Photoelectron Imaging of Vibrational Autodetachment from Nitromethane Anions

Photoelectron Imaging of Vibrational Autodetachment from Nitromethane Anions Chris L. Adams, Holger Schneider, J. Mathias Weber JILA, University of Colorado, Boulder, CO 80309 -0440 OSU International Symposium on Molecular Spectroscopy June 19, 2008

Low Energy Photoelectron Imaging of Nitromethane Anions Chris L. Adams, Holger Schneider, J. Mathias Weber JILA, University of Colorado, Boulder, CO 80309 -0440 OSU International Symposium on Molecular Spectroscopy June 19, 2008

Nitromethane - a model system • Binding energy of the excess electron in the anion is less than energies of the fundamental CH stretching vibrational transitions. interaction of vibrational excitation and electron emission • Vibrational autodetachment mediated internal vibrational relaxation (IVR) upon excitation of CH stretching fundamental observable in photoelectron spectrum? • Small molecule, tractable by theory • Relatively large error bars for current electron affinity (EA = 260 ± 80 me. V) Compton et al. , J. Chem. Phys. 105 (1996) 3472
So what’s already known?

Photoelectron Spectrum of bare Nitromethane
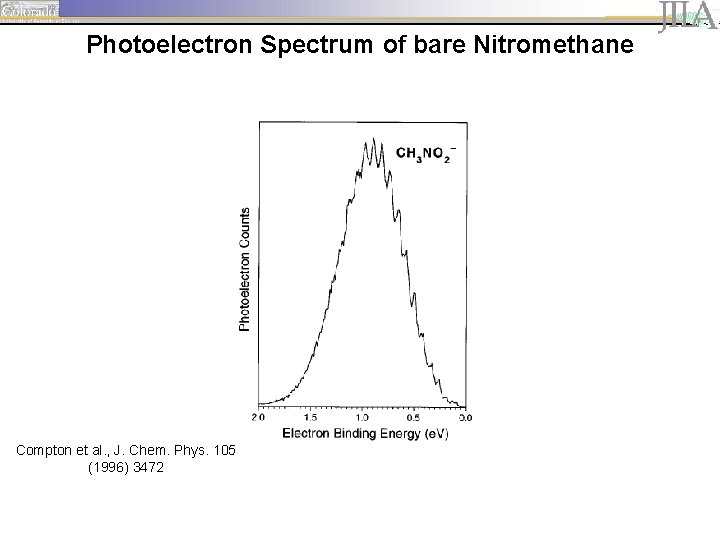
Photoelectron Spectrum of bare Nitromethane Compton et al. , J. Chem. Phys. 105 (1996) 3472
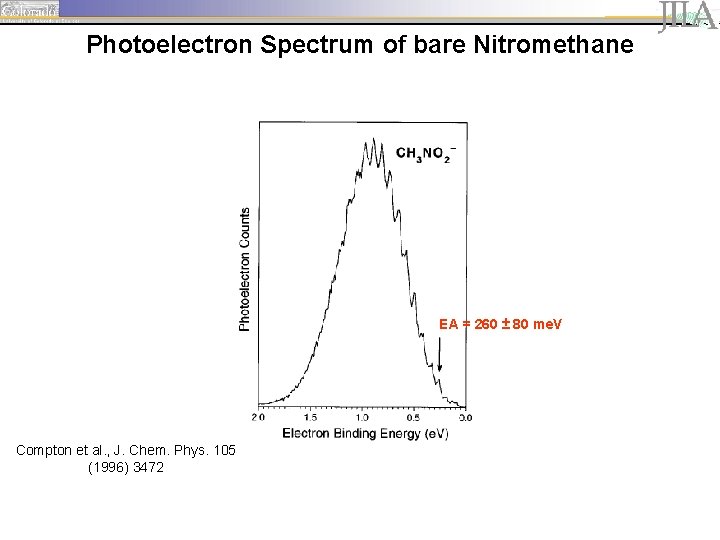
Photoelectron Spectrum of bare Nitromethane EA = 260 ± 80 me. V Compton et al. , J. Chem. Phys. 105 (1996) 3472
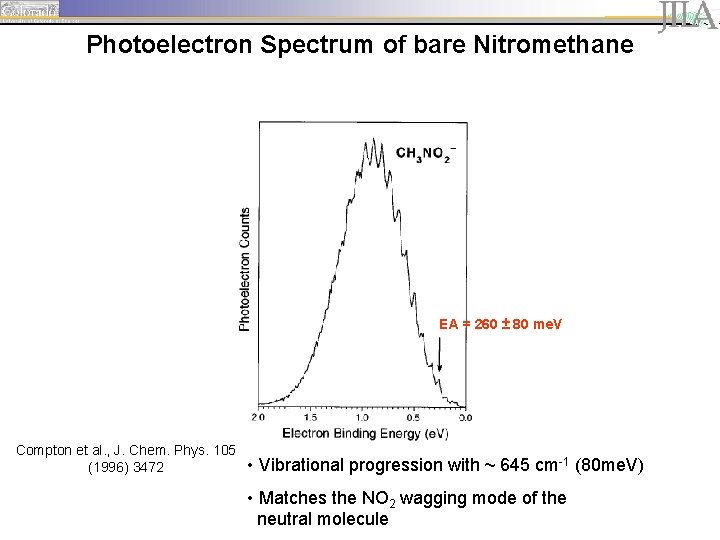
Photoelectron Spectrum of bare Nitromethane EA = 260 ± 80 me. V Compton et al. , J. Chem. Phys. 105 (1996) 3472 • Vibrational progression with ~ 645 cm-1 (80 me. V) • Matches the NO 2 wagging mode of the neutral molecule

