Photoelectron Velocity Map Imaging Spectroscopy of Be S

Photoelectron Velocity Map Imaging Spectroscopy of Be. S Amanda Dermer, Mallory Theis, Dr. Kyle Mascaritolo, Dr. Michael Heaven Emory University ISMS 2017

Interest in Beryllium Sulfide • Cluster evolution • Unique bonding properties • Be. O- 1 • Periodic Trends • Practical Applications • H 2 storage 4 • Rare gas bonding 5 • Blue-green laser diodes 2, 3 1. ) Mascaritolo, K. J. ; Dermer, A. R. ; Green, M. L. ; Gardner, A. M. ; Heaven, M. C. , Photodetachment spectroscopy of the beryllium oxide anion, Be. O. J. Chem. Phys. 2017, 146 (5), 054301/1 -054301/6. 2. ) Benosman, N. ; Amrane, N. ; Mecabih, S. ; Aourag, H. , Structural and electronic properties of bulk Be. S. Physica B: Condensed Matter (Amsterdam, Netherlands) 2001, 304 (1 -4), 214 -220. 3. ) Heciri, D. ; Beldi, L. ; Drablia, S. ; Meradji, H. ; Derradji, N. E. ; Belkhir, H. ; Bouhafs, B. , First-principles elastic constants and electronic structure of beryllium chalcogenides Be. S, Be. Se and Be. Te. Comput. Mater. Sci. 2007, 38 (4), 609 -617. 4. )Hwang, D. -Y. ; Mebel, A. M. , Theoretical Study of the Reversible Storage of H 2 by Be. S. J. Am. Chem. Soc. 2000, 122 (46), 11406 -11410. 5. )Wang, Q. ; Wang, X. , Infrared Spectra of Ng. Be. S (Ng = Ne, Ar, Kr, Xe) and Be. S 2 in Noble-Gas Matrices. J. Phys. Chem. A 2013, 117 (7), 1508 -1513

Previous Studies on Be. S 1 • Numerous theoretical studies of the neutral 1 -8 • Ground (X 1∑+) state and first excited states • Spectroscopic studies on the neutral 9, 10 No studies on Be. S-, as of yet • Ground (X 1∑+) state 1. ) De Oliveira-Filho, A. G. S. ; Alves, T. V. ; Ribas, V. W. ; Ferrao, L. F. A. ; Roberto-Neto, O. ; Machado, F. B. C. ; Ornellas, F. R. , A CASSCF/MRCI study of the low-lying electronic states of the Be. S molecule. International Journal of Quantum Chemistry 2011, 111 (7/8), 1694 -1700. 2. ) Larbi, T. ; Khadri, F. ; Ghalila, H. ; Lahmar, S. ; Hochlaf, M. , Theoretical spectroscopy and metastability of Be. S and its cation. Chem. Phys. 2010, 373 (3), 193 -202. 3. ) Liu, H. ; Xing, W. ; Shi, D. -h. ; Sun, J. -f. , Investigation on spectroscopic parameters and molecular constants of Be. S (X 1Σ+) using MRCI theory. Yuanzi Yu Fenzi Wuli Xuebao 2012, 29 (2), 216 -222. 4. ) Noga, J. ; Pluta, T. , Coupled cluster calculations of the electric properties of Be. S. An analysis of the fifth-order non-iterative corrections. Chem. Phys. Lett. 1997, 264 (1, 2), 101 -108. 5. ) Partridge, H. ; Langhoff, S. R. ; Bauschlicher, C. W. , Jr. , Theoretical study of the alkali and alkaline earth monosulfides. J. Chem. Phys. 1988, 88 (10), 6431 -7. 6. ) Pouilly, B. ; Robbe, J. M. ; Schamps, J. ; Field, R. W. ; Young, L. , Perturbations in the A 1Π-X 1Σ+ system of the beryllium sulfide molecule. The a 3Πi state. Journal of Molecular Spectroscopy 1982, 96 (1), 1 -55. 7. ) Rai, D. P. ; Ghimire, M. P. ; Thapa, R. K. , A DFT study of Be. X (X = S, Se, Te) semiconductor: Modified Becke Johnson (m. BJ) potential. Semiconductors 2014, 48 (11), 1411 -1422. 8. ) Srnec, M. ; Zahradnik, R. , Diatomics AB (A = Be, Mg; B = O, S) and oligomers thereof: A theoretical study. Chem. Phys. Lett. 2005, 407 (4 -6), 283 -288 9. ) Pouilly, B. ; Robbe, J. M. ; Schamps, J. ; Field, R. W. ; Young, L. , Perturbations in the A 1Π-X 1Σ+ system of the beryllium sulfide molecule. The a 3Πi state. Journal of Molecular Spectroscopy 1982, 96 (1), 1 -55. 10. ) Cheetham, C. J. ; Gissane, W. J. M. ; Barrow, R. F. , Electronic spectra of the Be. S molecule. Transactions of the Faraday Society 1965, 61 (511), 1308 -16.

