Photoelectron spectroscopy of the cyclopentadienide anion Analysis of









- Slides: 17

Photoelectron spectroscopy of the cyclopentadienide anion: Analysis of the Jahn. Teller effects in the cyclopentadienyl radical Takatoshi Ichino, Adam J. Gianola, and W. Carl Lineberger JILA and Department of Chemistry and Biochemistry University of Colorado, Boulder, Colorado 80309 John F. Stanton Department of Chemistry and Biochemistry and Institute for Theoretical Chemistry The University of Texas at Austin, TX 78712 Supported by

High-temperature oxidation of benzene cyclopentadienyl radical (C 5 H 5) • Cyclopentadienyl radical is formed in combustion of benzene. • Cyclopentadienyl radical is a resonance stabilized free radical which may play a role in growth of polycyclic aromatic hydrocarbons.

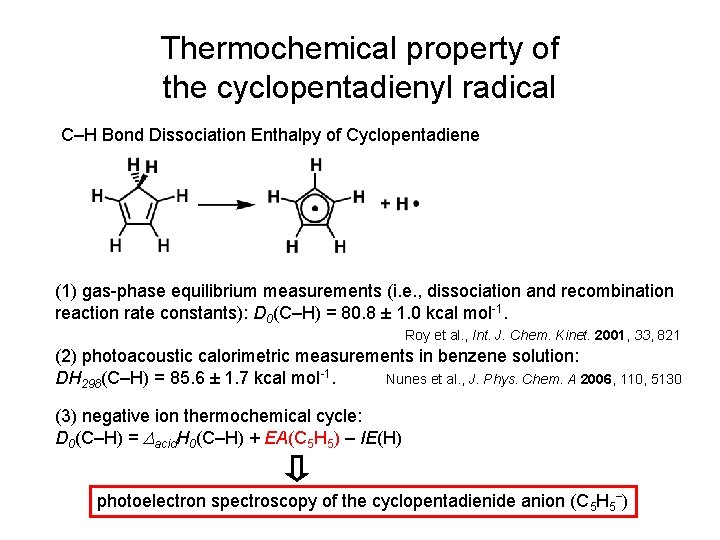
Thermochemical property of the cyclopentadienyl radical C–H Bond Dissociation Enthalpy of Cyclopentadiene (1) gas-phase equilibrium measurements (i. e. , dissociation and recombination reaction rate constants): D 0(C–H) = 80. 8 ± 1. 0 kcal mol-1. Roy et al. , Int. J. Chem. Kinet. 2001, 33, 821 (2) photoacoustic calorimetric measurements in benzene solution: DH 298(C–H) = 85. 6 ± 1. 7 kcal mol-1. Nunes et al. , J. Phys. Chem. A 2006, 110, 5130 (3) negative ion thermochemical cycle: D 0(C–H) = Dacid. H 0(C–H) + EA(C 5 H 5) – IE(H) photoelectron spectroscopy of the cyclopentadienide anion (C 5 H 5‾)

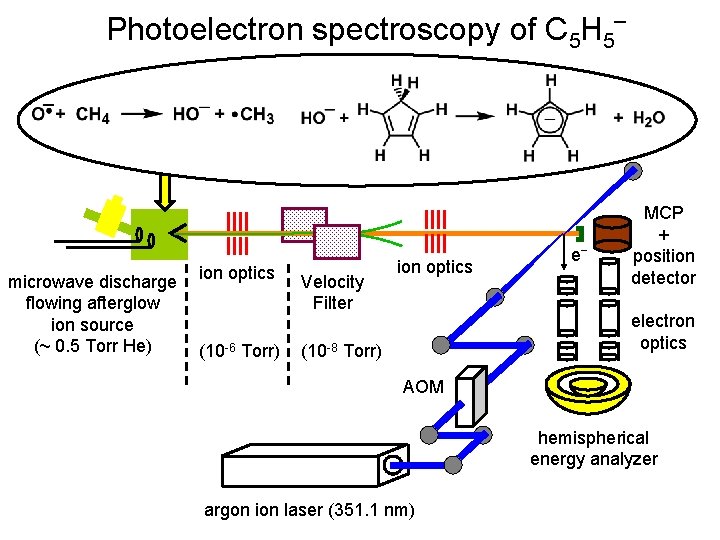
Photoelectron spectroscopy of C 5 H 5‾ microwave discharge flowing afterglow ion source (~ 0. 5 Torr He) ion optics (10 -6 Torr) Velocity Filter ion optics e‾ MCP + position detector electron optics (10 -8 Torr) AOM hemispherical energy analyzer argon ion laser (351. 1 nm)

