Photodynamic modulation of wound healing and inhibition of

Photodynamic modulation of wound healing and inhibition of tissue degradation Michael R Hamblin, Tatiana Demidova, Faten Gad, Touqir Zahra, Tayyaba Hasan. Wellman Center for Photomedicine Massachusetts General Hospital Harvard Medical School Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Outline Introduction to PDT Ø Antimicrobial PDT Ø Bioluminescence imaging of infection Ø PDT of infected wounds Ø Inhibition of tissue degradation by bacterial toxins Ø PDT stimulation of wound healing Ø Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
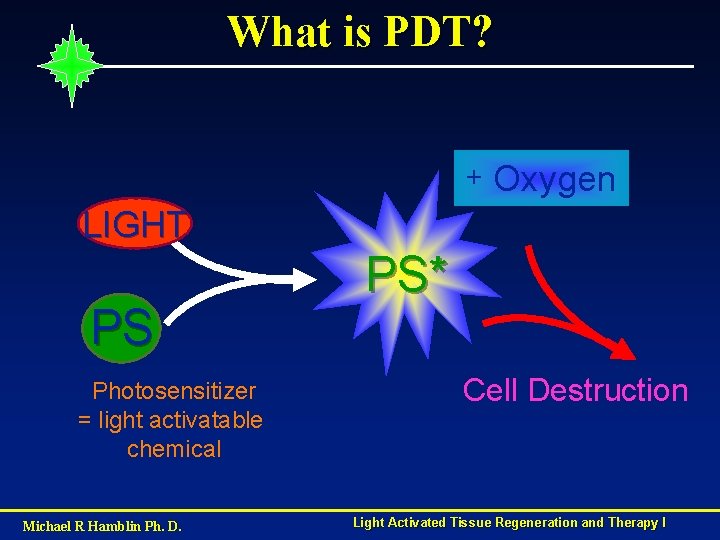
What is PDT? + Oxygen LIGHT PS Photosensitizer = light activatable chemical Michael R Hamblin Ph. D. PS* Cell Destruction Light Activated Tissue Regeneration and Therapy I

PDT for cancer LASER Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Components Photosensitizer (PS) : dye which absorbs visible light Visible light of the correct wavelength - red light penetrates tissue better Oxygen Target tissue or cells Dual selectivity for target - selective accumulation of PS spatial control of illumination Originally a cancer treatment - now other additional Applications have emerged Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
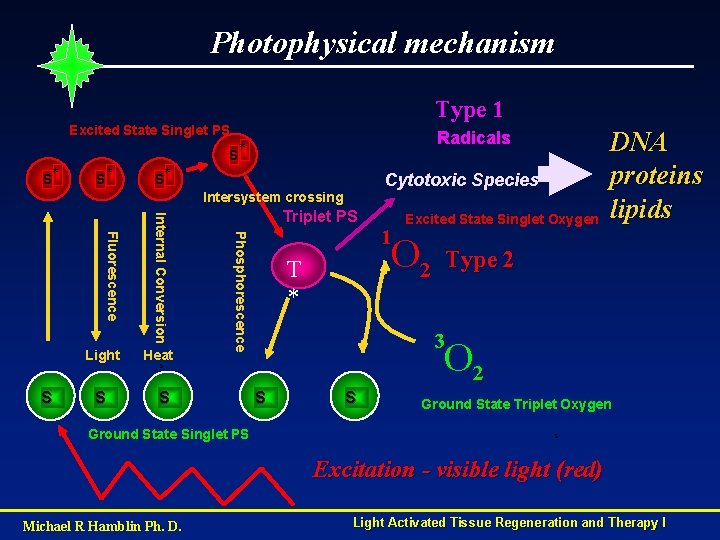