Be. S and Be. S- Theoretical Methods • Molpro computational package • RCCSD(T) with B 3 LYP reference wavefunction • CASSCF+MRCI+Q • Active space: • Be: 2 s, 2 p • S: 2 s, 2 p, 3 s, 3 p • Basis Sets: • Be: aug-cc-pw. CVXZ (X= T, Q, 5) • S: aug-cc-p. CVXZ (X= T, Q, 5) Ø Point-wise potential energy curves for Be. S and Be. S- • LEVEL 8. 0 program Ø Ro-vibrational levels Ø Franck Condon Factors • Morse potential fitting Ø ωe, ωeχe
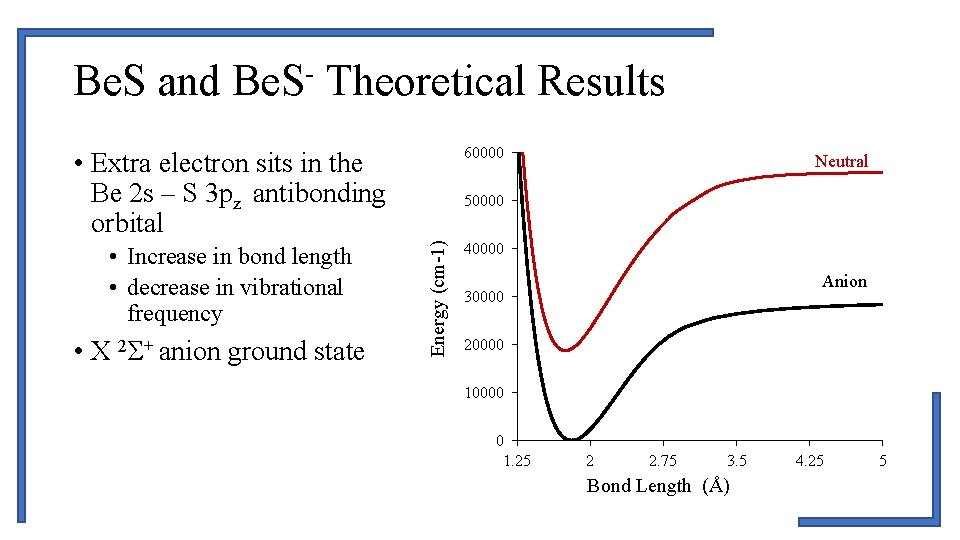
Be. S and Be. S- Theoretical Results 60000 • Extra electron sits in the Be 2 s – S 3 pz antibonding orbital • X 2Σ+ anion ground state 50000 Energy (cm-1) • Increase in bond length • decrease in vibrational frequency Neutral 40000 Anion 30000 20000 10000 0 1. 25 2 2. 75 3. 5 Bond Length (Å) 4. 25 5
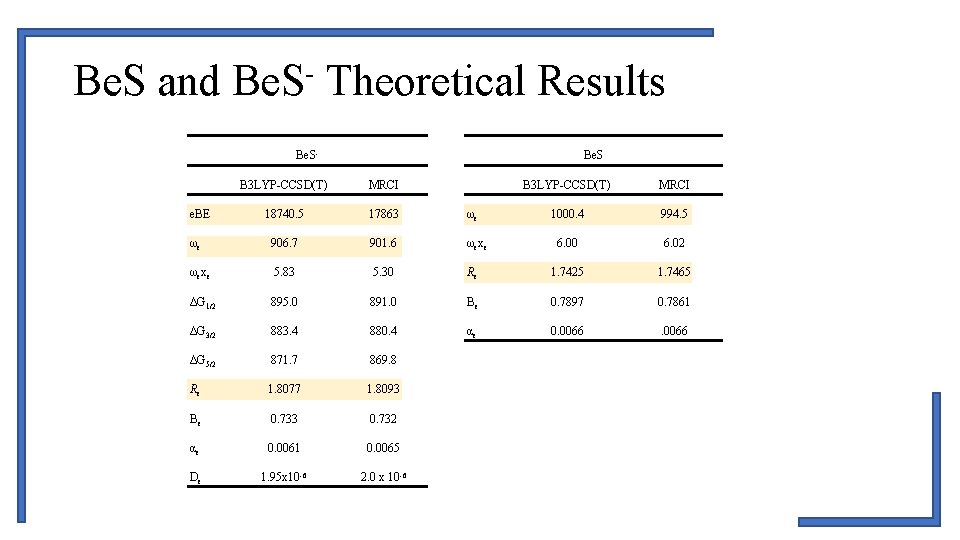
Be. S and Be. S- Theoretical Results Be. S B 3 LYP-CCSD(T) MRCI 18740. 5 17863 ωe ωe 906. 7 901. 6 ωexe 5. 83 5. 30 ΔG 1/2 895. 0 ΔG 3/2 B 3 LYP-CCSD(T) MRCI 1000. 4 994. 5 6. 00 6. 