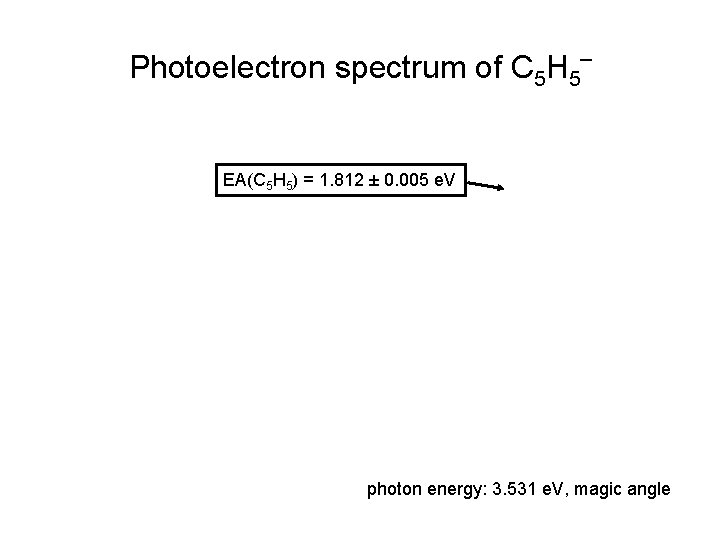
Photoelectron spectrum of C 5 H 5‾ EA(C 5 H 5) = 1. 812 ± 0. 005 e. V photon energy: 3. 531 e. V, magic angle

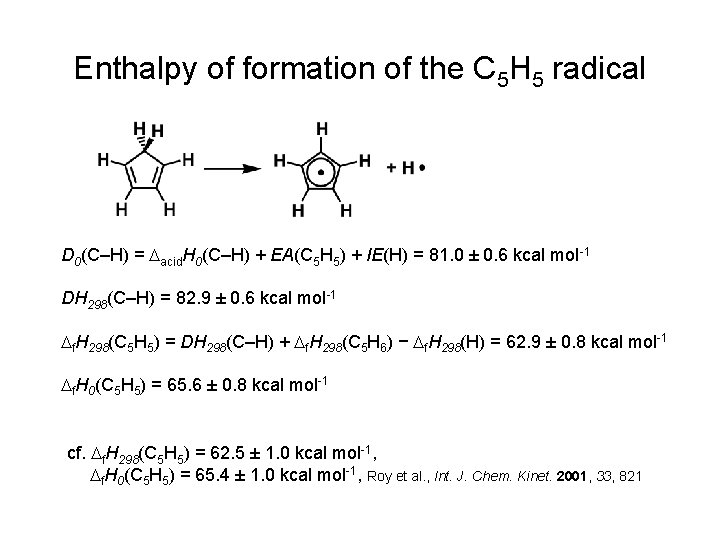
Enthalpy of formation of the C 5 H 5 radical D 0(C–H) = Dacid. H 0(C–H) + EA(C 5 H 5) + IE(H) = 81. 0 ± 0. 6 kcal mol-1 DH 298(C–H) = 82. 9 ± 0. 6 kcal mol-1 Df. H 298(C 5 H 5) = DH 298(C–H) + Df. H 298(C 5 H 6) − Df. H 298(H) = 62. 9 ± 0. 8 kcal mol-1 Df. H 0(C 5 H 5) = 65. 6 ± 0. 8 kcal mol-1 cf. Df. H 298(C 5 H 5) = 62. 5 ± 1. 0 kcal mol-1, Df. H 0(C 5 H 5) = 65. 4 ± 1. 0 kcal mol-1, Roy et al. , Int. J. Chem. Kinet. 2001, 33, 821