Photophysical mechanism Type 1 Excited State Singlet PS Radicals S* S* Cytotoxic Species Intersystem crossing S Heat Phosphorescence S Internal Conversion Fluorescence Light Triplet PS O 2 T * Ground State Singlet PS Type 2 3 O 2 S S 1 Excited State Singlet Oxygen DNA proteins lipids S S Ground State Triplet Oxygen S Excitation - visible light (red) Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
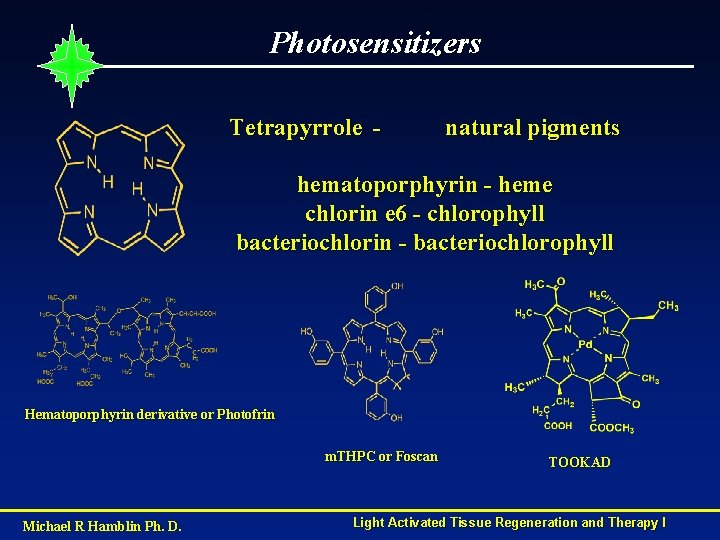
Photosensitizers Tetrapyrrole - natural pigments hematoporphyrin - heme chlorin e 6 - chlorophyll bacteriochlorin - bacteriochlorophyll Hematoporphyrin derivative or Photofrin m. THPC or Foscan Michael R Hamblin Ph. D. TOOKAD Light Activated Tissue Regeneration and Therapy I

Why do we use PDT against microorganisms? n For localized infections (not systemic) n Photosensitizer is delivered locally to infection n World-wide increase in multi-antibiotic resistant bacteria n Systemic antibiotics cannot get into dead or damaged tissue n Even if antibiotics work they take several days Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Antibiotic resistance • 70 percent of bacteria that cause infections in hospitals are resistant to at least one antibiotic • Some organisms are resistant to all approved antibiotics • Food-producing animals • Antibiotics are given to patients more often than necessary • Patients who are prescribed antibiotics don't complete course • Previously treatable diseases that have again become untreatable, as in the days before antibiotics were developed Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