Assignment of vibrational features: compare geometry of the neutral and the anion
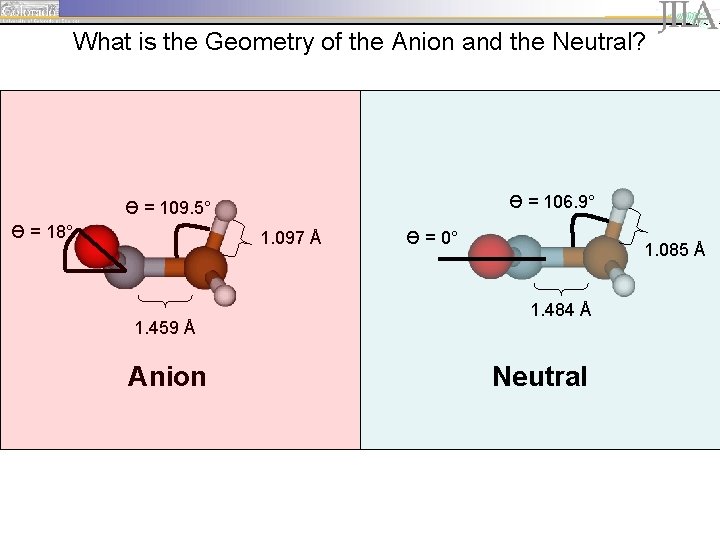
What is the Geometry of the Anion and the Neutral? Ө = 106. 9° Ө = 109. 5° Ө = 18° 1. 097 Å 1. 459 Å Anion Ө = 0° 1. 085 Å 1. 484 Å Neutral
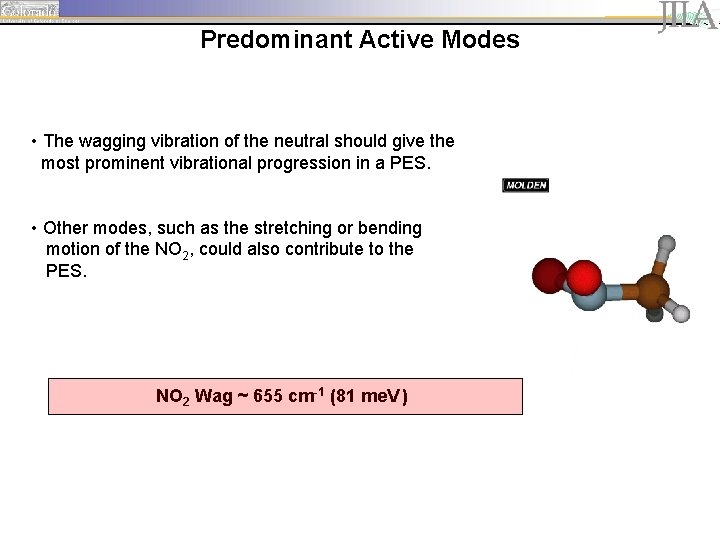
Predominant Active Modes • The wagging vibration of the neutral should give the most prominent vibrational progression in a PES. • Other modes, such as the stretching or bending motion of the NO 2, could also contribute to the PES. NO 2 Wag ~ 655 cm-1 (81 me. V )
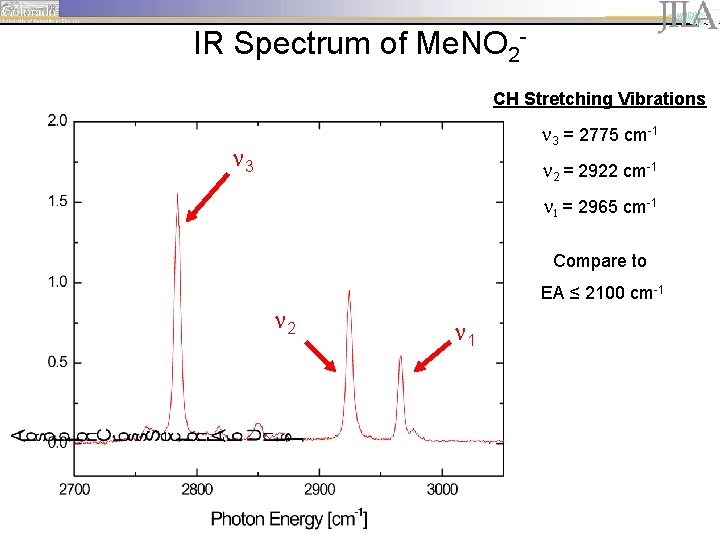
IR Spectrum of Me. NO 2 CH Stretching Vibrations 3 = 2775 cm-1 3 2 = 2922 cm-1 1 = 2965 cm-1 Compare to EA ≤ 2100 cm-1 2 1
Vibrational Autodetachment Dark States ZOBS
