Photosynthesis AGRI 6203 Pathway of Electron Flow the

















- Slides: 30

Photosynthesis AGRI 6203

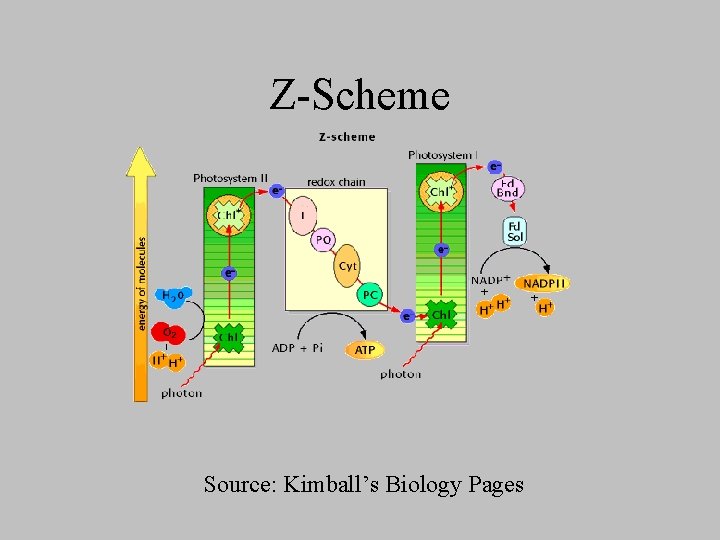
Pathway of Electron Flow: the Zig Zag Scheme • Raise energy level of electrons derived from water to the energy level required to reduce NADP+ to NADPH. – Each electron must be boosted twice • ATP production is coupled with electron flow

Z-Scheme Source: Kimball’s Biology Pages

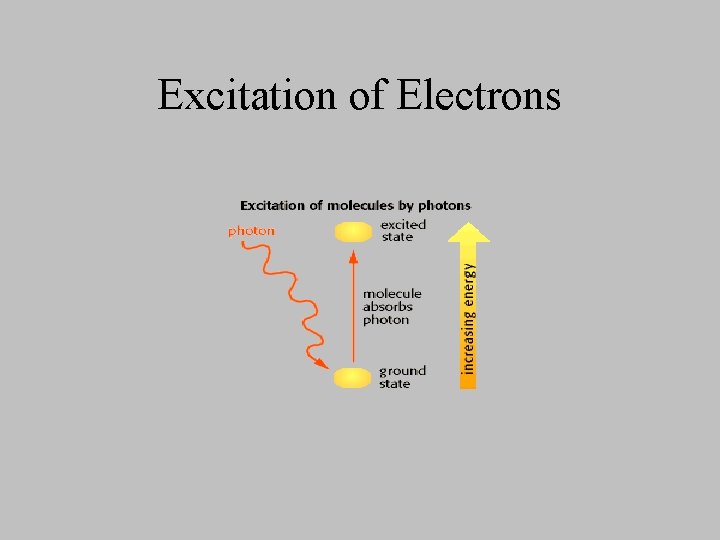
Excitation of Electrons

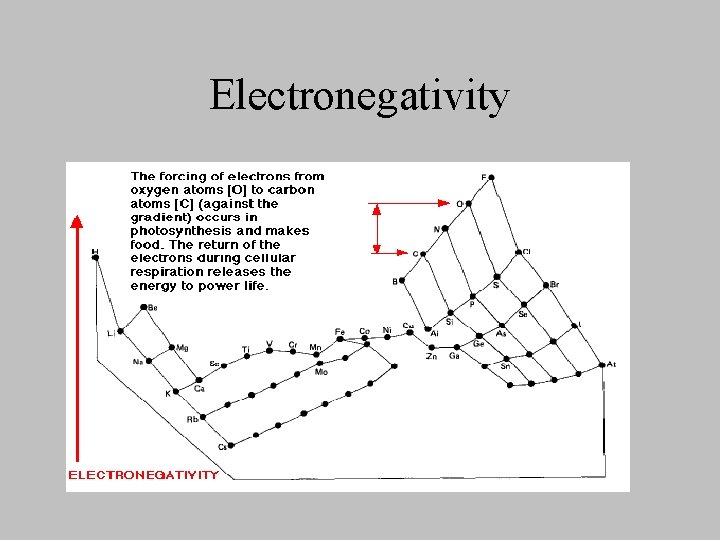
Redox Potentials (E) • Measure (volts) of a an affinity of a substance for electrons - its electronegativity- compared with hydrogen (which is set a 0) • positive redox potentials – capable of oxidizing – substance more electronegative than H (+E) • negative redox potentials – capable of reducing – substances less electronegative than H (-E)

