Photochemistry can be dramatically different from ordinary chemistry

Photochemistry can be dramatically different from “ordinary” chemistry See example below

Cl Ar Cl
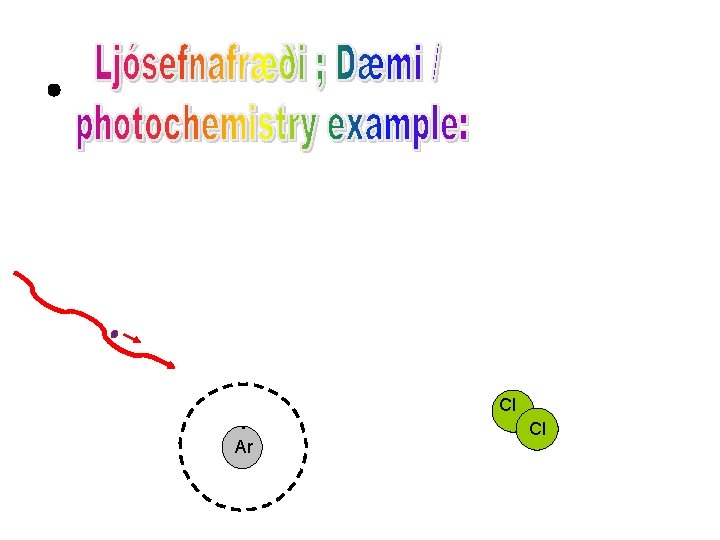
. Ar Cl Cl

Cl Ar Cl
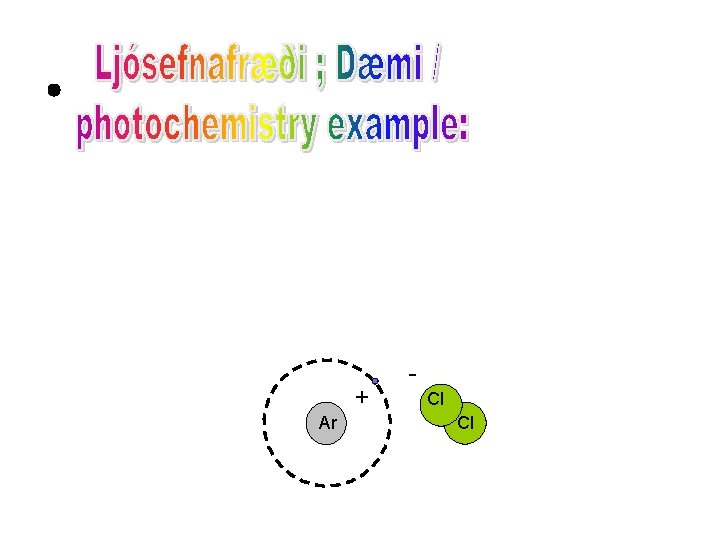
+ Ar Cl Cl
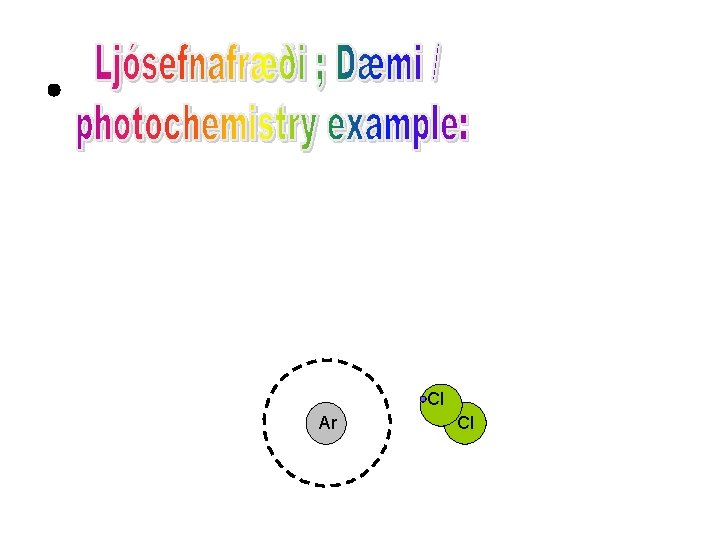
Cl Ar Cl

Hvernig er efnafræði ósoneyðingarinnar? / Ozon depletion? O 3 Cl
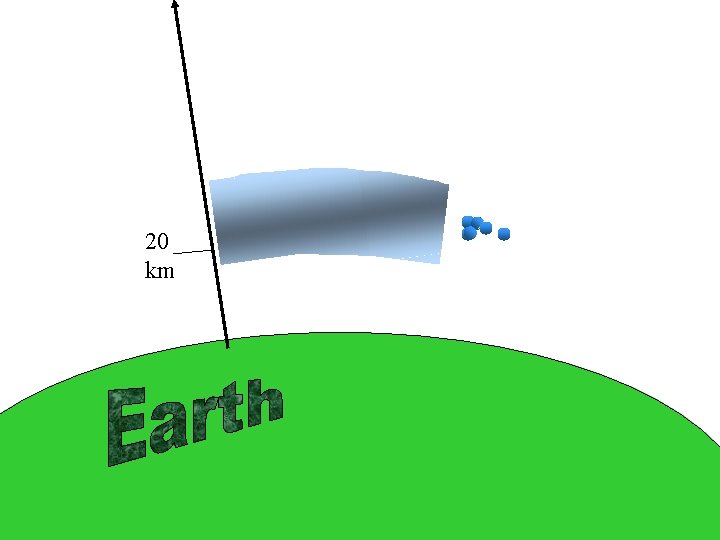