What do we know about antimicrobial PDT? n n n n Known for over 100 years Gram (+) species easily killed by usual (tumor) PS + light Gram (-) species need cationic PS or special means to increase bacterial permeability Positively charged PS are more effective Some PS do not have to bind or penetrate to be effective Other PS must penetrate Mechanisms of killing: cytoplasmic membrane damage, (DNA damage? ) Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Background Many naturally occurring antibacterial peptides are polycationic - e. g. defensins, cecropins, magainins, histatins, and defensins. Synthetic polycations are used as antibacterials Polycations permeabilize gram negative bacteria Cationic dyes are known to be efficient photosensitizers of both classes of bacteria Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Hypothesis Polycationic covalent conjugates between poly-l-lysine and chlorin e 6 may efficiently target photodestruction to bacteria Gram positive - increased binding and diffusion through porous peptidoglycan layer Gram negative - increased binding and penetration by perturbation of outer lipid bilayer Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Choice of PS Good absorption peak at 660 nm Good fluorescence & singlet oxygen quantum yields Easy conjugation chemistry via carbodiimide activation of COOH group Readily available Purification of conjugates is crucial Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Synthesis Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
Conjugates H 2 C pl-ce 6 37 lysines 1 ce 6 CH 3 H 2 N H 3 C H O N H N CH 3 NH 2 H 2 N H COOH O O O NH NH N N H H HN NH 2 H 2 N O O NH NH O H N NH O O O NH 2 H 2 N O O NH 2 HN O N H O H 2 N NH 2 N H H 2 N H N O O N H H 2 N HN H N O H 2 N O HN H 2 N O NH 2 HN O NH 2 H N O NH 2 O N H NH 2 H N NH 2 O H O O N N H NH O N H NH 2 O O O HO HN NH 2 O N H HN NH H 2 N Michael R Hamblin Ph. D. NH O O NH COOH H 2 N O H 2 N NH 2 CH 3 H 2 N NH 2 O O N H NH 2 Light Activated Tissue Regeneration and Therapy I
Bioluminescence imaging =Bacteria have been genetically engineered to emit light by transfection with plasmid containing lux operon from Photorhabdus luminescens (antibiotic selection necessary) =Bacteria have been stably transduced with entire lux operon (lux ABCDE) or (lux CDABE) (chromosomal integration) =Genes code for luciferase and biosynthetic enzymes responsible for producing substrate =Bacteria “glow in the dark” using their own ATP and oxygen Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
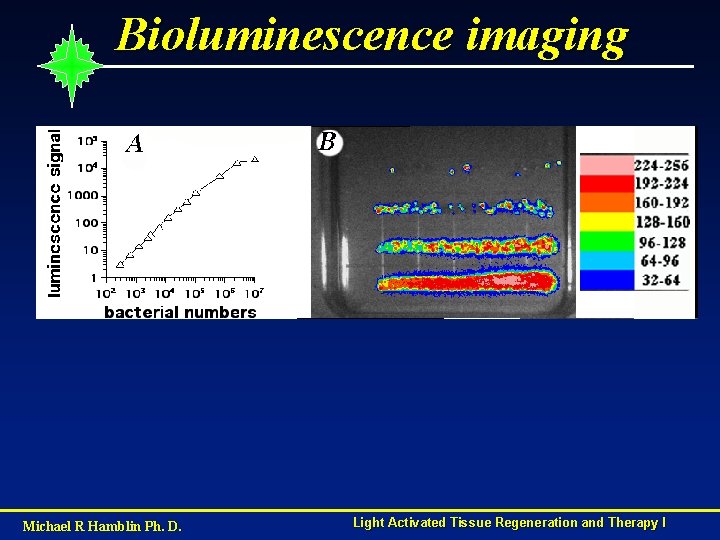
Bioluminescence imaging Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
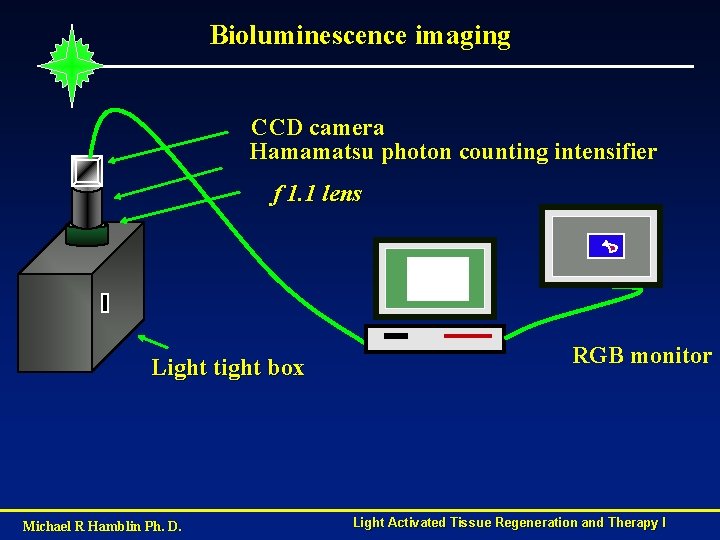
Bioluminescence imaging CCD camera Hamamatsu photon counting intensifier f 1. 1 lens Light tight box Michael R Hamblin Ph. D. RGB monitor Light Activated Tissue Regeneration and Therapy I