Electronegativity

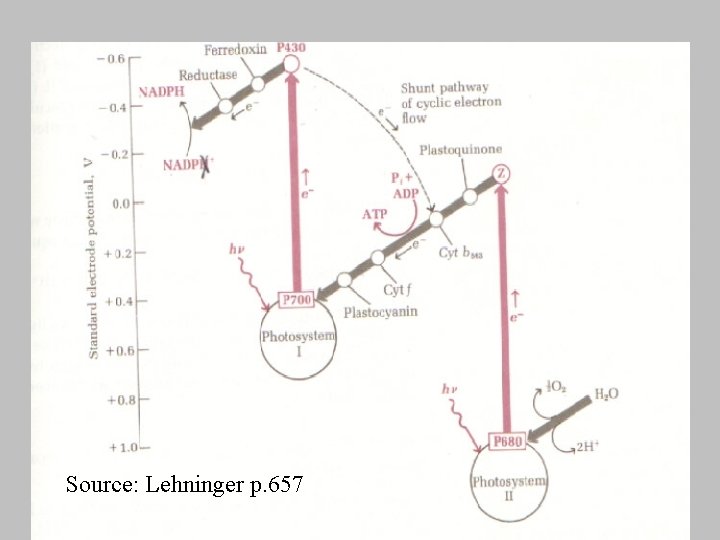
Source: Lehninger p. 657

“Downhill” flow of electrons • Free energy is released • - G

“Uphill” flow of electrons • Input of free energy • + G

E • Greater the E between two substances, the greater the vigor with which electrons will flow spontaneously from the less positive to the more positive substance

Available free energy • G = -n(23. 062 kcal) ( E) – n = number of electrons transferred – 23. 0672 = amount of energy released when one electron passes through a potential drop of 1 volt

Synthesis of glucose by Photosynthesis • 24 electrons must be removed from water – water redox potential = +0. 82 v – must be pumped “uphill” to – carbon atoms which they partially reduce to carbohydrate with a redox potential = -0. 42 v – difference is 1. 24 v ( E), so • G = -24(23. 062 kcal) (1. 24) = +686 kcal

Light energy causes electrons to flow uphill • Chlorophyll absorbs light and the electron in its structure is “boosted” • excited chlorophyll migrates to the reaction center of the photosystem – causes an electron to acquire a large amount of energy • “hot” electron is expelled from the reaction center and accepted by the first electron carrier

• First electron carrier becomes reduced and the reaction center has become _____ – oxidized reaction center now has an “electron hole” • energy-rich electron has high reducing “pressure” – passes to NADP+ – must be sufficiently negative to reduce NADP+

Filling the electron holes • PS I electron hole is filled by PSII • PSII electron hole is filled by water – 2 H 2 O -----> 4 H+ 4 e- = O 2 • electron flow from water to NADP • 2 H 2 O + 2 _______ ---light---> O 2 + _____ + 2 H+

Energy Profile of Photosynthetic Electron Transport • Transfer processes occurring the upward direction require energy • downward flow of electrons proceed with a loss of free energy • electrons flow from water (+0. 82 V) to NADP (-0. 32 V).