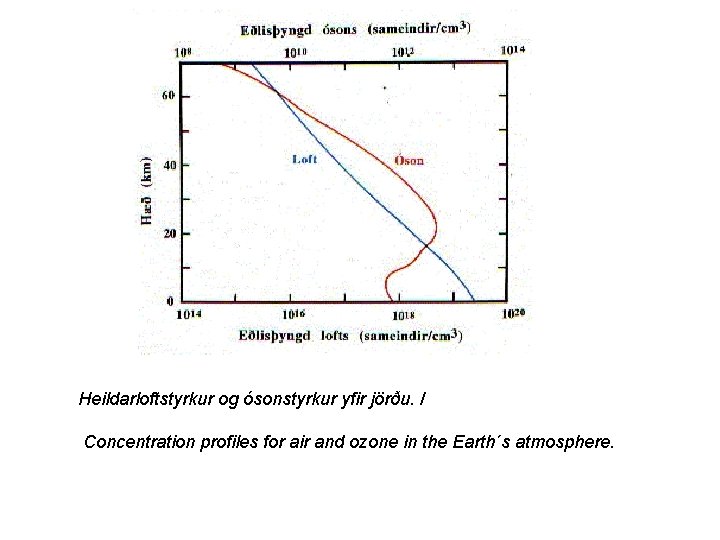
Heildarloftstyrkur og ósonstyrkur yfir jörðu. / Concentration profiles for air and ozone in the Earth´s atmosphere.
20 km
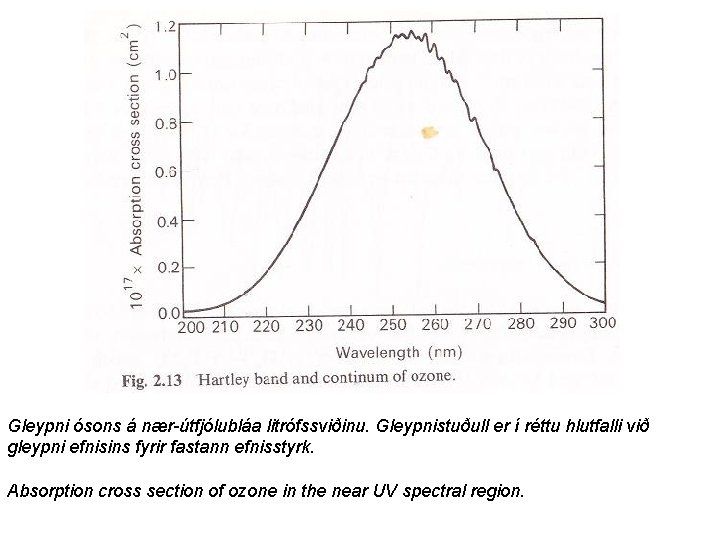
Gleypni ósons á nær-útfjólubláa litrófssviðinu. Gleypnistuðull er í réttu hlutfalli við gleypni efnisins fyrir fastann efnisstyrk. Absorption cross section of ozone in the near UV spectral region.
