Photocopies l Occasionally need uncontrolled copies induction information








































- Slides: 41

Photocopies l Occasionally need uncontrolled copies Ø induction? Ø information for client? l Mark copy as uncontrolled l Explain how in documentation

Handwritten Amendments l May be permitted l Explain how in documentation l Must be authorised l All copies must be amended l Minimise

Control of Records l Documented procedures, records of: Ø testing Ø equipment Ø internal calibration audits Ø management Ø corrective, review preventive actions

Control of Records l What was done l Who did it l Immediate recording l Preservation l Alterations - no erasure

Internal Audits

Outline l What is an internal audit ? l Types of Audits l How to meet standard requirements l Effective auditing l Preparing for an audit l Conducting an audit

Internal Audits Required: l By ISO 9000 Ø clause l 4. 17 By ISO/IEC 17025 Ø clause 4. 13 (management requirements)

What is an Internal Audit ? l Systematic and independent examination of the quality management system (QMS) l By someone within the organisation l In addition to external audits

The Internal Audit Process l Scheduled Audits Ø programme Ø examine Ø identify managed by Quality Manager documents, results, processes problems Ø improve l Unscheduled Audits Ø investigate Ø improve problem

Why Internal Auditing ? l Is QMS implemented exactly as intended ? l To investigate a problem Ø why did it occur ? Ø how can it be resolved ? Ø how can it be prevented in future ? l Identify opportunities to improve l Does the QMS meet requirements of standards ?

Types of Internal Audits l Horizontal Ø all departments audited against one element of standard or procedure l Vertical Ø one Ø department audited against all element of standard or procedure

Standard Requirements: ISO 9001 and 17025 l Audit program l Documented procedures l Auditors independent of activity l Audit results documented and reported to management l Prompt action after problems identified l Follow up activities

Management’s Responsibility l Define internal auditing policy l Assign responsibility of internal audit program Ø Quality l Manager Must be advised of internal audit outcomes Ø discussed at management review

Quality Manager’s Responsibility l Establish & maintain internal audit system l Develop schedule l Coordinate audits l Manage corrective action system l Advise management audit outcomes

Who Audits ? l Trained & qualified auditors l Quality Manager selects and trains internal auditors Ø observer on Quality Manager’s audits Ø fist audit under supervision of qualified auditor l Independent of the activity to be audited

Audit Schedule l Annual l Address all elements of the quality system Ø not l all departments Frequency ? Ø critical areas

Documents used in Internal Auditing l Checklists l Corrective action request forms l Audit report forms

Documenting the Audit Program (1) l Quality Manual Ø quality policy on internal auditing Ø responsibility for internal audits

Documenting the Audit Program (2) l Internal audit procedure(s) Ø selection and training of auditors Ø scheduling audits Ø responsibilities Ø preparation, of auditors conducting and reporting on audits Ø identifying, resolving and following up corrective actions Ø reporting audit results to management

Effective Auditing (1) l Gather evidence about compliance with quality system or standard

Effective Auditing (2) l Gather information about: Ø process, Ø staff, operating procedures equipment, test methods Ø environment, Ø quality handling of samples control, verification activities Ø recording and reporting practices. l Compare with documented system l Identify breakdown in system or departure from procedures

What to Audit l Systems audit Ø adherence l to documented procedures Technical audit Ø Technical correctness Ø adherence to documented procedures/test methods Ø auditor must have technical knowledge of test l Combination Ø vertical audit

What to Audit - Technical Audit l Staff l Methods l Equipment l Testing Environment l Samples and Test Items l Quality Control l Computers l Records and Reports

Audit Preparation l Quality Manager determines Ø audit team ð lead Ø auditor details ð scope ð time, l of audit date, duration Contact auditee Ø date, time, type & duration

1. Audit Plan 3. Opening Meeting g tin uc nd Co 2. Develop Checklists 5. Record Results t di Au e th 4. Gather Evidence 6. Closing Meeting 7. Audit Report