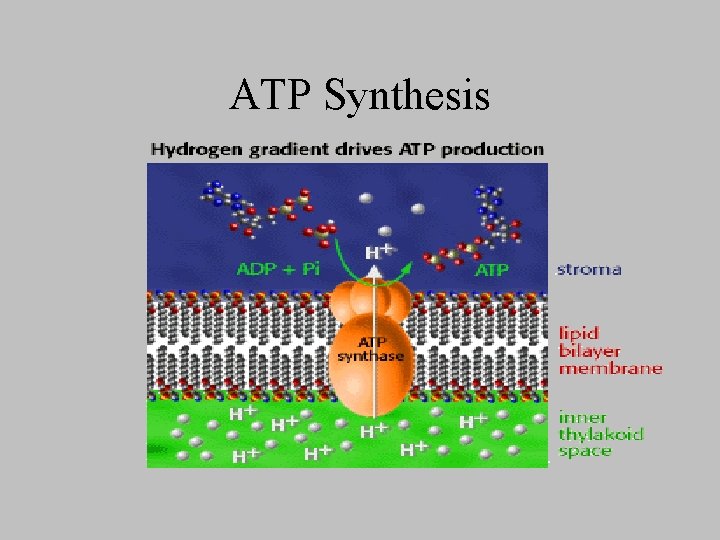
Photosynthetic Phosphorylation • ADP to ATP is coupled with the energy released as high-energy electrons flow down the photosynthetic electron-transport chain from excited photosystem ____ to the electron holes in photosystem ___. • One ATP is formed per pair of electrons

ATP Synthesis

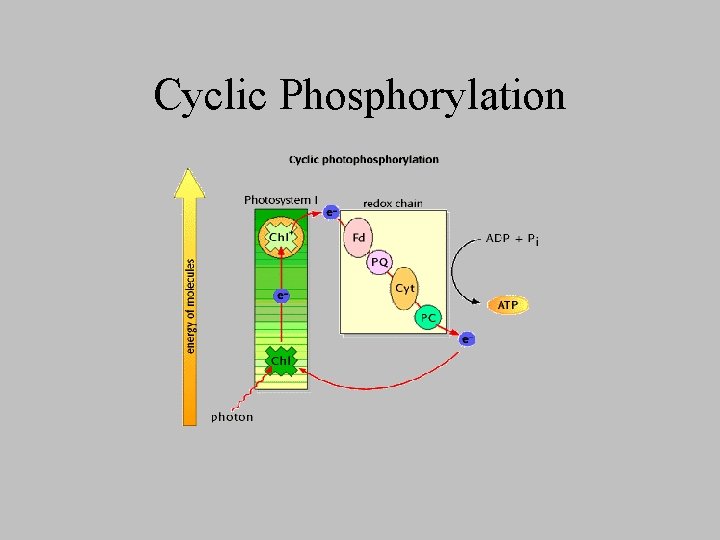
Cyclic Phosphorylation • Involves only PSI, cyclic because – electron boosted to P 430 by illumination of PSI, instead of passing to NADP+, flows back into the electron hole of PSI • overall reaction – Pi + ADP + light energy ---> ATP + H 20 • Why? – light reactions: 1: 1 ATP: NADPH formed – dark reactions: 3: 2 ATP: NADPH consumed