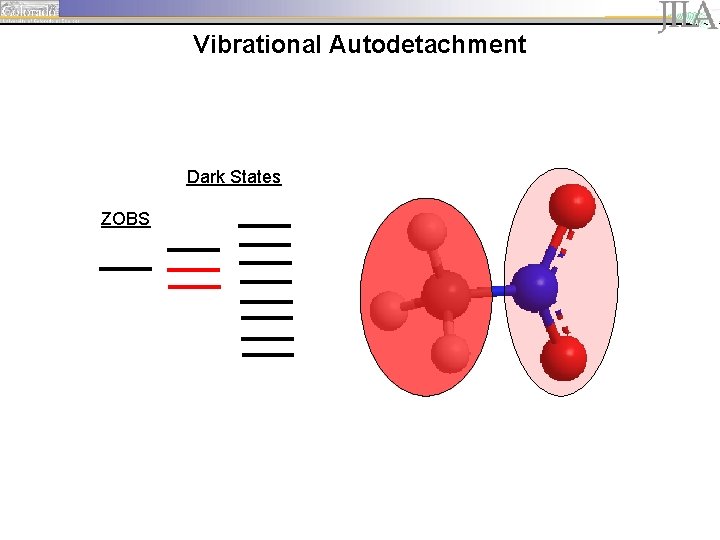
Vibrational Autodetachment Dark States ZOBS
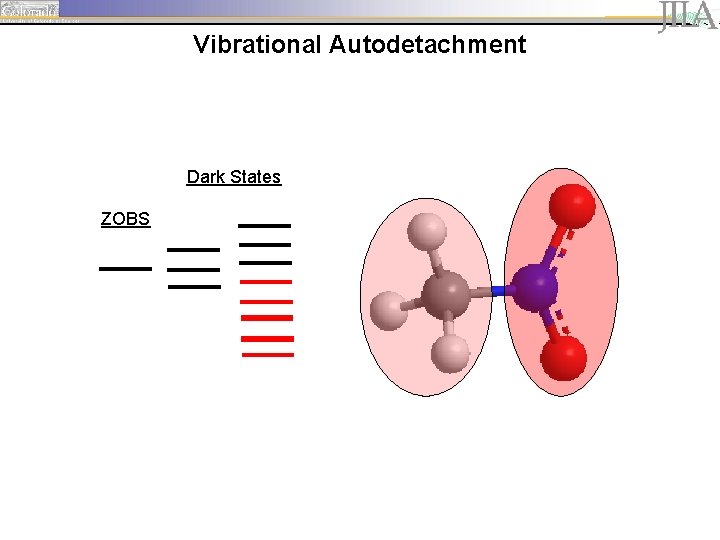
Vibrational Autodetachment Dark States ZOBS
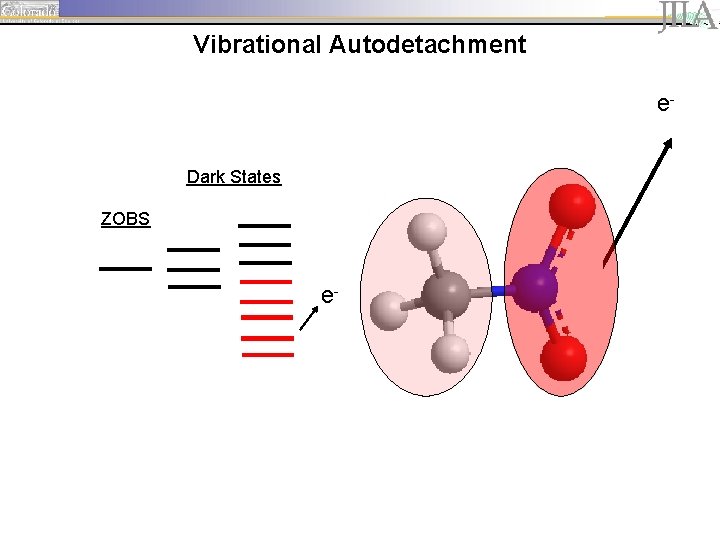
Vibrational Autodetachment e. Dark States ZOBS e-
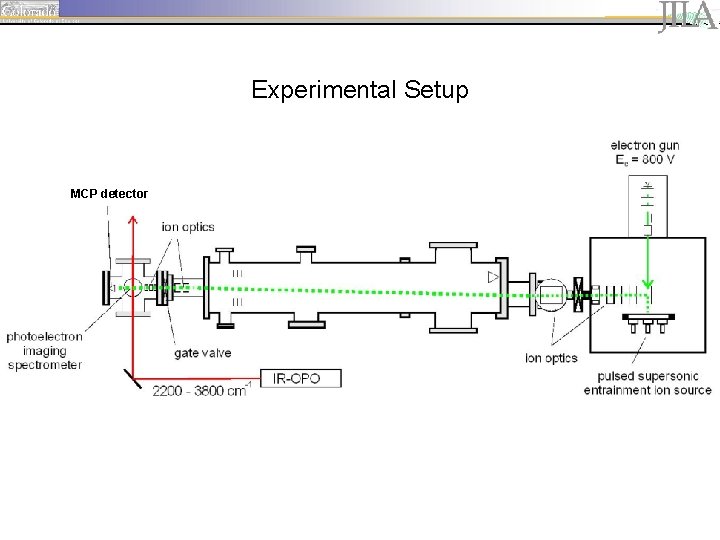
Experimental Setup MCP detector
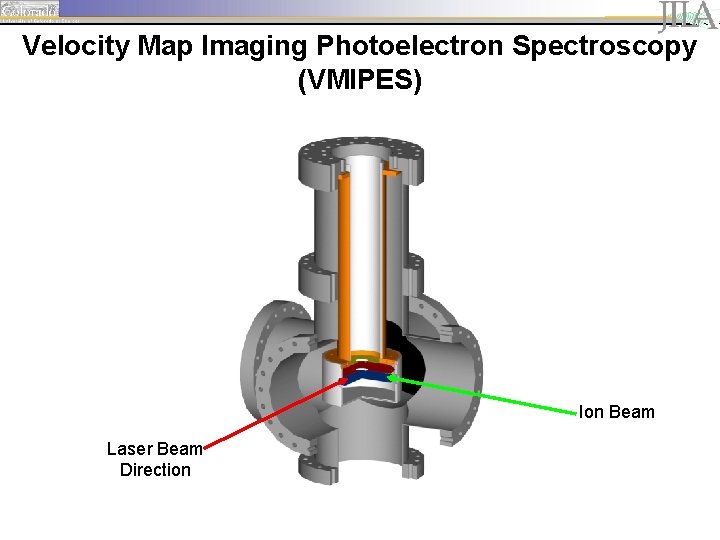
Velocity Map Imaging Photoelectron Spectroscopy (VMIPES) Ion Beam Laser Beam Direction
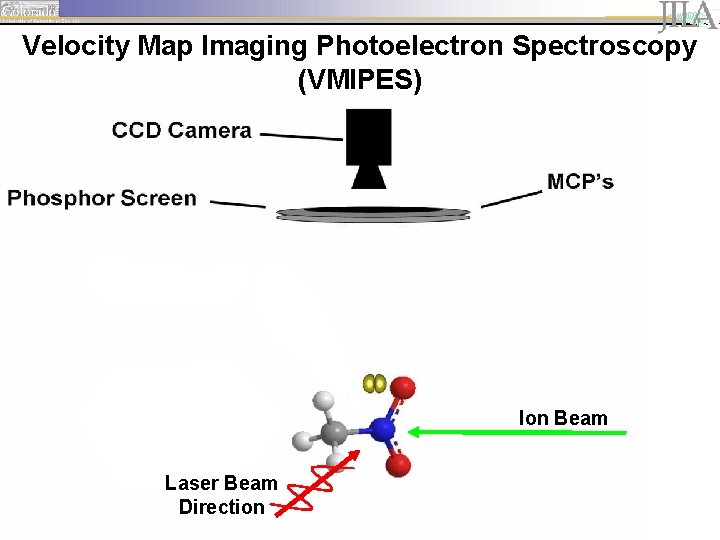
Velocity Map Imaging Photoelectron Spectroscopy (VMIPES) Ion Beam Laser Beam Direction
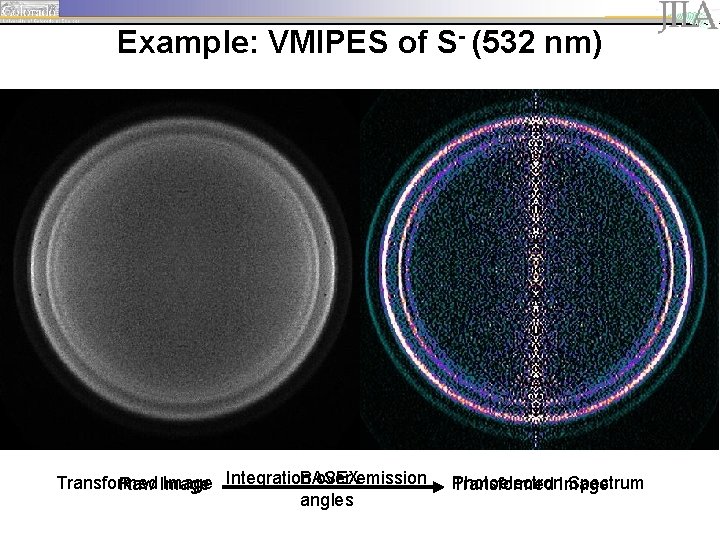
Example: VMIPES of S- (532 nm) BASEX over emission Transformed Image Integration Raw Image angles Photoelectron Spectrum Transformed Image