Application of bacteria to wounded mouse Hamblin MR, O’Donnell DA, Murthy N, Contag CH, and Hasan T. Rapid control of wound infections by targeted photodynamic therapy monitored by in vivo bioluminescence imaging. Photochem Photobiol, 75: 51 -57, 2002 Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Addition of PS to infected wounds Non-pathogenic strain of E. coli (DH 5 alpha) Add 50 µL of a 100 µM solution of p. L-ce 6 conjugate to wounds 1 & 4 30 minutes after bacteria and deliver light after a further 30 minutes Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Light delivery from a 660 nm diode laser Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
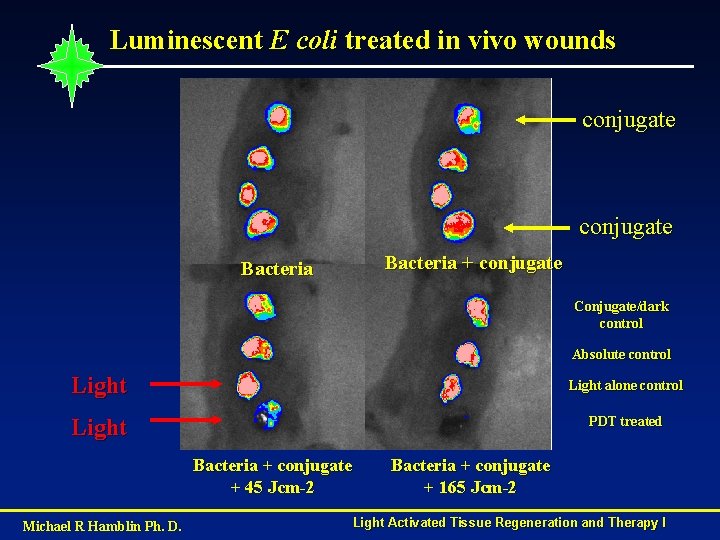
Luminescent E coli treated in vivo wounds conjugate Bacteria + conjugate Bacteria Conjugate/dark control Absolute control Light alone control Light PDT treated Bacteria + conjugate + 45 Jcm-2 Michael R Hamblin Ph. D. Bacteria + conjugate + 165 Jcm-2 Light Activated Tissue Regeneration and Therapy I

Luminescent E coli treated in vivo wounds Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Treatment does not impair healing Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

However…… This strain of E. coli (DH 5 alpha) is non-pathogenic Use a pathogenic Gram-ve bacterium Pseudomonas aeruginosa (strain 180, causes septicemia in rats and mice from infected burns) Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Single wound for P aeruginosa infection Hamblin MR, Zahra T, Contag CH, Mc. Manus AT, and Hasan T. Optical monitoring and treatment of potentially lethal wound infections in vivo. J Infectious Disease, 2003, 187: 1717 -1725 Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
