Photon or Planck Statistics Blackbody Radiation Experiments on

Photon (or Planck) Statistics

Blackbody Radiation • Experiments on EM Radiation from a Hot Body: “Black Body Radiation” • Energy loss of a hot body is attributable to the emission of electromagnetic waves from the body. • The distribution of the energy flux over the wavelength spectrum does not depend on the nature of the body but does depend on its temperature. In Quantum Electrodynamics: EM radiation is regarded as a photon gas.
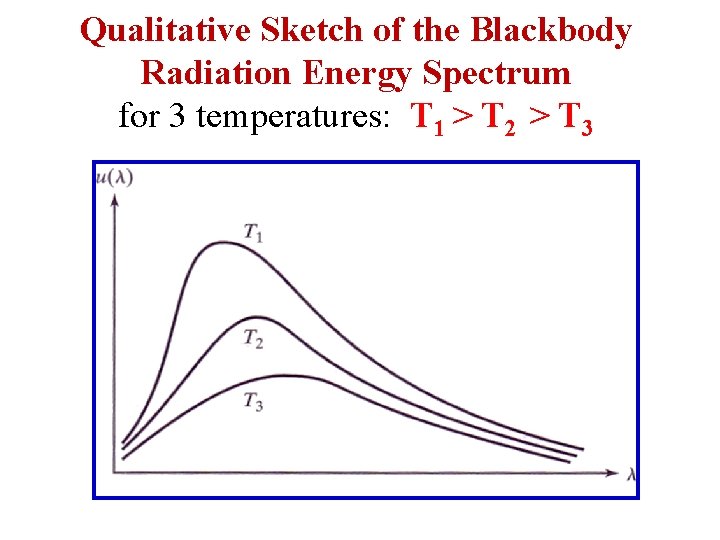
Qualitative Sketch of the Blackbody Radiation Energy Spectrum for 3 temperatures: T 1 > T 2 > T 3

• Photons emitted by an atomic energy level can be absorbed at another, so The total number of photons is not constant, i. e. ΣNJ = N does not apply • Lack of Photon Number Conservation can be shown to imply that the Chemical Potential in the Bose. Einstein Distribution = Zero for Photons. So, photon = 0 • Photons are Bosons of Spin 1 & they obey Bose-Einstein Statistics:

• For a continuous spectrum of energy • The energy of each photon is = hν, • So: • Definition: f(ν)dν number of photon states with frequency v in the range ν to ν + dν
• In a photon gas, there are 2 EM polarization states, corresponding to the 2 independent directions of polarization of an EM wave. • As a result t • c speed of EM wave (light) in vacuum.

• The energy within the frequency range v to v + dv equals the number of photons within that range times the energy of each photon: u(ν) So we finally have: u(ν)

• So the total photon energy at frequency is: u(ν)dν • This is the Planck radiation formula, which gives the energy per unit frequency. • Re-expressing this in terms of the wavelength gives: u u(λ )

u(λ) • u(λ) is the energy per unit wavelength. • The Stephan-Boltzmann Law states that The total radiation energy is proportional to T 4 • This can be derived from the above expression by integrating u( ) over all wavelengths , as follows:

u(λ ) • The total energy can be calculated by integrating this over all : • Setting one has

• The total energy of the photon gas is: From a table, the integral has a value of • So

• The particle flux equals , where ν is the mean speed and n is the number density. In a similar way, the energy flux e can be calculated: • This is the Stephan-Boltzmann Law with the Stephan-Boltzmann constant =

• The wavelength at which u(λ is a ) the derivative maximum can be found by setting of u(λ equal to zero: ) • This is known as Wien’s Displacement Law

• For long wavelengths: u (Rayleigh- Jeans formula) • For short wavelengths: • Sun’s Surface T≈ 6000 K, thus
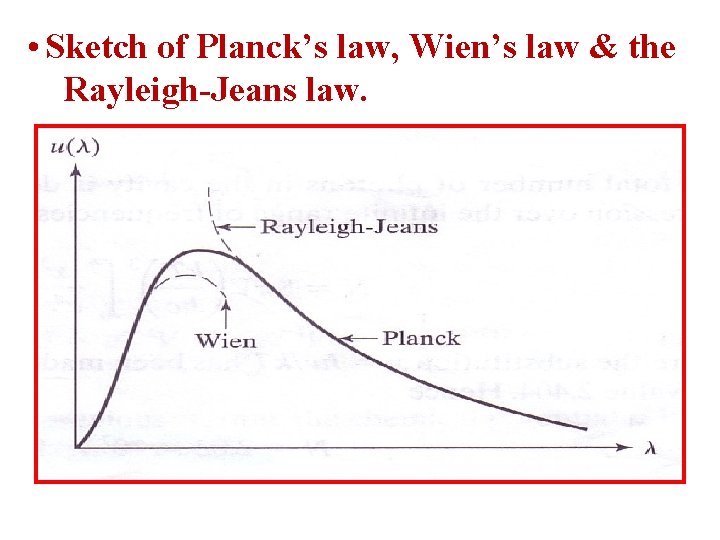
• Sketch of Planck’s law, Wien’s law & the Rayleigh-Jeans law.
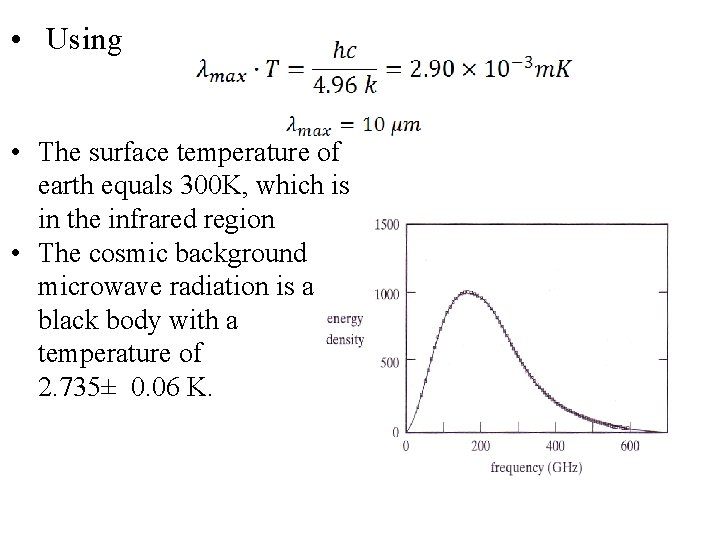
• Using • The surface temperature of earth equals 300 K, which is in the infrared region • The cosmic background microwave radiation is a black body with a temperature of 2. 735± 0. 06 K.

Properties of a Photon Gas The number of photons having frequencies between v and v + d v is The total number of photons in the cavity is determined by integrating over the infinite range of frequencies:

Leads to Where T is in Kelvin and V is in m 3 • The mean energy of a photon • The ratio of is therefore of the order of unity. • The heat capacity

• Substitute • Entropy

• Therefore, both the heat capacity and the entropy increase with the third power of the temperature! • For photon gas: • It shows that F does not explicitly depend on N. Therefore,

• Calculate the total electromagnetic energy inside an oven of volume 1 m 3 heated to a temperature of 400 F • Solution

• Show that thermal energy of the air in the oven is a factor of approximately 1010 larger than the electromagnetic energy. • Solution:
- Slides: 22