…and what can we learn from the experiment? • determine electron affinity • assign vibrational features in PES
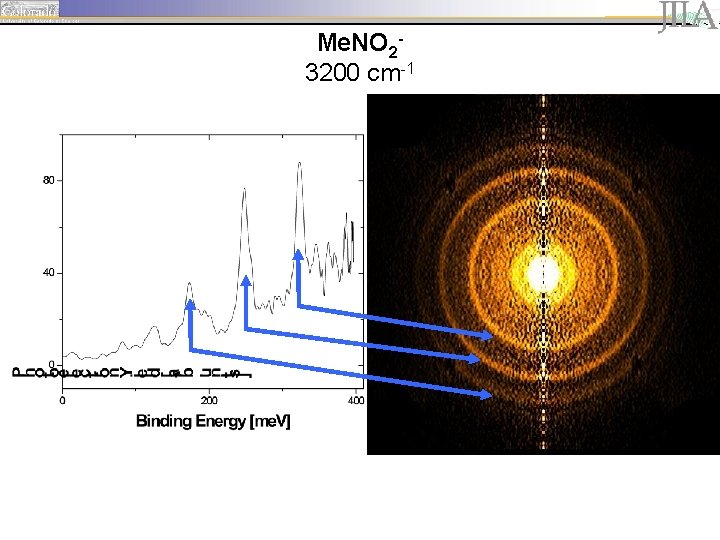
1 Me. NO 23200 cm-1
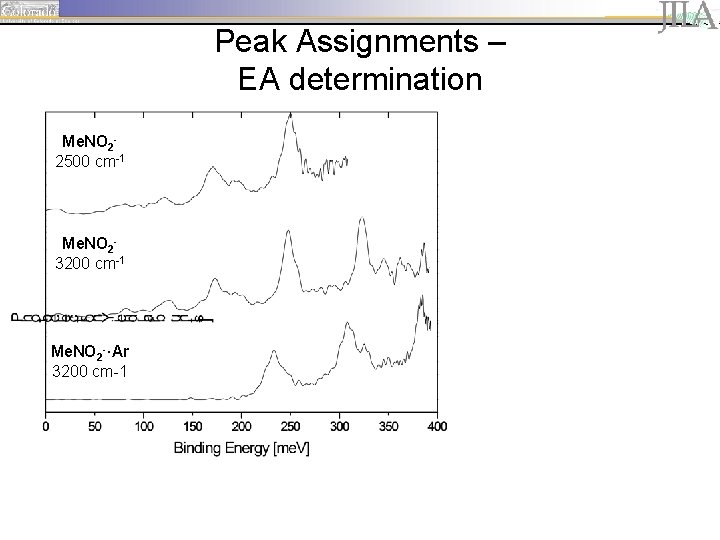
Peak Assignments – EA determination Me. NO 22500 cm-1 Me. NO 23200 cm-1 Me. NO 2 -·Ar 3200 cm-1
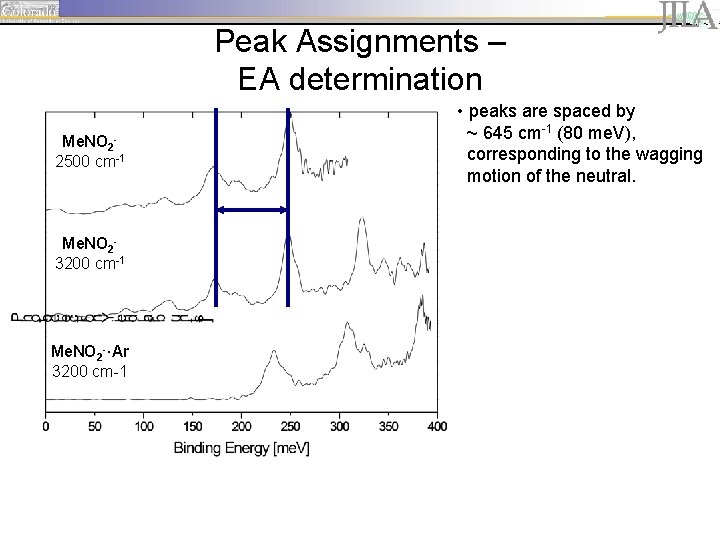
Peak Assignments – EA determination Me. NO 22500 cm-1 Me. NO 23200 cm-1 Me. NO 2 -·Ar 3200 cm-1 • peaks are spaced by ~ 645 cm-1 (80 me. V), corresponding to the wagging motion of the neutral.
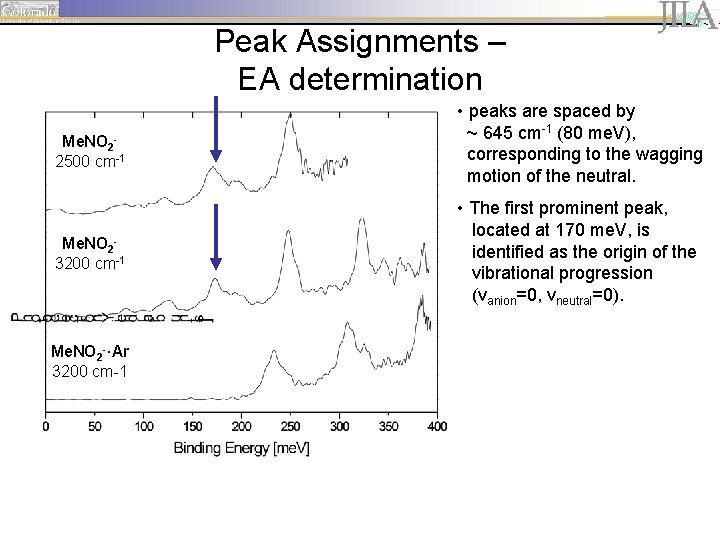
Peak Assignments – EA determination Me. NO 22500 cm-1 Me. NO 23200 cm-1 Me. NO 2 -·Ar 3200 cm-1 • peaks are spaced by ~ 645 cm-1 (80 me. V), corresponding to the wagging motion of the neutral. • The first prominent peak, located at 170 me. V, is identified as the origin of the vibrational progression (vanion=0, vneutral=0).
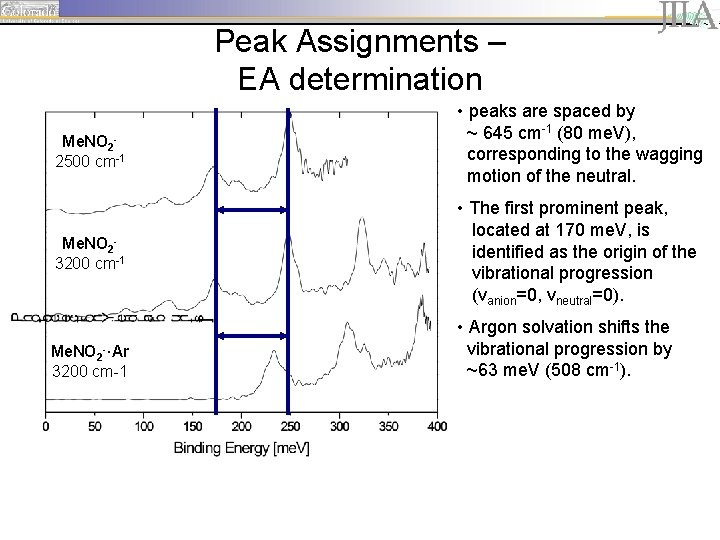
Peak Assignments – EA determination Me. NO 22500 cm-1 Me. NO 23200 cm-1 Me. NO 2 -·Ar 3200 cm-1 • peaks are spaced by ~ 645 cm-1 (80 me. V), corresponding to the wagging motion of the neutral. • The first prominent peak, located at 170 me. V, is identified as the origin of the vibrational progression (vanion=0, vneutral=0). • Argon solvation shifts the vibrational progression by ~63 me. V (508 cm-1).
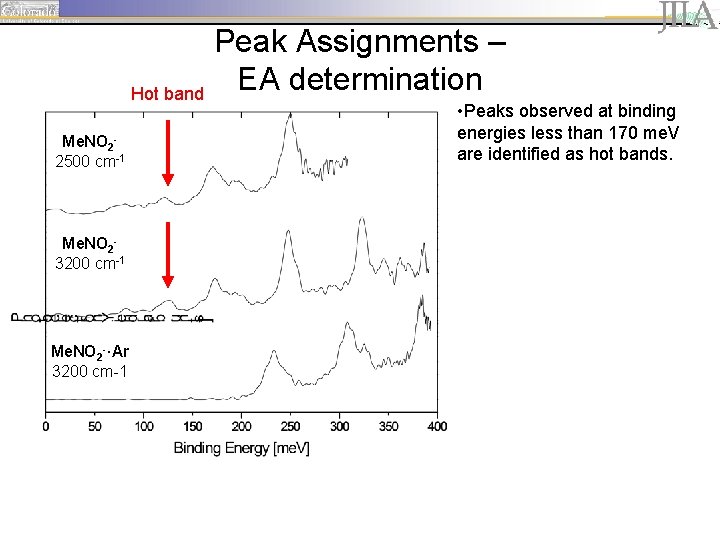
Hot band Me. NO 22500 cm-1 Me. NO 23200 cm-1 Me. NO 2 -·Ar 3200 cm-1 Peak Assignments – EA determination • Peaks observed at binding energies less than 170 me. V are identified as hot bands.
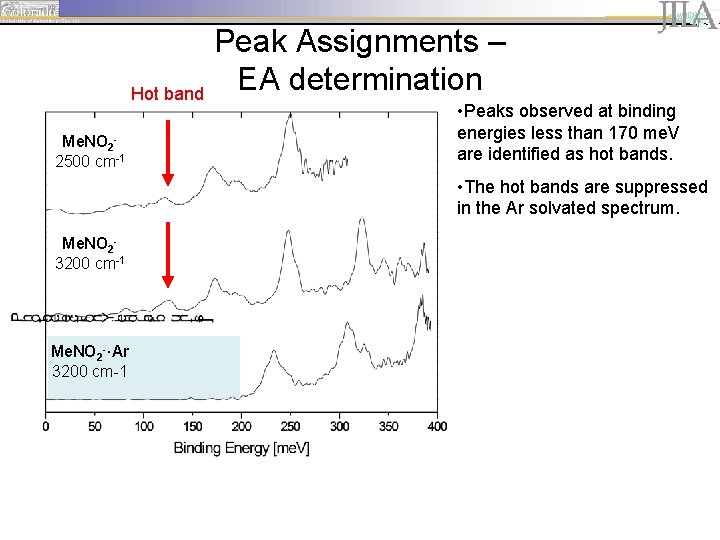
Hot band Me. NO 22500 cm-1 Peak Assignments – EA determination • Peaks observed at binding energies less than 170 me. V are identified as hot bands. • The hot bands are suppressed in the Ar solvated spectrum. Me. NO 23200 cm-1 Me. NO 2 -·Ar 3200 cm-1
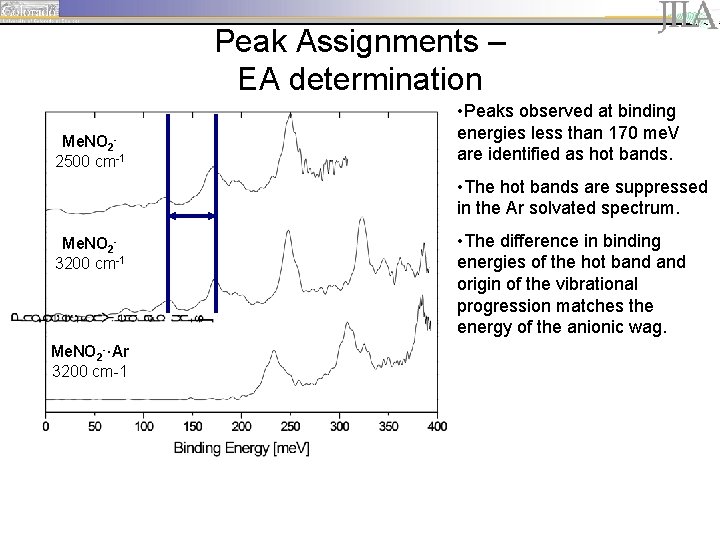
Peak Assignments – EA determination Me. NO 22500 cm-1 • Peaks observed at binding energies less than 170 me. V are identified as hot bands. • The hot bands are suppressed in the Ar solvated spectrum. Me. NO 23200 cm-1 Me. NO 2 -·Ar 3200 cm-1 • The difference in binding energies of the hot band origin of the vibrational progression matches the energy of the anionic wag.

