Photoelectric Effect And Quantum Mechanics The Ultraviolet Catastrophe










- Slides: 11

Photoelectric Effect And Quantum Mechanics

The Ultraviolet Catastrophe • Classical physics could not explain the distribution of radiation from a hot body. (Intensity as a function of wavelength)

Oscillators • Max Plank postulated that energy was emitted by resonators which could only absorb and give off discrete amounts of energy. • The energy of each resonator is given by E = nhf where n is an integer, f is the frequency, and h is PLANK’S CONSTANT.

Quanta • Because the energy from the resonators comes in discrete units (multiples of Plank’s constant x frequency) it is said to be quantized. • The quantum of light is called a PHOTON. • A “resonator” (atom or molecule) radiates or absorbs energy when it changes quantum states. This energy corresponds to the difference between two energy levels.

The Photoelectric Effect • A beam of light shining on a metal surface adds energy to the atoms’ electrons, sometimes enough to loosen them from their atoms and “eject” them.

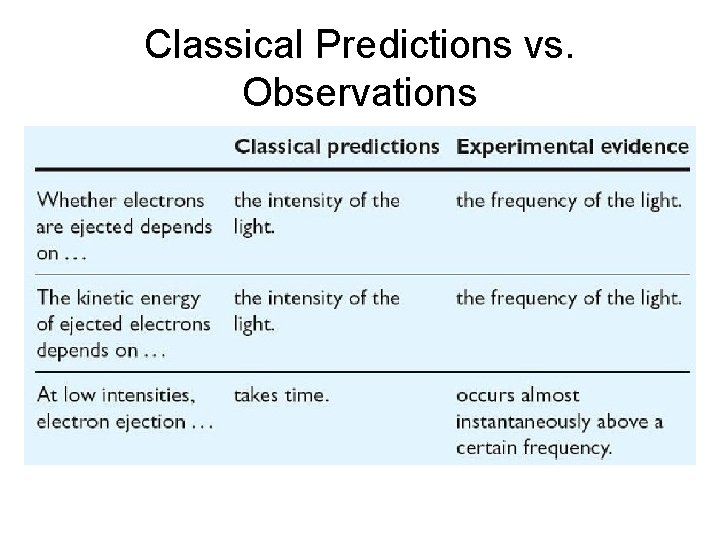
Predictions of Classical Phyiscs • Classical physical laws predicted – Any wavelength of light, if shined long enough, should add enough energy to eject electrons – The stronger the intensity of the light the more electrons should be ejected – The kinetic energy of the ejected electrons depend should depend on the intensity of the light.

Experimental Evidence • Whether electrons are ejected or not is not a function of intensity but of FREQUENCY of the incident light beam. • The kinetic energy of the ejected electrons depends on the FREQUENCY of the incident light beam, not the intensity. • Even at low intensities, electrons are ejected almost immediately.

Einstein • Einstein won a Nobel Prize, not for his work in relativity, but for explaining the photoelectric effect. • He proposed that electromagnetic waves are quantized. • EM waves can be understood as a stream of particles (photons) • Each photon has Energy = hf

Threshold Frequency • The minimum frequency of light needed to eject an electron is called the threshold frequency. • If the frequency of the incident light exceeds the threshold frequency, the photoelectric effect is observed; if the frequency is lower than the threshold frequency, no electrons are ejected.

Work Function • The amount of energy each electron needs to escape the metal is known as the WORK FUNCTION. • The work function is equal to hft where ft is the threshold frequency of the metal (and h is Plank’s constant). • Different metals have different work functions.

Classical Predictions vs. Observations