02 Re 1. 7425 1. 7465 891. 0 Be 0. 7897 0. 7861 883. 4 880. 4 αe 0. 0066 ΔG 5/2 871. 7 869. 8 Re 1. 8077 1. 8093 Be 0. 733 0. 732 αe 0. 0061 0. 0065 De 1. 95 x 10 -6 2. 0 x 10 -6 e. BE

Photoelectron Velocity Map Imaging Spectroscopy e. KE Detection Range Neutral Ground State Detachment Threshold EA Tunable hν Anion Ground State
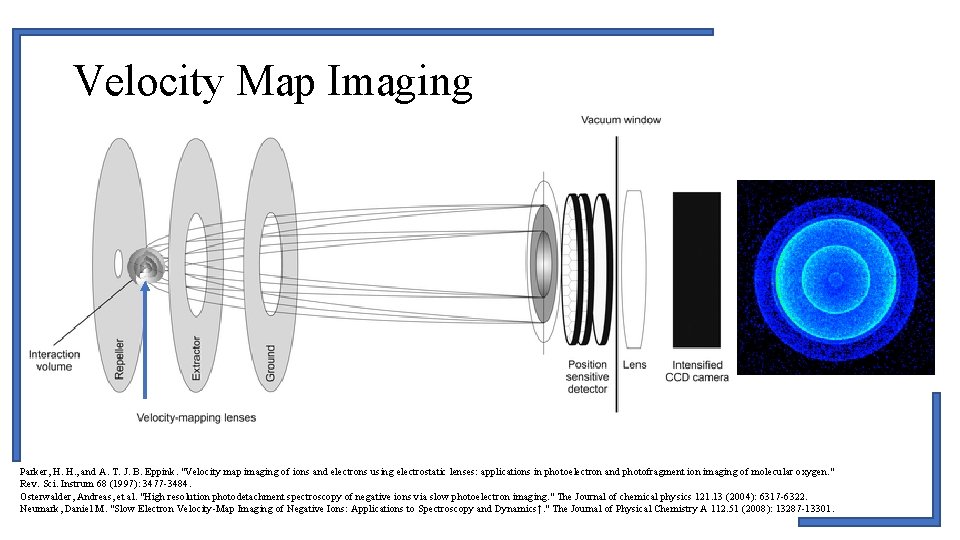
Velocity Map Imaging Parker, H. H. , and A. T. J. B. Eppink. "Velocity map imaging of ions and electrons using electrostatic lenses: applications in photoelectron and photofragment ion imaging of molecular oxygen. " Rev. Sci. Instrum 68 (1997): 3477 -3484. Osterwalder, Andreas, et al. "High resolution photodetachment spectroscopy of negative ions via slow photoelectron imaging. " The Journal of chemical physics 121. 13 (2004): 6317 -6322. Neumark, Daniel M. "Slow Electron Velocity-Map Imaging of Negative Ions: Applications to Spectroscopy and Dynamics†. " The Journal of Physical Chemistry A 112. 51 (2008): 13287 -13301.