New Assignments New value for EA: 170 ± 4 me. V 1370 ± 30 cm-1 Old value: 260 ± 80 me. V
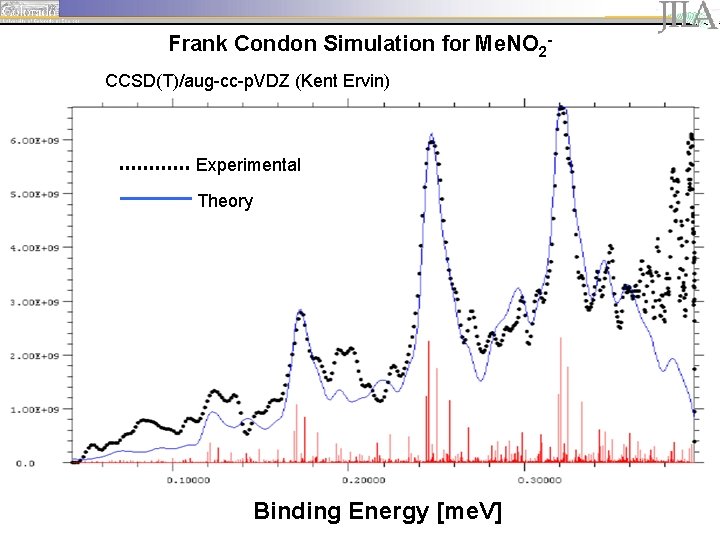
Frank Condon Simulation for Me. NO 2 CCSD(T)/aug-cc-p. VDZ (Kent Ervin) Experimental Theory Binding Energy [me. V]