20 km
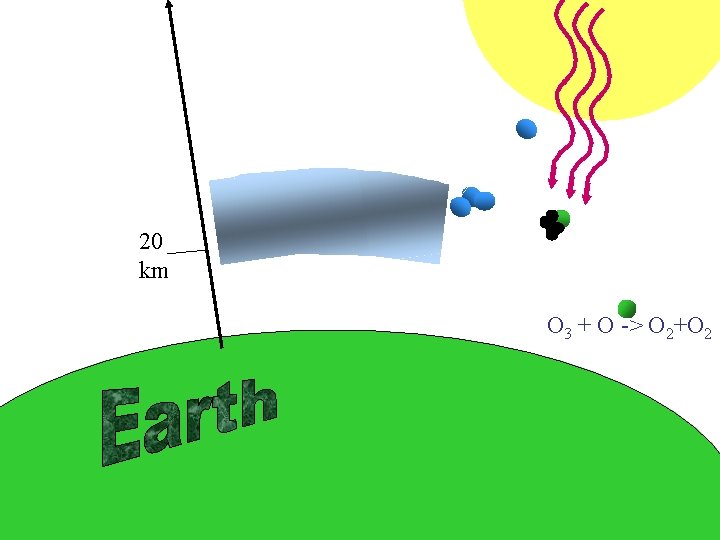
20 km O 3 + O -> O 2+O 2

Chemistry and spectroscopy of the greenhouse effect/ Global warming effects(? )
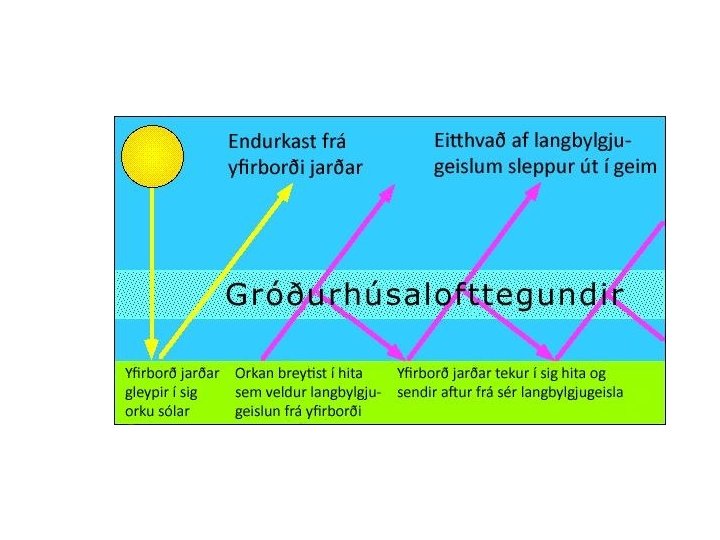
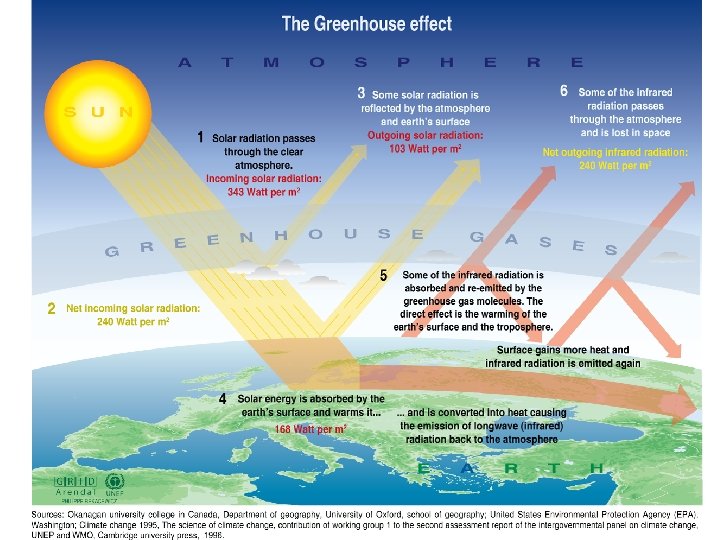