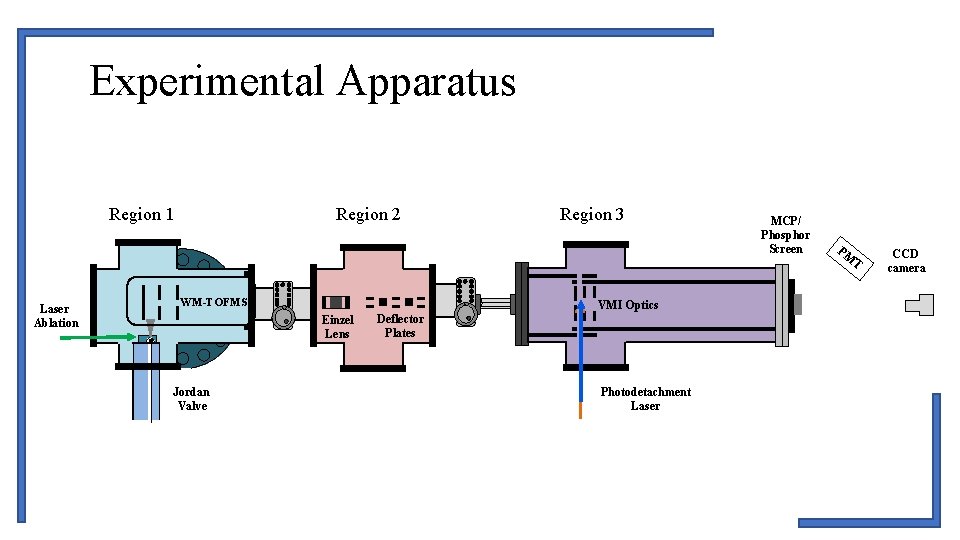
Experimental Apparatus Region 1 Laser Ablation Region 2 WM-TOFMS VMI Optics Einzel Lens Jordan Valve Region 3 Deflector Plates Photodetachment Laser MCP/ Phosphor Screen PM T CCD camera
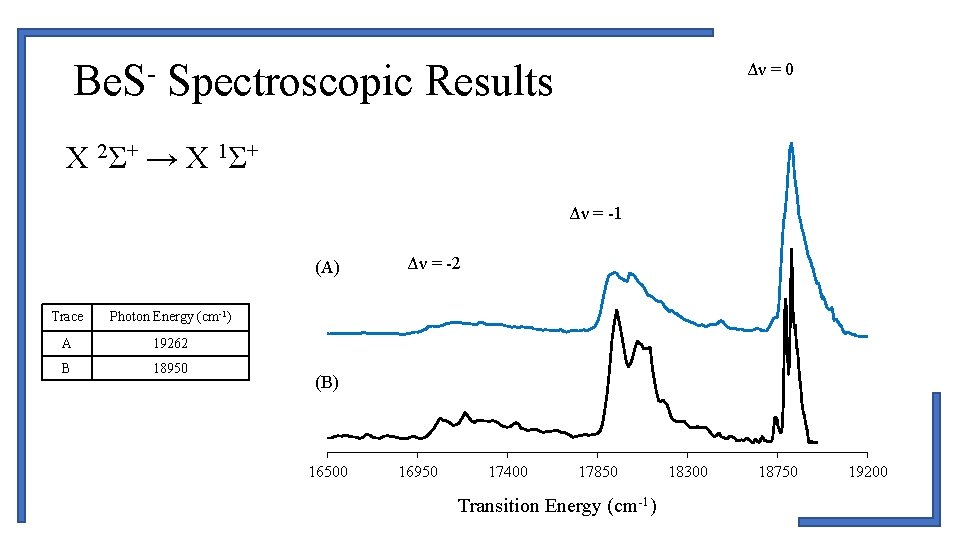
Be. S- Spectroscopic Results Δν = 0 X 2Σ+ → X 1Σ+ Δν = -1 (A) Trace Photon Energy (cm-1) A 19262 B 18950 Δν = -2 (B) 16500 16950 17400 17850 Transition Energy (cm-1) 18300 18750 19200
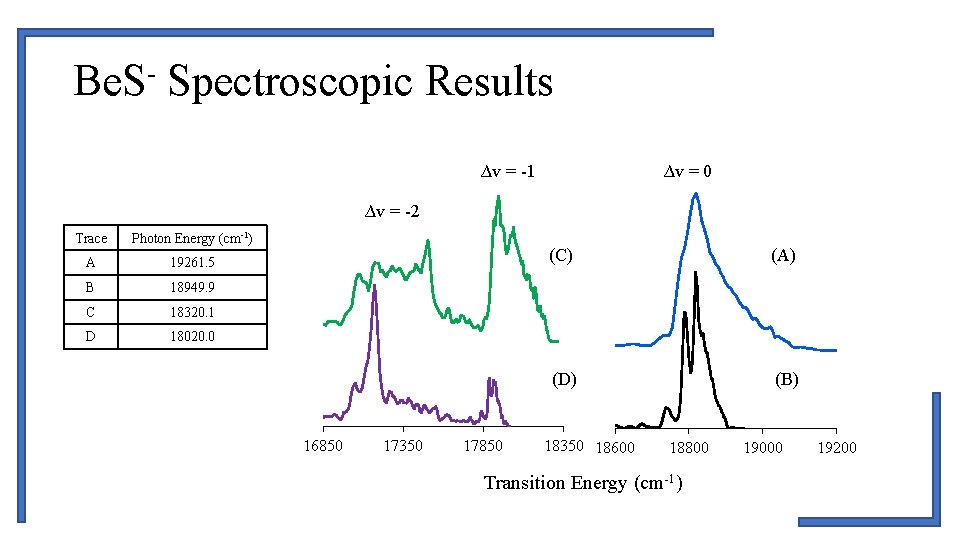
Be. S- Spectroscopic Results ∆v = -1 ∆v = 0 ∆v = -2 Trace Photon Energy (cm-1) A 19261. 5 B 18949. 9 C 18320. 1 D 18020. 0 16850 17350 17850 (C) (A) (D) (B) 18350 18600 18800 Transition Energy (cm-1) 19000 19200
