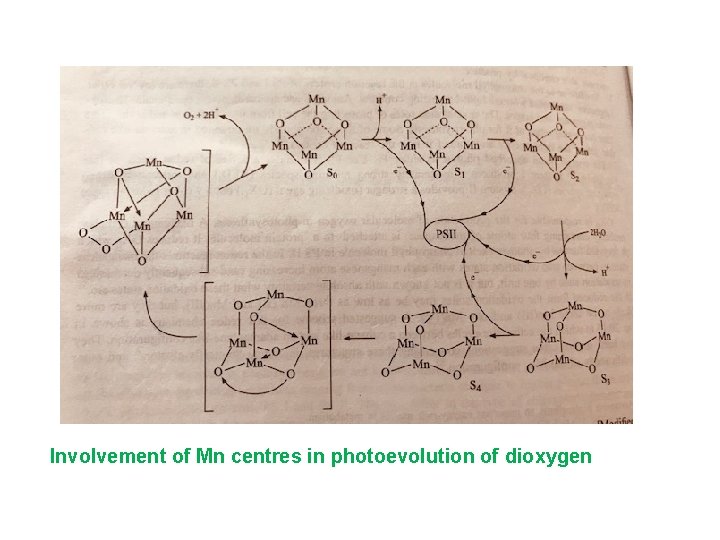
Involvement of Mn centres in photoevolution of dioxygen

• PS-I and PS-II absorb energy to initiate several redox reaction • PS-I have strong Reducing Species (REDI)strong oxidising (OXl) • PS-II have strong oxidising (OX II) weaker Reducing (REDII) • In redox reaction Mn is present in Mn(II) -Mn(III) and Mn(III)-Mn(IV).

Important photoactive redox centres to be studied in next coming lectures………. • • • Fe-S centers, Ferredoxin Reieske’s center Fe-S , Fe(II)-Fe(II) Cytochrome Fe(III)-Fe(II) Mn cluster
- Slides: 13