Reproducibility of wounds Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Infection of wounds Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Determination of LD 50 200, 000 P. aeruginosa Treatment dose selected to be 5, 000 (25 X LD 50) Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
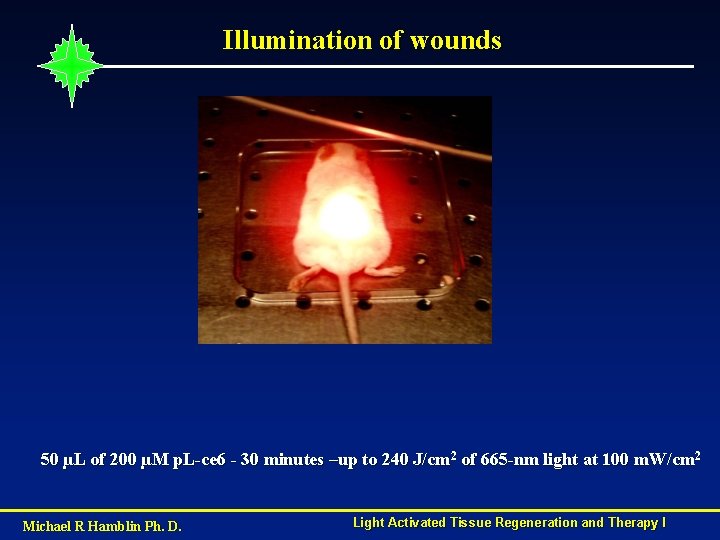
Illumination of wounds 50 µL of 200 µM p. L-ce 6 - 30 minutes –up to 240 J/cm 2 of 665 -nm light at 100 m. W/cm 2 Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Luminescent P. aeruginosa treated in vivo wounds Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
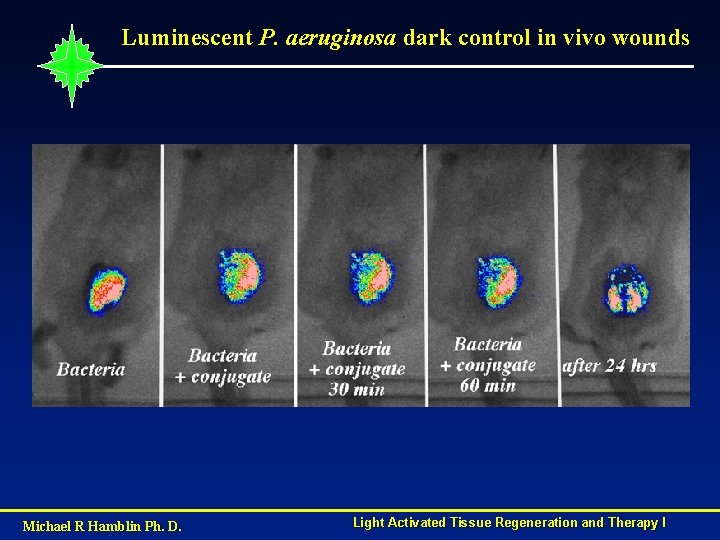
Luminescent P. aeruginosa dark control in vivo wounds Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Luminescent P aeruginosa treated in vivo wounds Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
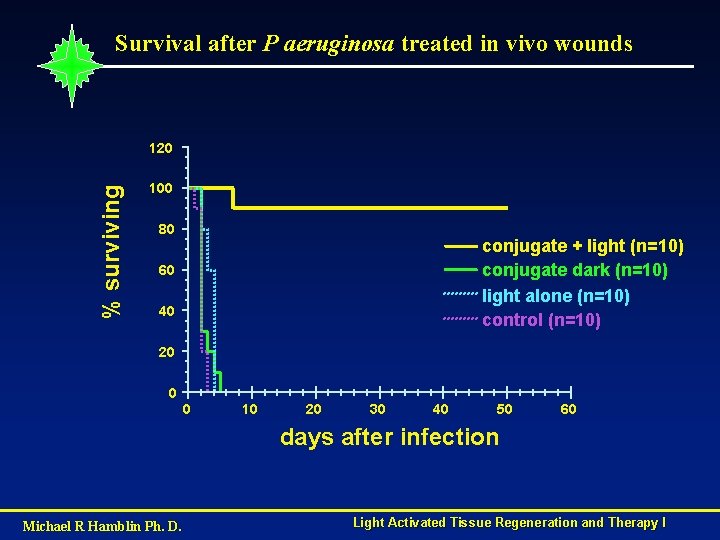
Survival after P aeruginosa treated in vivo wounds % surviving 120 100 80 conjugate + light (n=10) conjugate dark (n=10) light alone (n=10) control (n=10) 60 40 20 0 0 10 20 30 40 50 60 days after infection Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
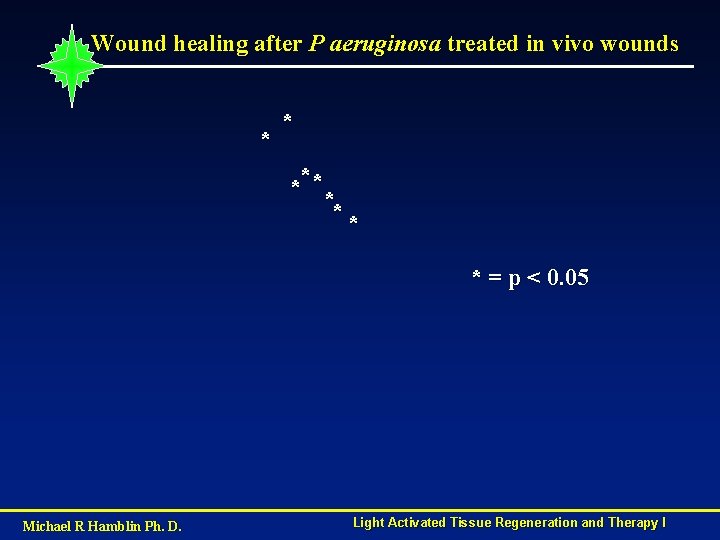
Wound healing after P aeruginosa treated in vivo wounds * * **** ** * = p < 0. 05 Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
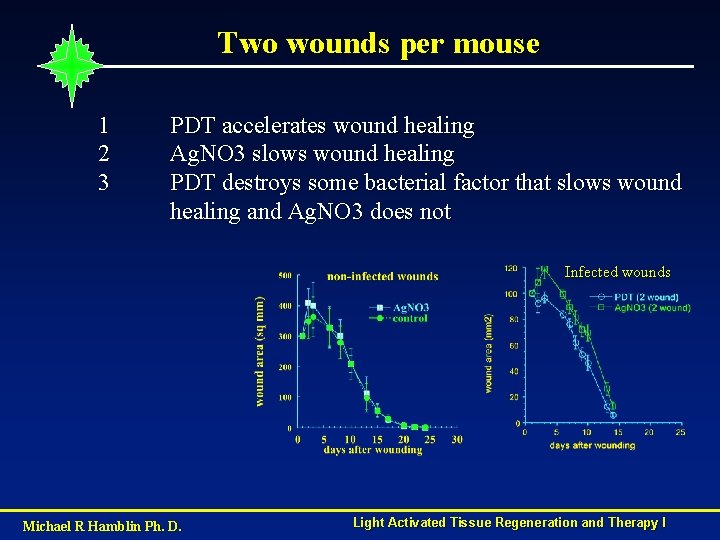