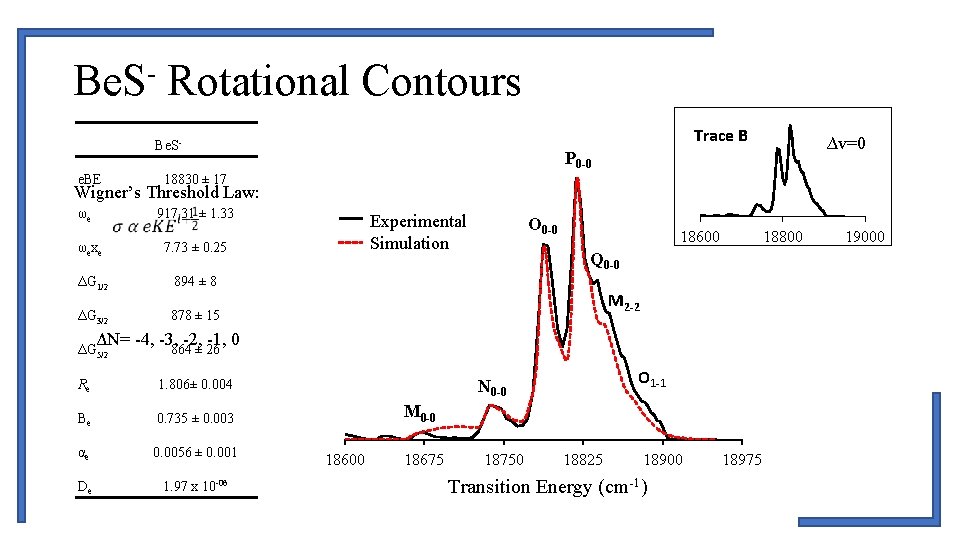
Be. S- Rotational Contours Trace B Be. Se. BE 18830 ± 17 Wigner’s Threshold Law: ωe 917. 31 ± 1. 33 ωexe 7. 73 ± 0. 25 ΔG 1/2 894 ± 8 ΔG 3/2 878 ± 15 Series 1 Experimental Simulation Series 2 O 0 -0 18600 18800 Q 0 -0 M 2 -2 ΔN= -4, -3, -2, -1, 0 ΔG 5/2 ∆v=0 P 0 -0 864 ± 26 Re 1. 806± 0. 004 Be 0. 735 ± 0. 003 αe 0. 0056 ± 0. 001 De 1. 97 x 10 -06 O 1 -1 N 0 -0 M 0 -0 18600 18675 18750 18825 18900 Transition Energy (cm-1) 18975 19000
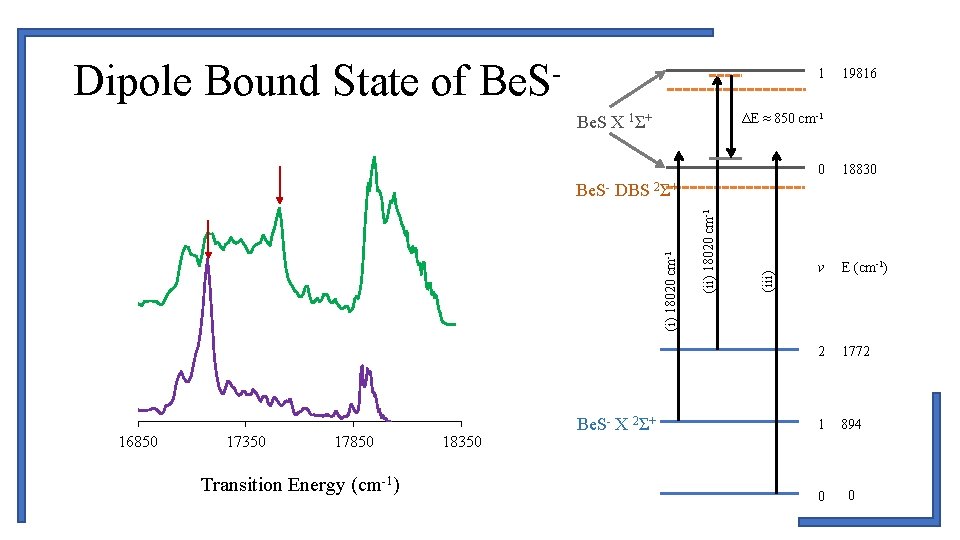
Dipole Bound State of Be. S- 1 19816 ΔE ≈ 850 cm-1 Be. S X 1Σ+ 0 18830 v E (cm-1) 2 1772 1 894 0 0 16850 17350 17850 Transition Energy (cm-1) 18350 Be. S- X 2Σ+ (iii) (ii) 18020 cm-1 (i) 18020 cm-1 Be. S- DBS 2Σ+