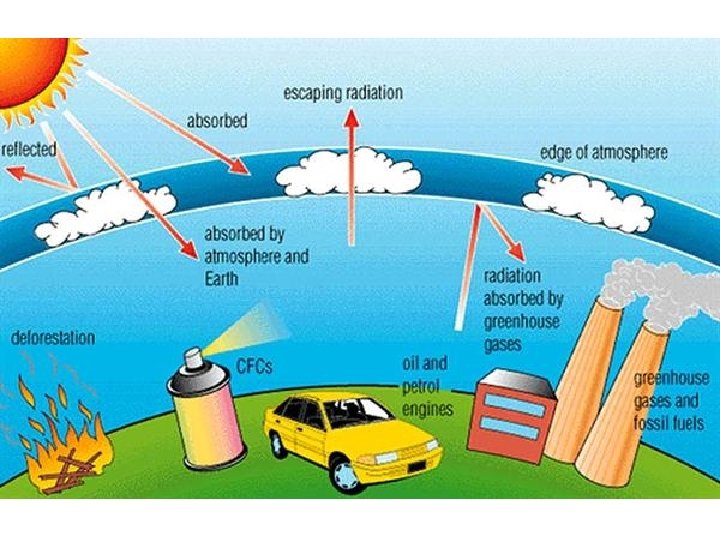
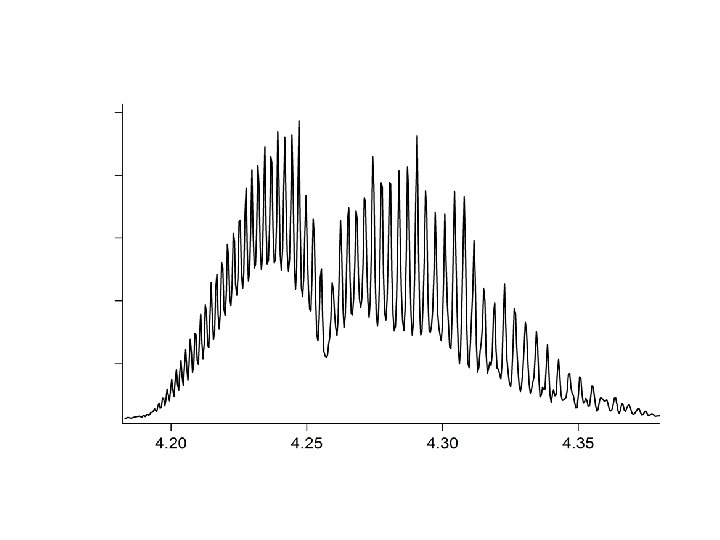
Gegnskin og gleypni andrúmsloftsins ÚF SÝN . . IR. . . .
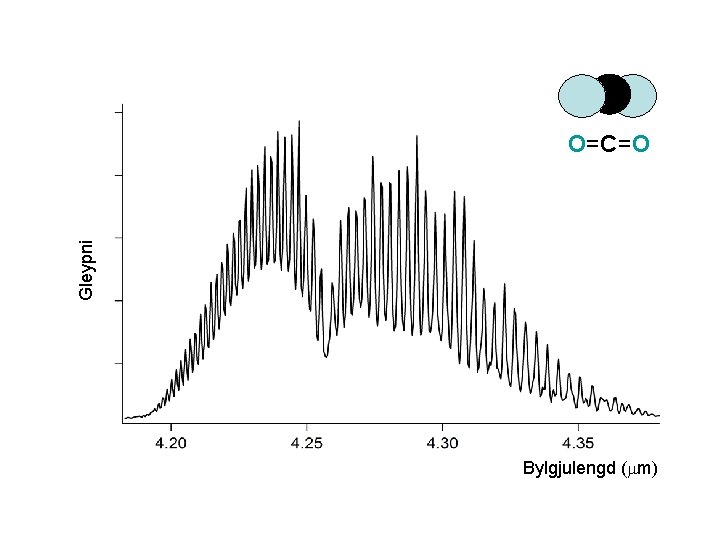
Gleypni O=C=O Bylgjulengd (mm)