Cyclic Phosphorylation

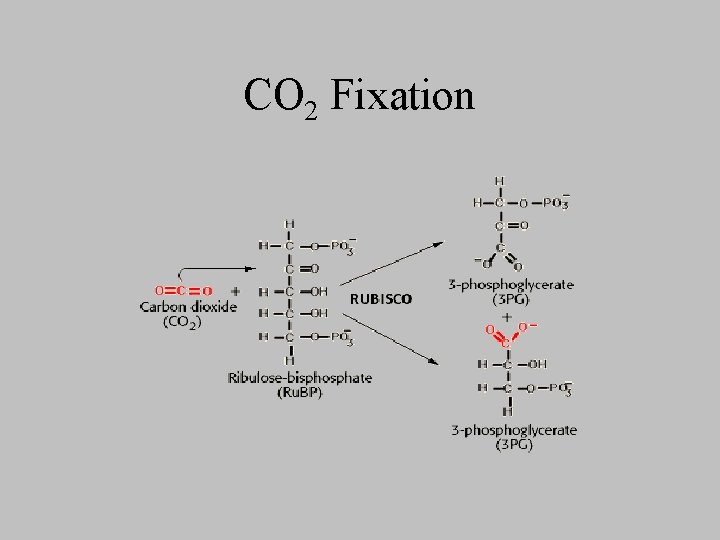
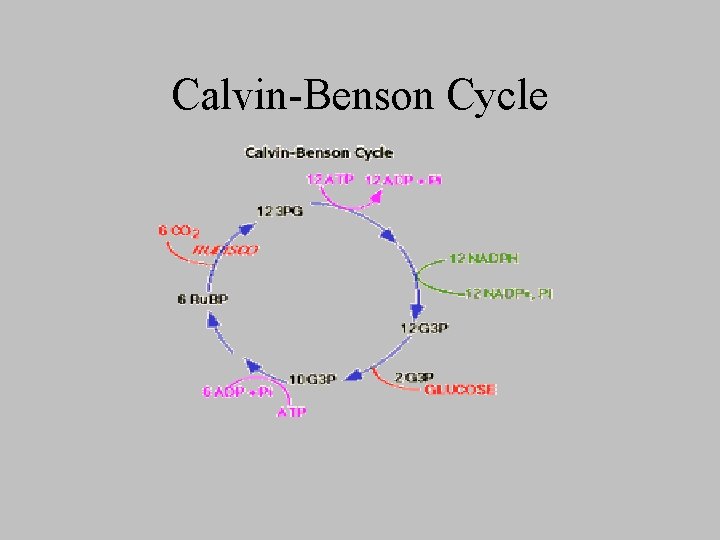
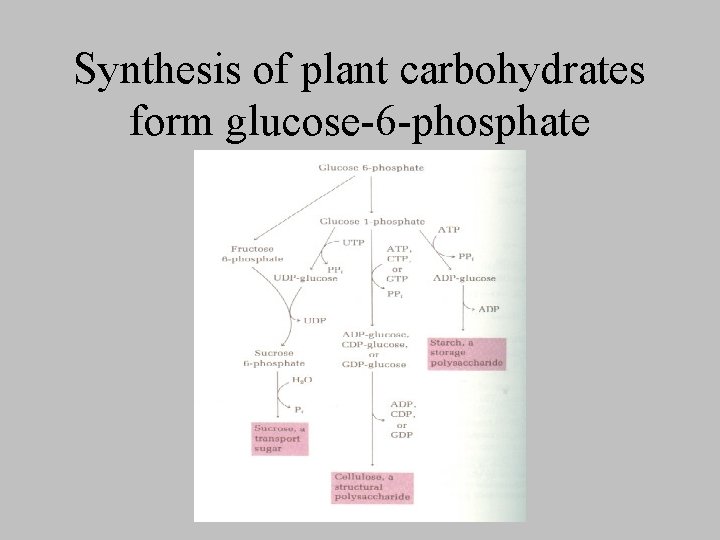
Photosynthetic formation of glucose • Generation of glucose and other carbs – from ____and _____, – at the expense of _______ and ____ produced in the light reactions • overall equation • What prevents animals from being able to carry out net conversion of CO 2 to glucose?