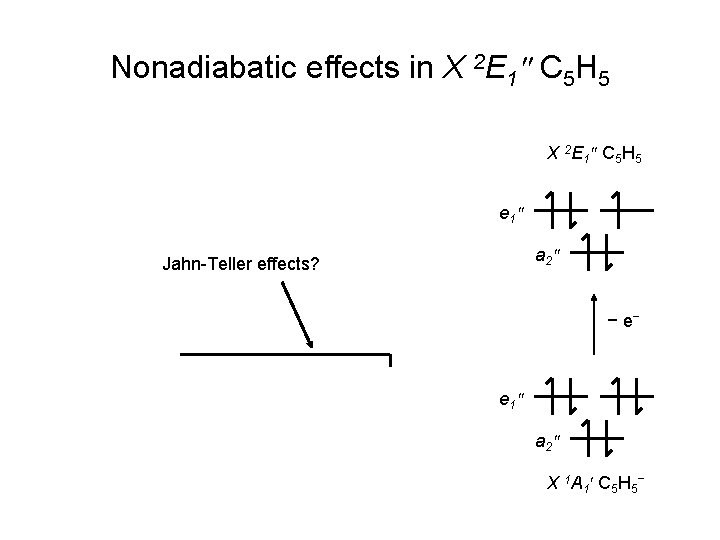
Nonadiabatic effects in X 2 E 1″ C 5 H 5 X 2 E 1 ″ C 5 H 5 e 1 ″ a 2 ″ Jahn-Teller effects? − e‾ e 1 ″ a 2 ″ X 1 A 1 ′ C 5 H 5 ‾

Laser excited dispersed fluorescence A 2 A 2″ ― X 2 E 1″ C 5 H 5 electronic transition • Emission from the vibraional ground state as well as from the vibrationally excited states of the Jahn-Teller active modes. • Spectral simulations based on a model Hamiltonian in terms of JT eigenfunctions. • Ab initio evaluation of JT coupling constants. • Measurements of isotopomers. Applegate et al. , J. Chem. Phys. 2001, 114, 4855, 4869

Model Hamiltonian for a degenerate system with linear Jahn-Teller coupling where nuclear kinetic energy operator harmonic potential energy of the reference state linear intrastate coupling linear and bilinear JT coupling E : energy of the degenerate states at the reference geometry Model potential has an expansion form around the reference geometry in terms of the reduced normal coordinates of the reference state (qi). Köppel et al. , Adv. Chem. Phys. 1984, 57, 59; Mayer et al. , J. Chem. Phys. 1994, 100, 899

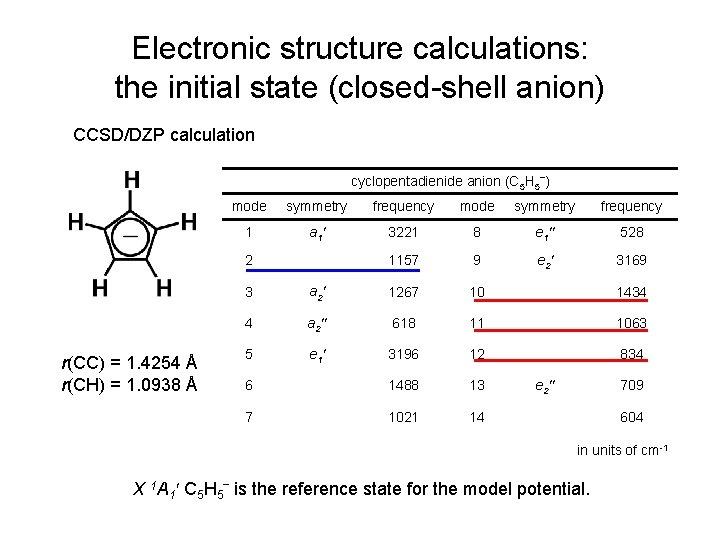
Electronic structure calculations: the initial state (closed-shell anion) CCSD/DZP calculation cyclopentadienide anion (C 5 H 5‾) mode symmetry frequency 1 a 1′ 3221 8 e 1″ 528 1157 9 e 2′ 3169 2 r(CC) = 1. 4254 Å r(CH) = 1. 0938 Å 3 a 2′ 1267 10 1434 4 a 2″ 618 11 1063 5 e 1′ 3196 12 834 6 1488 13 7 1021 14 e 2″ 709 604 in units of cm-1 X 1 A 1′ C 5 H 5‾ is the reference state for the model potential.