…and what can we learn from the experiment? • determine electron affinity • assign vibrational features in PES

…and what can we learn from the experiment? • determine electron affinity • assign vibrational features in PES • effects of vibrational excitation on photoelectron spectra
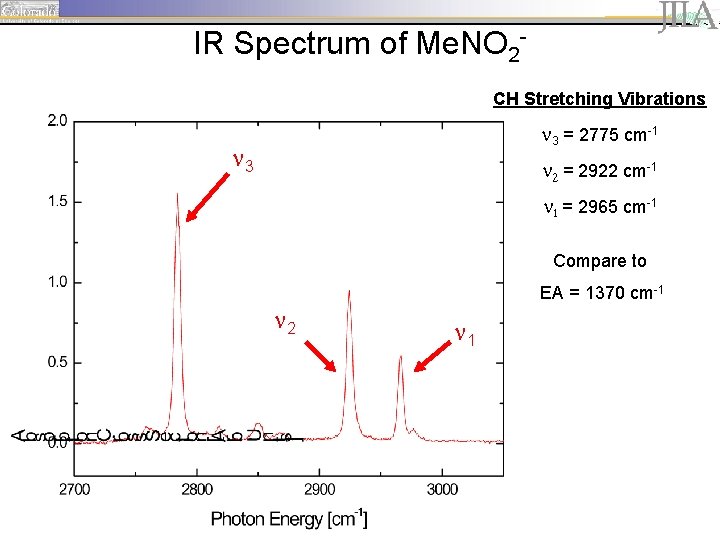
IR Spectrum of Me. NO 2 CH Stretching Vibrations 3 = 2775 cm-1 3 2 = 2922 cm-1 1 = 2965 cm-1 Compare to EA = 1370 cm-1 2 1
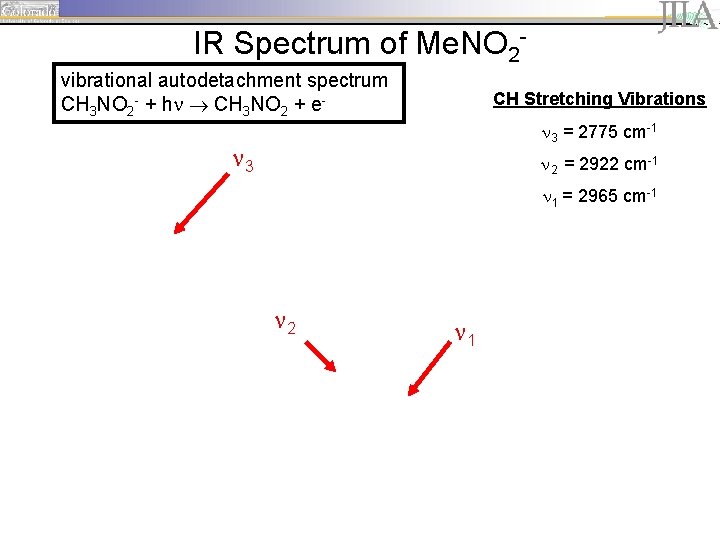
IR Spectrum of Me. NO 2 vibrational autodetachment spectrum CH 3 NO 2 - + h CH 3 NO 2 + e- CH Stretching Vibrations 3 = 2775 cm-1 3 2 = 2922 cm-1 1 = 2965 cm-1 2 1
IR Spectrum of Me. NO 2 3