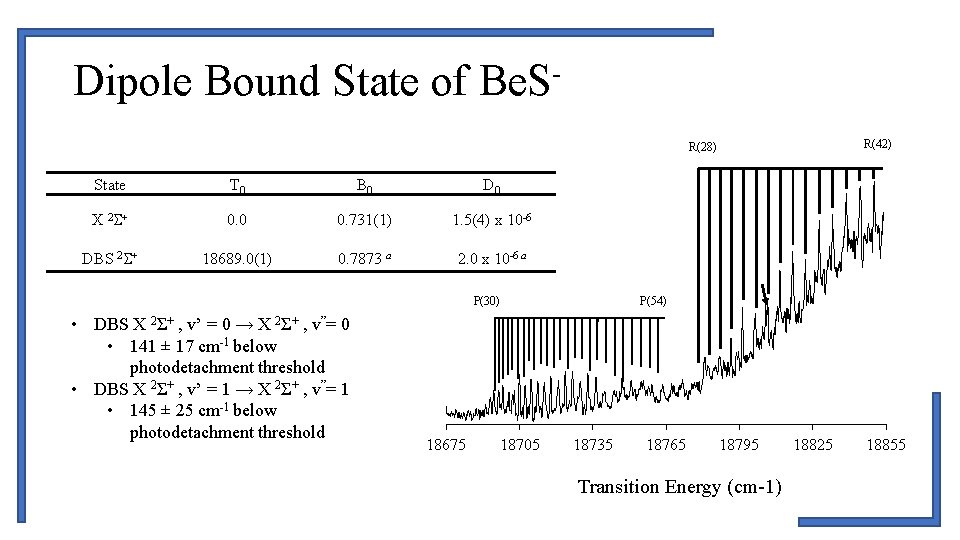
Dipole Bound State of Be. SR(42) R(28) State T 0 B 0 D 0 X 2 S+ 0. 0 0. 731(1) 1. 5(4) x 10 -6 DBS 2 S+ 18689. 0(1) 0. 7873 a 2. 0 x 10 -6 a P(30) • DBS X 2Σ+ , v’ = 0 → X 2Σ+ , v”= 0 • 141 ± 17 cm-1 below photodetachment threshold • DBS X 2Σ+ , v’ = 1 → X 2Σ+ , v”= 1 • 145 ± 25 cm-1 below photodetachment threshold 18675 18705 P(54) 18735 18765 18795 Transition Energy (cm-1) 18825 18855
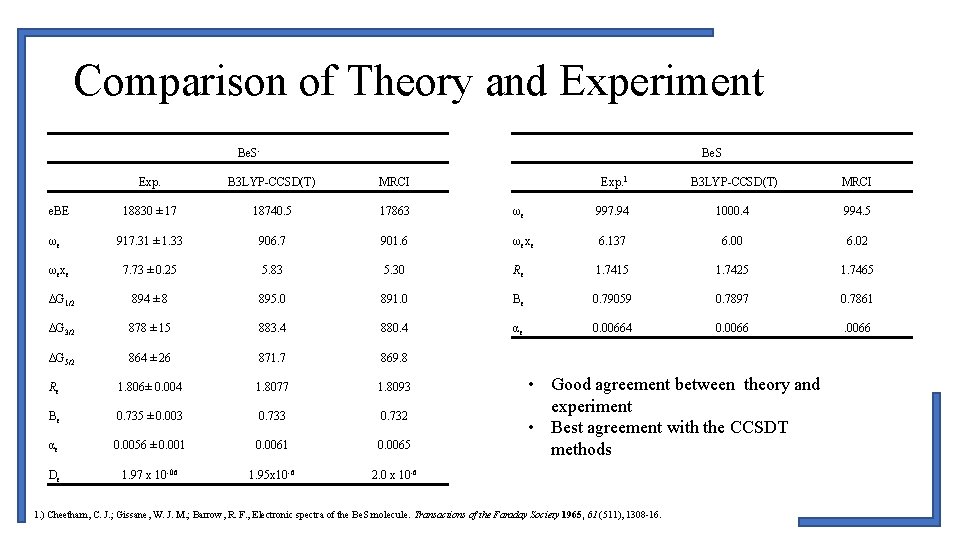
Comparison of Theory and Experiment Be. S Exp. B 3 LYP-CCSD(T) MRCI 18830 ± 17 18740. 5 17863 917. 31 ± 1. 33 906. 7 ωexe 7. 73 ± 0. 25 ΔG 1/2 Exp. 1 B 3 LYP-CCSD(T) MRCI ωe 997. 94 1000. 4 994. 5 901. 6 ωexe 6. 137 6. 00 6. 02 5. 83 5. 30 Re 1. 7415 1. 7425 1. 7465 894 ± 8 895. 0 891. 0 Be 0. 79059 0. 7897 0. 7861 ΔG 3/2 878 ± 15 883. 4 880. 4 αe 0. 00664 0. 0066 ΔG 5/2 864 ± 26 871. 7 869. 8 Re 1. 806± 0. 004 1. 8077 1. 8093 Be 0. 735 ± 0. 003 0. 732 αe 0. 0056 ± 0. 001 0. 0065 De 1. 97 x 10 -06 1. 95 x 10 -6 2. 0 x 10 -6 e. BE ωe • Good agreement between theory and experiment • Best agreement with the CCSDT methods 1. ) Cheetham, C. J. ; Gissane, W. J. M. ; Barrow, R. F. , Electronic spectra of the Be. S molecule. Transactions of the Faraday Society 1965, 61 (511), 1308 -16.