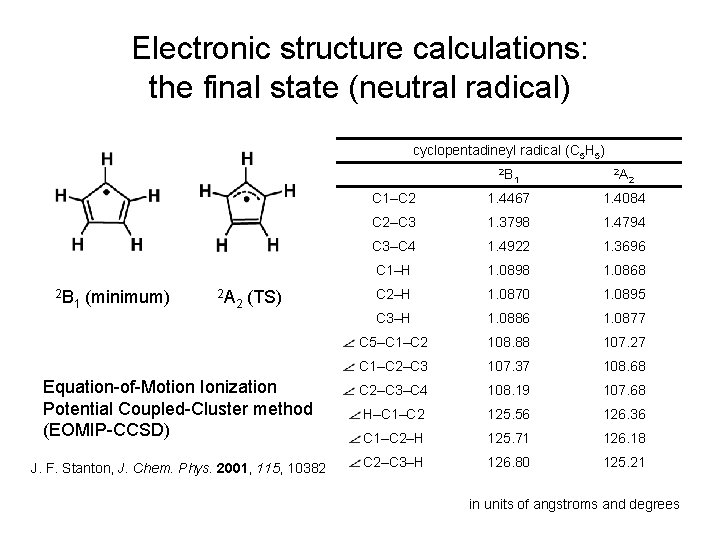
Electronic structure calculations: the final state (neutral radical) cyclopentadineyl radical (C 5 H 5) 2 B 1 2 A 2 C 1–C 2 1. 4467 1. 4084 C 2–C 3 1. 3798 1. 4794 C 3–C 4 1. 4922 1. 3696 C 1–H 1. 0898 1. 0868 C 2–H 1. 0870 1. 0895 C 3–H 1. 0886 1. 0877 C 5–C 1–C 2 108. 88 107. 27 C 1–C 2–C 3 107. 37 108. 68 Equation-of-Motion Ionization Potential Coupled-Cluster method (EOMIP-CCSD) C 2–C 3–C 4 108. 19 107. 68 H–C 1–C 2 125. 56 126. 36 C 1–C 2–H 125. 71 126. 18 J. F. Stanton, J. Chem. Phys. 2001, 115, 10382 C 2–C 3–H 126. 80 125. 21 2 B 1 (minimum) 2 A 2 (TS) in units of angstroms and degrees

Ab initio parametrization of the model potential linear coupling constants: Geometry displacements from the initial (anion) to the final (radical) states are multiplied by the quadratic force constant matrix of the final state at its equilibrium geometry in terms of the anion reduced normal coordinates. bilinear coupling constants: The off-diagonal elements of the quadratic force constant matrix of the final state at its equilibrium geometry in terms of the anion reduced normal coordinates. X 2 E 1″ C 5 H 5 model potential parameter (e. V) linear intrastate coupling k 2 0. 0234 linear JT coupling l 10 0. 0626 l 11 0. 1149 l 12 0. 2125 bilinear JT coupling k 2, 10 − 0. 0001 k 2, 11 0. 0116 k 2, 12 0. 0281 No energy barrier is assumed along the pseudorotation path in the model potential.