Comparison of Off and On Resonance Spectra 3200 cm-1 (off resonance) 2775 cm-1 (on resonance) Kinetic Energy
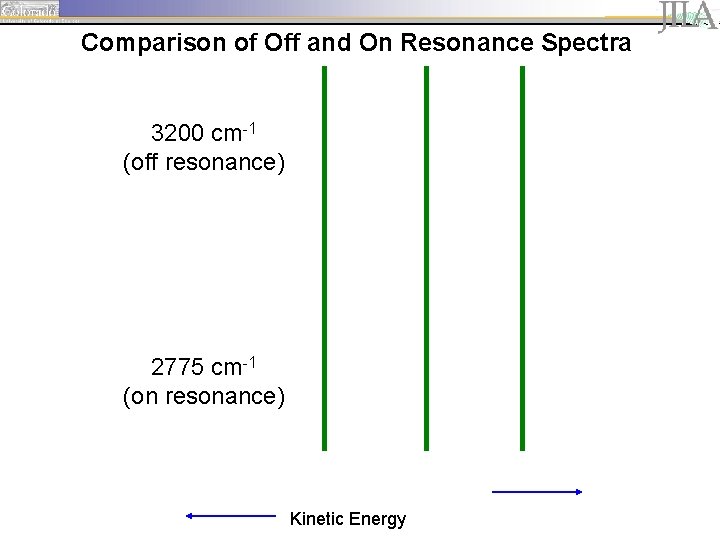
Comparison of Off and On Resonance Spectra 3200 cm-1 (off resonance) 2775 cm-1 (on resonance) Kinetic Energy

Summary • The adiabatic electron affinity is found to be (170 4) me. V (1370 32) cm-1 • A progression belonging to the NO 2 wagging mode is the most prominent feature in the PES • Considerable contrast is observed between the electron kinetic energy distribution in vibrational autodetachment and direct photodetachment. • The data suggest redistribution of vibrational energy before electron emission and retention of vibrational energy in the molecule, leading to emission of low-energy electrons

Acknowledgements Weber group: • Mathias Weber • Holger Schneider • Jesse Marcum Lineberger Lab • Carl Lineberger • Lenny Sheps • Elisa Miller • Kent M. Ervin, University of Nevada, Reno