1. Audit Plan l Objectives & scope l Collect documents Ø standard, procedure, work instructions, forms Ø desk l top review History

2. Developing Checklists l Guidelines l Review documents Ø identify important aspects of the activity Ø list in logical order Ø set of questions

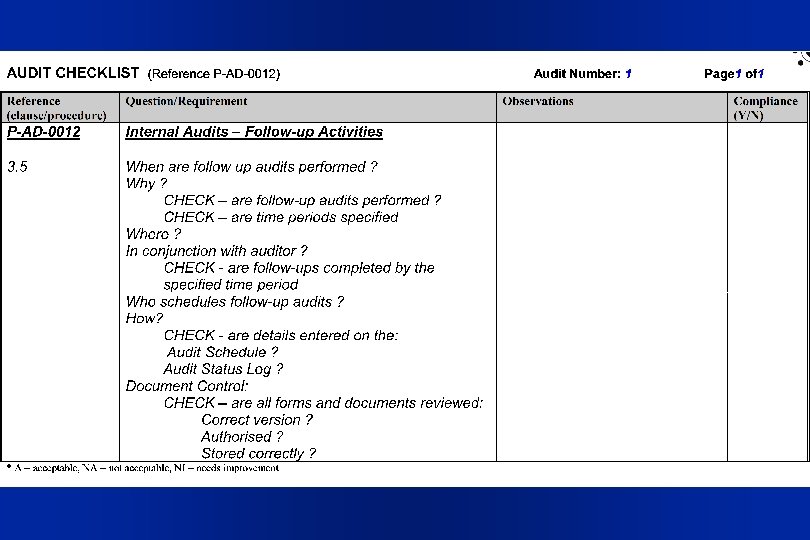
P-AD-0012 3. 5 Audit Follow-up Activities l It may be necessary for a follow-up audit to be performed to verify the effectiveness of any corrective action carried out. Corrective action, and subsequent follow-up audits, should be completed within a time period agreed to by the auditee, in consultation with the auditor. l The Quality Manager should schedule the follow-up audit and enter details on the Audit Schedule and the Audit Status Log.


3. Opening Meeting l l Who ? Ø auditor/audit team Ø auditee Ø any staff from area to be audited that may be interviewed What ? Ø Scope Ø expected duration

4. 1 Gather Evidence about Compliance l Interviews Ø ask questions about system and its implementation Ø who, what, when, how, where, why ? Ø other questions ð direct ð hypothetical ð clarifying

4. 2 Gather Evidence about Compliance l Examine documents Ø procedures, work instructions, forms, quality manual Ø copies controlled ? Ø available ? Ø correct issue status ? Ø used in manner intended ? l Quality Records Ø stored correctly ? Ø used as objective evidence Ø many forms

4. 3 Gather Evidence about Compliance l Observe activities Ø what is said or written may not reflect practice Ø “show me” l Examine facilities Ø as travel through laboratory/offices Ø examine: ð equipment ð standard of housekeeping ð size and layout of working area ð environment eg. temperature in lab

5. Recording Results l Record on checklists Ø activities which do not adhere to quality system Ø may be classified ð major non-conformance ð minor non-conformance Ø areas for improvement

6. Closing Meeting(s) l Audit team meeting Ø discuss l audit results Closing meeting Ø discuss corrective actions Ø determine l resolution dates Identify corrective actions Ø use corrective action forms

7. Audit Report Audit details l Summary of findings l Ø corrective actions ð numbered ð objective evidence ð reference the document Ø observations l Distribute

Corrective and Preventive Action

Outline l What is a corrective action ? l What is a preventive action ? l Corrective and preventive action program l Corrective and preventive action process

Corrective & Preventive Action Required: l By ISO 9000 Ø clause l 4. 14 By ISO/IEC 17025 Ø clause 4. 10 corrective action Ø clause 4. 11 preventive action

Corrective Action l An action taken to correct a problem Ø incorrect result Ø departure from procedure

Preventive Action l A proactive process to identify Ø improvement Ø potential opportunities sources of non-conformance