Photoelectrochemistry ch 18 Introduction of Luminescence Electrogenerated Chemiluminescence













- Slides: 15

Photoelectrochemistry (ch. 18) Introduction of Luminescence Electrogenerated Chemiluminescence Photochemistry at Semiconductors

Radiation energy electrical or chemical energy e. g. , ECL, electrochromic device, EL, sensors 1. General Concepts of luminescence the type of excitation - Photoluminescence: light emission by UV or visible light - Radioluminescence (scintillation): excited by radioactive substances - Cathodoluminescence: excited by high velocity electron bombardment - X-ray luminescence: by X-rays - Chemiluminescence: by chemical reactions -Electrochemiluminescence or electrogenerated chemiluminescence: by electrochemical reactions - Electroluminescence: by electric voltage Luminescent materials (or luminophors): substances which exhibit luminescence - organic (organoluminophors) - inorganic (phosphors)

2. Organoluminophors cf. B. M. Krasovitskii, B. M. Bolotin, Organic Luminescent Materials, VCH (1988). Electronic spectra - by energy transitions between unexcited (ground) and excited states of molecules absorption ( ) vs. emission (luminescence, ) spectrum - sublevels (vibrational & rotational), 0 -0 band, Stoke’s law (by nonradiative losses) - deviation from mirror symmetry of absorption & luminescence; intra- & intermolecular processes, e. g. , changes in the structure of molecules in the excited state


- luminescence intensity: quantum yield or quantum efficiency: ratio between the emitted and absorbed quanta (occurrence of nonradiative processes lower the quantum yield) -time interval during which they emit light in the excited state; duration of light emission after excitation has stopped fluorescence (10 -9 -10 -7 s) or phosphorescence (10 -4 -10 -2 s) excited states of molecules - singlet state (S*); antiparallel spins, multiplicity, 2 S + 1 = 1 - triplet state (T); multiplicity = 3



- sensitization & inhibition of fluorescence applications of organic luminescent materials fluorescent pigments & paints. dye for plastics & fibers, optical brightening agents, organic scintillators, lasers, electrochemiluminescent or chemiluminescent compositions, analytical chemistry, biology & medicine

3. Inorganic phosphors phosphor: a solid which converts certain types of energy into electromagnetic radiation over and above thermal radiation luminescence

e. g. , Al 2 O 3: Cr 3+ (ruby, red), Y 2 O 3: Eu 3+, host lattice + luminescenter (activator) - host lattice: hold luminescent ion tightly - efficient luminescent materials: need to suppress nonradiative process - if exciting radiation is not absorbed by the activator add another ion to transfer the excitation radiation to the activator “sensitizer” e. g. , Ca 5(PO 4)3 F: Sb 3+, Mn 2+

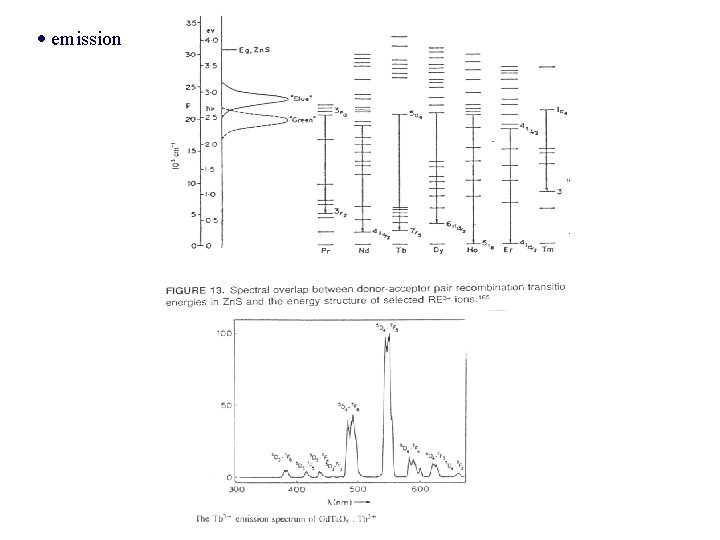
- luminescent molecules e. g. , bipyridine + Eu 3+ i) the bipyridine cage protects Eu 3+ ion against aqueous surroundings which try to quench luminescence ii) excitation radiation bipyridine molecule absorb & transfer it to Eu 3+ ion red luminescence How does a luminescent material absorb its excitation energy? - quantum mechanics: coordination diagram, energy level diagrams of ions

emission


nonradiative transitions; efficiency? energy transfer applications lamps, cathode ray, X-ray phosphor, electroluminescence, laser probe, immunoassay, 4. Electroluminescence luminescent material can be excited by application of an electric voltage applied voltage - low field EL: light emitting diodes (LED, energy is injected into a p-n junction, a few volts), laser diodes (semiconductor lasers); normally DC - high field EL (> 106 Vcm-1): display, thin film EL, Zn. S EL; normally AC (ACEL)

low field EL: LED & semiconductor lasers - LED band to band transition