CO 2 Fixation

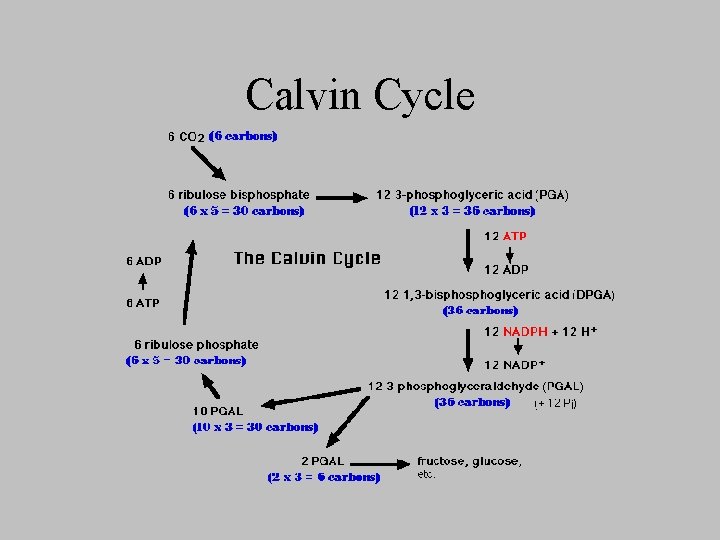
Calvin-Benson Cycle

Calvin Cycle

Synthesis of plant carbohydrates form glucose-6 -phosphate

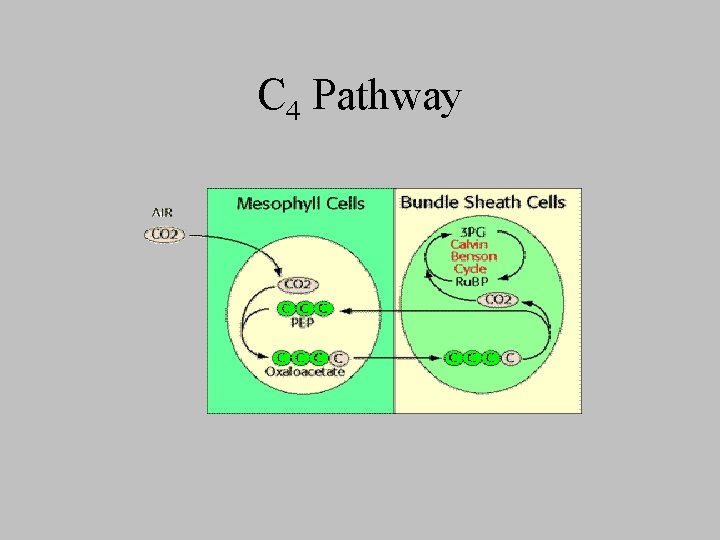
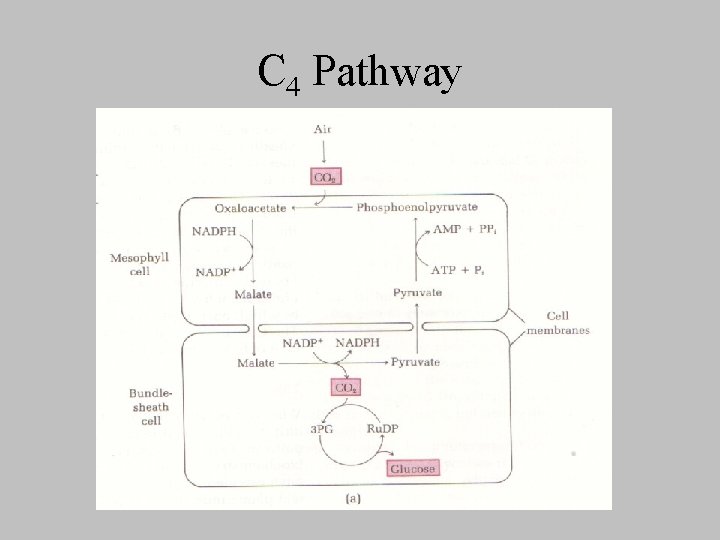
C 4 Plants • Tropics • all plant ultimately use the C 3 pathway • C 3 pathway is preceded by preliminary fixation of CO 2 into oxaloacetate • phosphoenopyruvate caboxylase – OAA produced

C 4 Pathway

C 4 Pathway

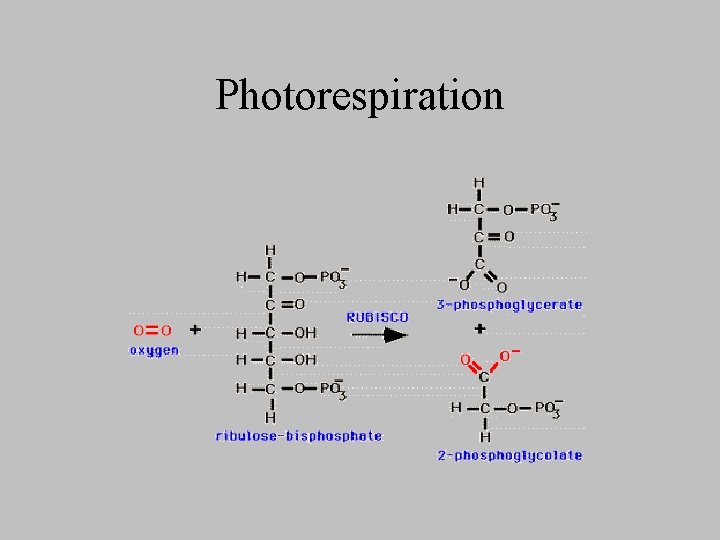
Photorespiration

Sources of Images Kimball’s Biology Pages Principles of Biochemistry, 1982, Lehninger,