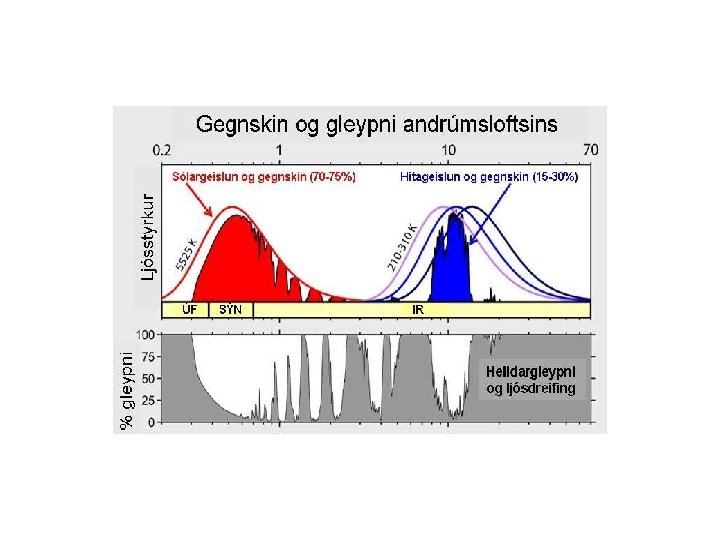
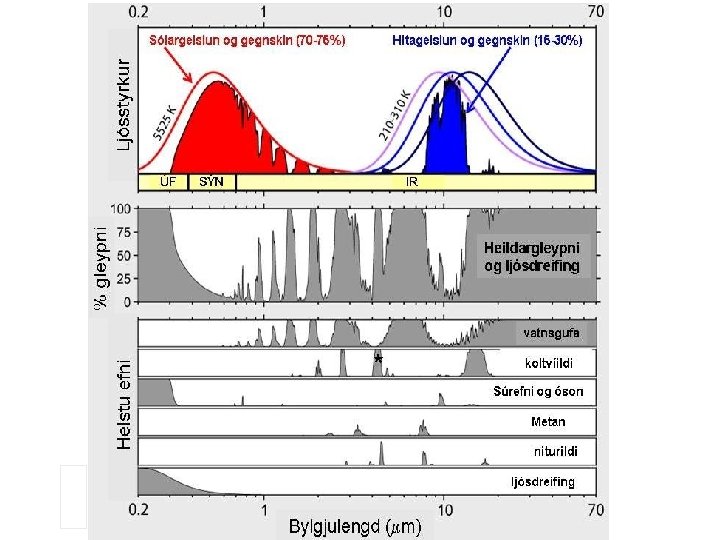
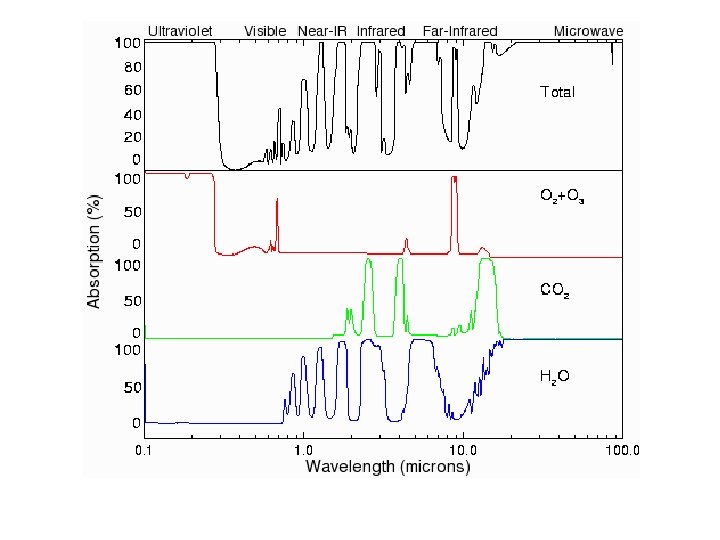
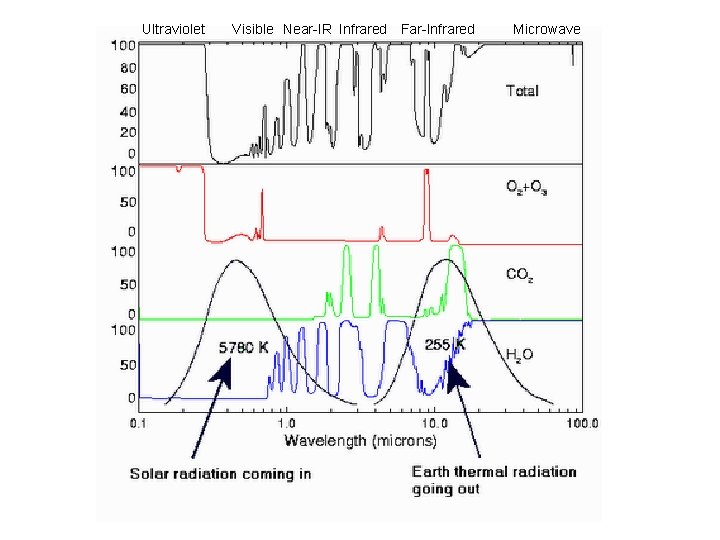
Ultraviolet Visible Near-IR Infrared Far-Infrared Microwave