Two wounds per mouse 1 2 3 PDT accelerates wound healing Ag. NO 3 slows wound healing PDT destroys some bacterial factor that slows wound healing and Ag. NO 3 does not Infected wounds Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Conclusions Photodestruction of Gram bacteria in wounds is possible in vivo Mice can be saved from an otherwise lethal infection Bacterial virulence factors that slow wound healing may also be destroyed For P. aeruginosa these may be exotoxin A, proteases, lipase elastase etc. Since many non-healing wounds are infected PDT may have dual function Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Stimulation of wound healing by PDT Systemic PDT is likely to inhibit wound healing due to cellular and vascular damage Therefore topical PDT is tested for wound healing Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
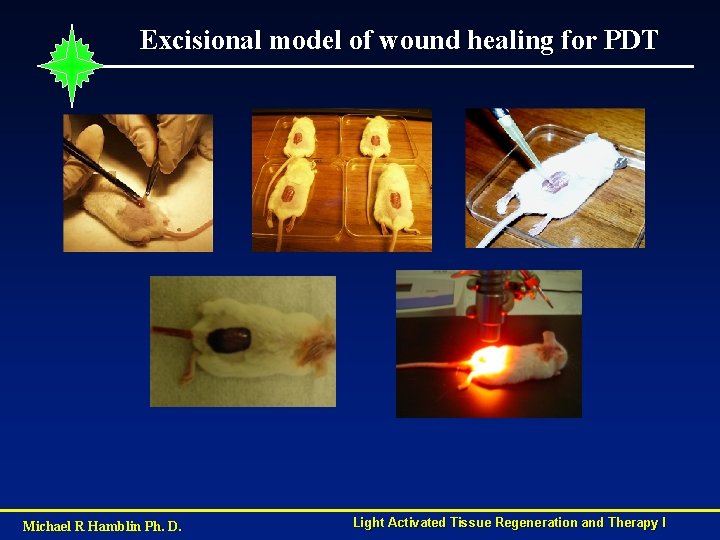
Excisional model of wound healing for PDT Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
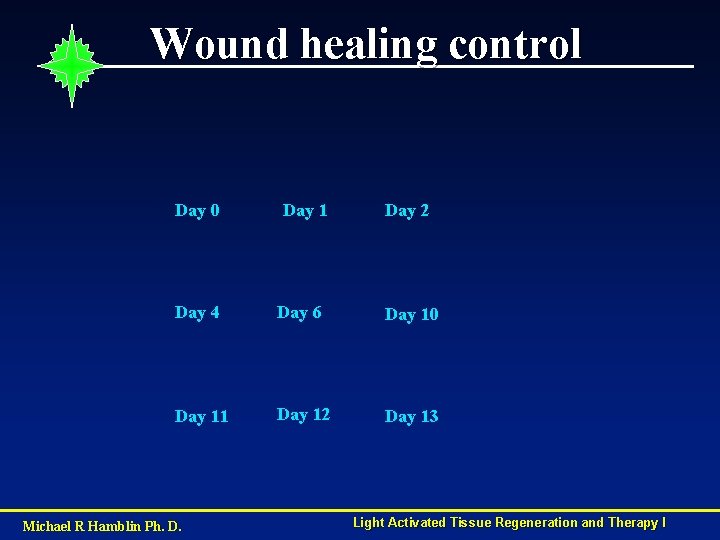
Wound healing control Day 0 Day 1 Day 2 Day 4 Day 6 Day 10 Day 11 Day 12 Day 13 Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Topical PDT with BPD + 690 -nm light improves wound healing 50 µL of 100 µM BPD - 30 minutes -100 J/cm 2 of 690 -nm light at 100 m. W/cm 2 Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
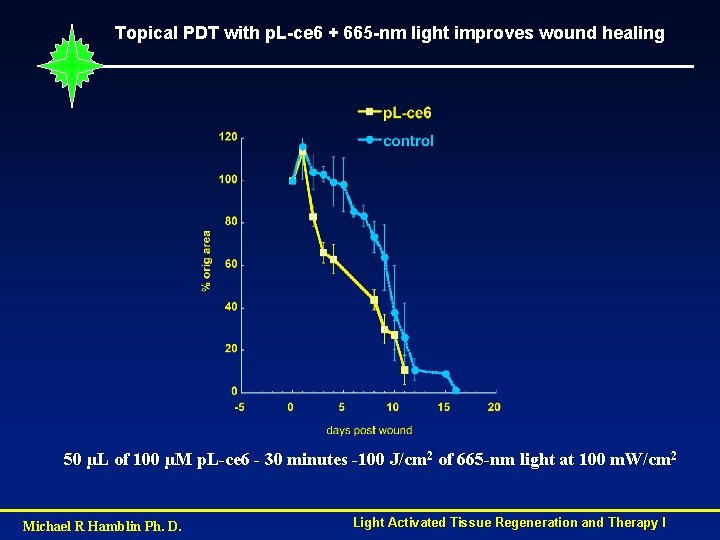