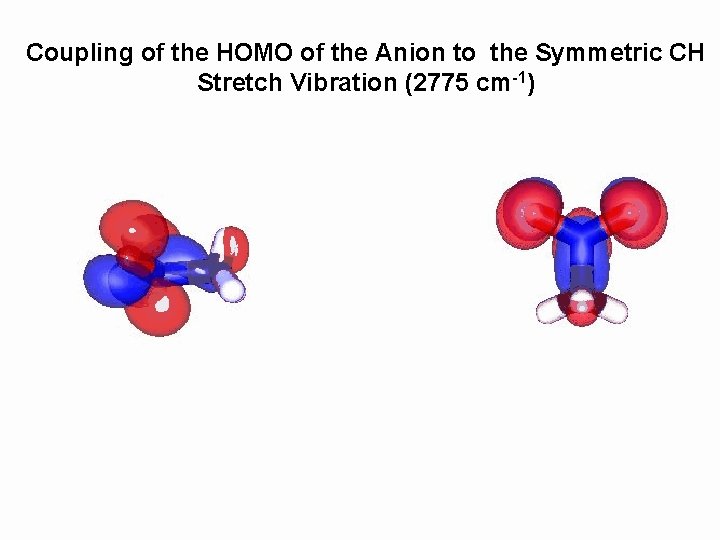
Coupling of the HOMO of the Anion to the Symmetric CH Stretch Vibration (2775 cm-1)

Comparison of On and Off Resonance Images On Resonance Image Off Resonance Image
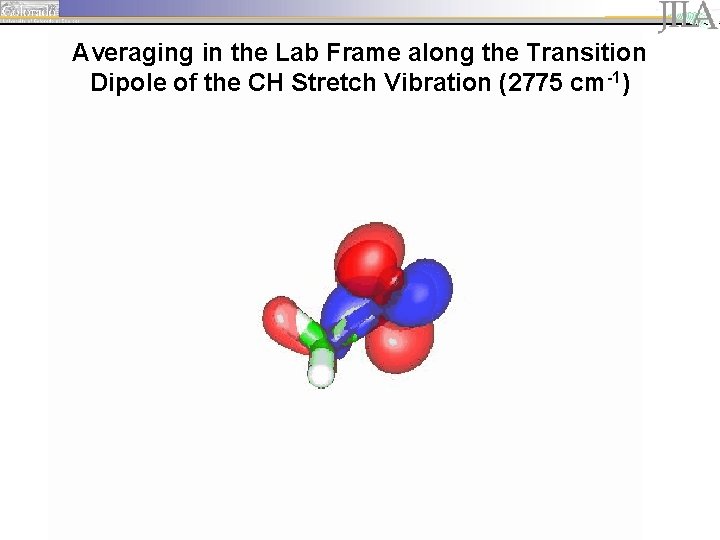
Averaging in the Lab Frame along the Transition Dipole of the CH Stretch Vibration (2775 cm-1)

Potential Energy Surfaces for Neutral Me. NO 2 and the Valence Anion J. M. Weber et al. , JCP 115 (2001) 10718
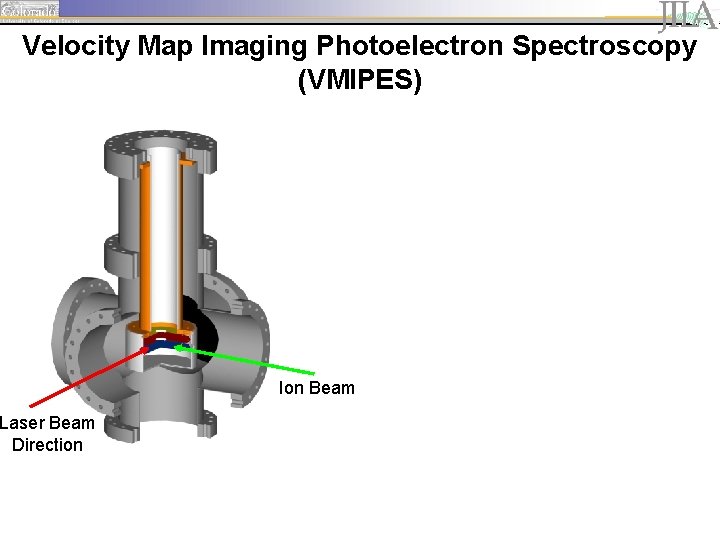
Velocity Map Imaging Photoelectron Spectroscopy (VMIPES) Laser Beam Direction Ion Beam
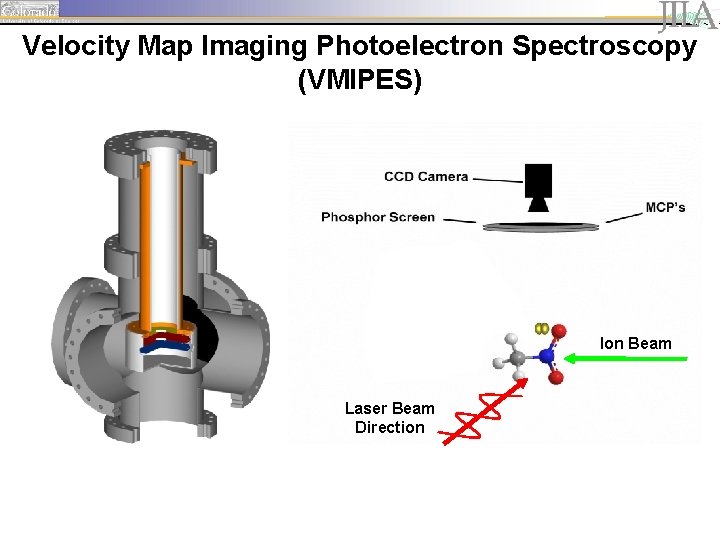
Velocity Map Imaging Photoelectron Spectroscopy (VMIPES) Ion Beam Laser Beam Direction
- Slides: 47