Conclusions • Be. S- was studied for the first time, experimentally and theoretically • EA, constants • EA = 18830 ± 17 cm-1 • Be. S- ωe =917. 31 ± 1. 33 cm-1, ωeχe = 7. 73 ± 0. 25 cm-1 • Dipole bound state was found for the anion. • Be. S anion featured similar characteristics to the Be. O anion. • Our theoretical methods agree well with our experimentally determined constants. • Future Directions: • Diatomic beryllium-containing molecules • Be. C-, Be. F- • Larger beryllium species • Be. O 2 -, Be. C 2 -, Be. S 2 - Ø Addition of a cold ion trap

Acknowledgements • Heaven Group: • • Dr. Michael Heaven Dr. Kyle Mascaritolo Amanda Reed Robert Van. Gundy Sean Bresler Dr. Michael Sullivan Dr. Jiande Han • Funding: • NSF

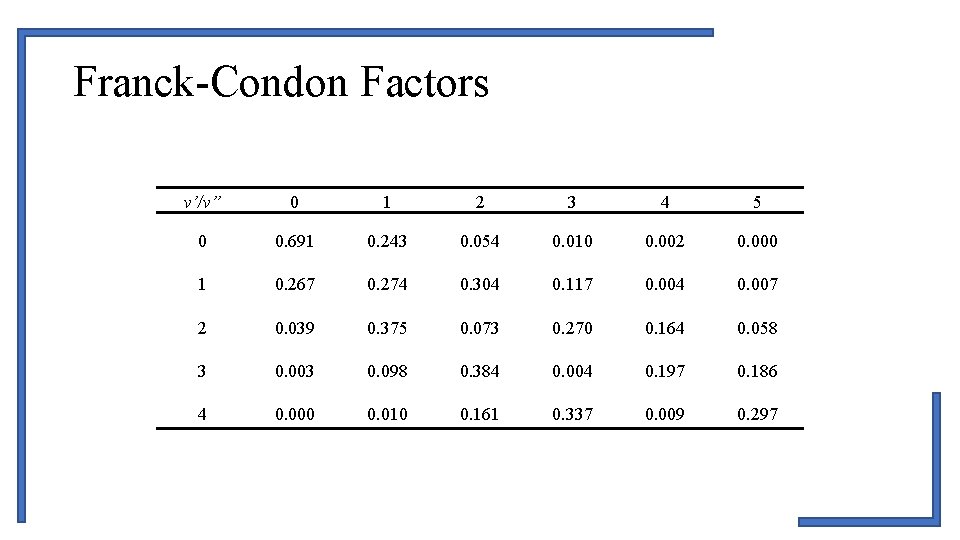
Franck-Condon Factors v’/v” 0 1 2 3 4 5 0 0. 691 0. 243 0. 054 0. 010 0. 002 0. 000 1 0. 267 0. 274 0. 304 0. 117 0. 004 0. 007 2 0. 039 0. 375 0. 073 0. 270 0. 164 0. 058 3 0. 003 0. 098 0. 384 0. 004 0. 197 0. 186 4 0. 000 0. 010 0. 161 0. 337 0. 009 0. 297
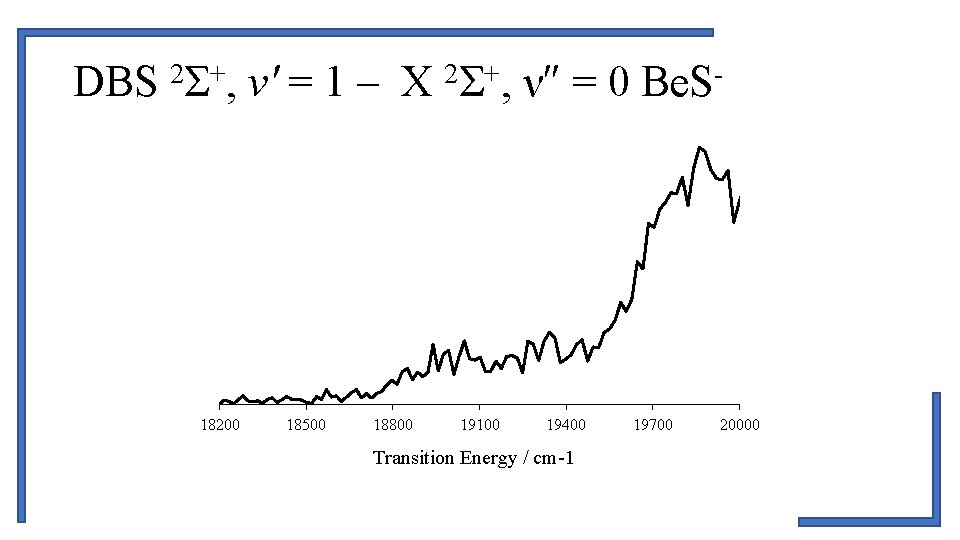
2 + DBS Σ , v′ = 1 – X Σ , ν″ = 0 Be. S 18200 18500 18800 19100 19400 Transition Energy / cm-1 19700 20000

Rotational Simulations
Sulfur Calibrations 3 P 1 EA 2 4 3 5 e. KE / cm-1 4 0 3 P 1 3 P 2 6 22 hv Pixel 5 6 16100 16300 2 P 1/2 2 P 3/2 1 3 16500 16700 16900 17100 Transition Energy (cm-1) 17300 17500
- Slides: 22