Topical PDT with p. L-ce 6 + 665 -nm light improves wound healing 50 µL of 100 µM p. L-ce 6 - 30 minutes -100 J/cm 2 of 665 -nm light at 100 m. W/cm 2 Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
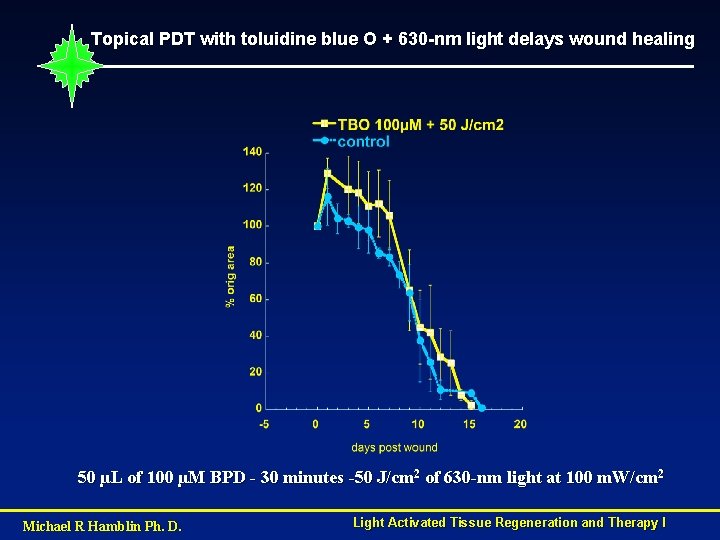
Topical PDT with toluidine blue O + 630 -nm light delays wound healing 50 µL of 100 µM BPD - 30 minutes -50 J/cm 2 of 630 -nm light at 100 m. W/cm 2 Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
Low dose 630 -nm light improves wound healing Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

High dose (50 J/cm 2) 630 -nm light has no effect Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
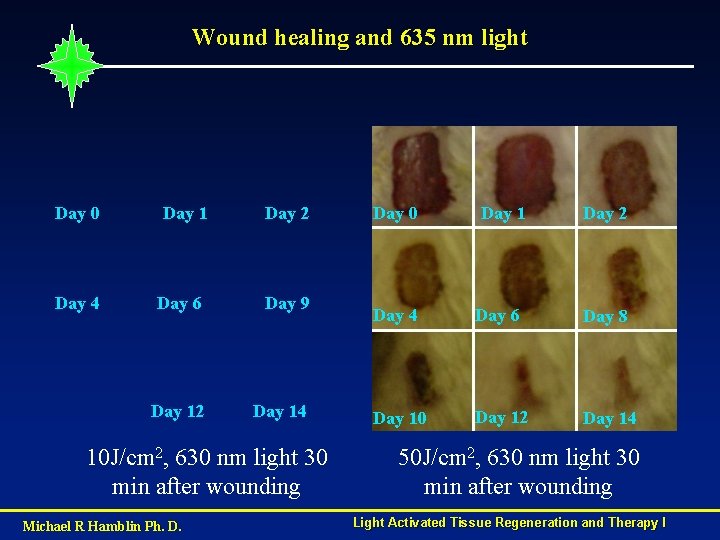
Wound healing and 635 nm light Day 0 Day 1 Day 2 Day 4 Day 6 Day 9 Day 4 Day 6 Day 8 Day 12 Day 14 10 J/cm 2, 630 nm light 30 min after wounding Michael R Hamblin Ph. D. Day 10 50 J/cm 2, 630 nm light 30 min after wounding Light Activated Tissue Regeneration and Therapy I

Conclusions Stimulation of wound healing is complicated Light alone may have an effect Ø Ø Ø Ø PS identity PS dose PS delivery route time between PS and light wavelength fluence rate Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I

Acknowledgements Faten Gad, MD Touqir Zahra, MD David O’Donnell, BSc Tayyaba Hasan, Ph. D Tatiana N Demidova, BSc Christopher H Contag - Stanford University Kevin P Francis - Xenogen Corp Funding: NIH-NIAID R 01 AI 050875 to M. R. Hamblin). Do. D-MFEL program Michael R Hamblin Ph. D. Light Activated Tissue Regeneration and Therapy I
- Slides: 49