Simulation based on the model Hamiltonian: linear intrastate coupling (a 1′) only X 2 E 1 ″ C 5 H 5 No obeserved peak can be assigned to a 1′ mode.

Simulation based on the model Hamiltonian: linear intrastate (a 1′) + linear JT (e 2′) coupling X 2 E 1 ″ C 5 H 5 Observed peak positions are well reproduced by linear JT coupling.

Simulation based on the model Hamiltonian: add bilinear coupling X 2 E 1 ″ C 5 H 5 Relative peak intensities are well reproduced by addition of bilinear coupling. cf. photoelectron spectrum of CH 3 O‾, Schmidt-Klügmann et al. , Chem. Phys. Lett. 2003, 369, 21

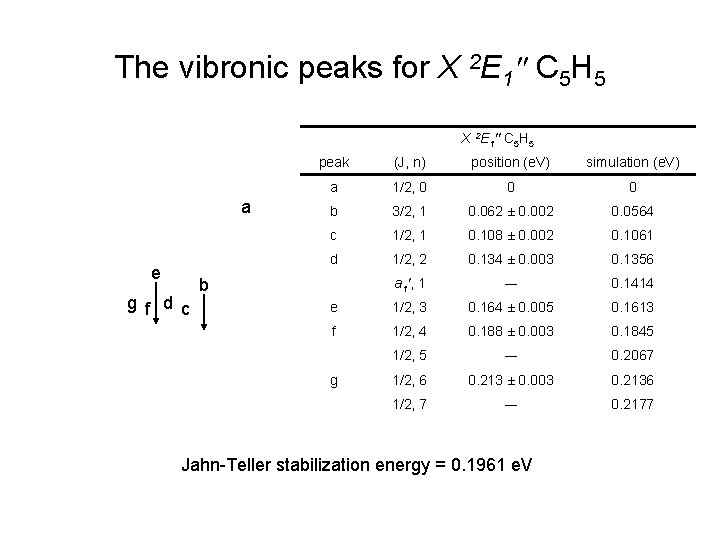
The vibronic peaks for X 2 E 1″ C 5 H 5 a e g f d c peak (J, n) position (e. V) simulation (e. V) a 1/2, 0 0 0 b 3/2, 1 0. 062 ± 0. 002 0. 0564 c 1/2, 1 0. 108 ± 0. 002 0. 1061 d 1/2, 2 0. 134 ± 0. 003 0. 1356 a 1′, 1 ― 0. 1414 e 1/2, 3 0. 164 ± 0. 005 0. 1613 f 1/2, 4 0. 188 ± 0. 003 0. 1845 1/2, 5 ― 0. 2067 1/2, 6 0. 213 ± 0. 003 0. 2136 1/2, 7 ― 0. 2177 b g Jahn-Teller stabilization energy = 0. 1961 e. V

Conclusion • The 351. 1 nm photoelectron spectrum of the cyclopentadienide anion has been measured. The electron affinity of the cyclopentadienyl radical has been determined to be 1. 812 ± 0. 005 e. V. • The C–H bond dissociation enthalpy of cyclopentadiene has been derived as D 0(C–H) = 81. 0 ± 0. 6 kcal mol-1 from a negative ion thermochemical cycle. The enthalpy of formation of the cyclopentadienyl radical has been derived to be Df. H 298 = 62. 9 ± 0. 8 kcal mol-1. • Model potentials of X 2 E 1″ C 5 H 5 have been constructed around the equilibrium geometry of X 1 A 1′ C 5 H 5‾ in terms of the anion reduced normal coordinate, based on the EOMIP-CCSD calculations. A simulation based on the model Hamiltonian reproduces the observed vibronic structure very well, revealing strong Jahn-Teller activity for e 2′ modes in the spectrum. It is important to include the bilinear coupling between a 1′ and e 2′ modes in the model potential. • The simulation is completely ab initio.