Organic photochemistry: photoisomerization
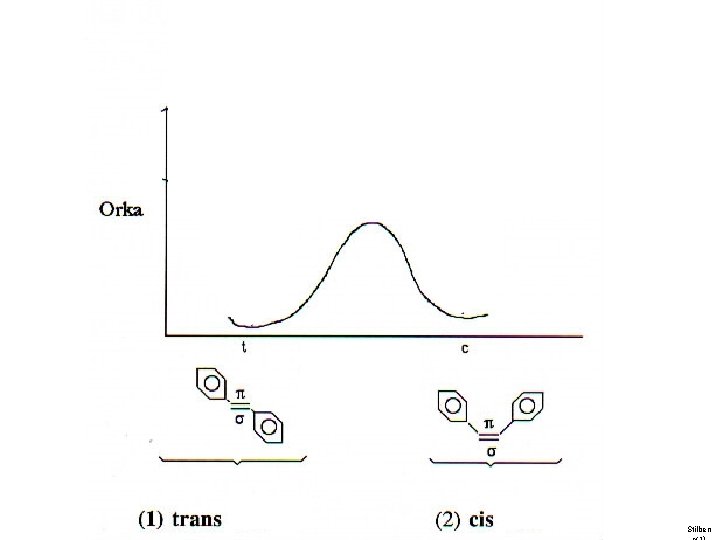
Stilben
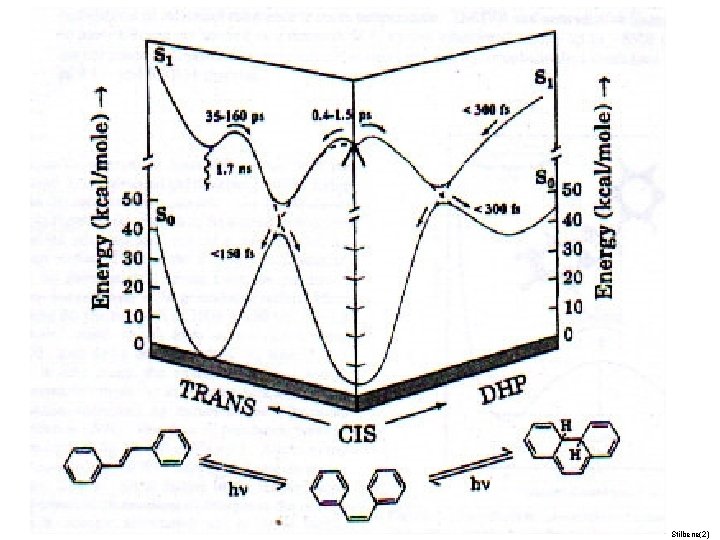
Stilbene(2)

Astrochemistry
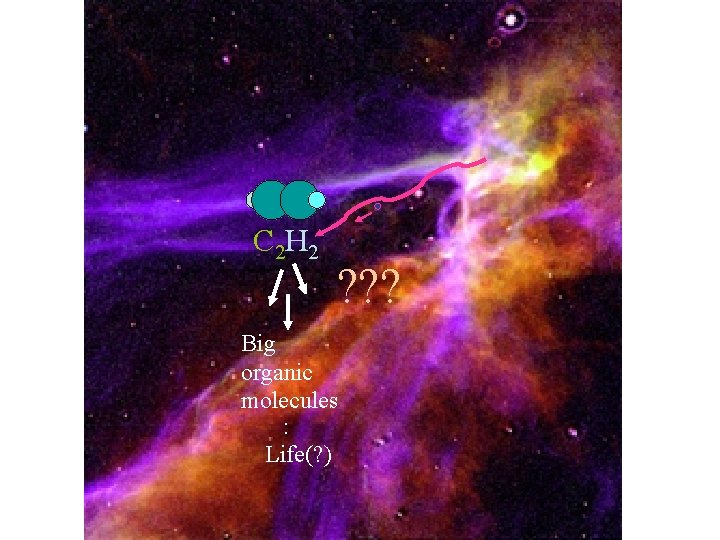
C 2 H 2 ? ? ? Big organic molecules : Life(? )
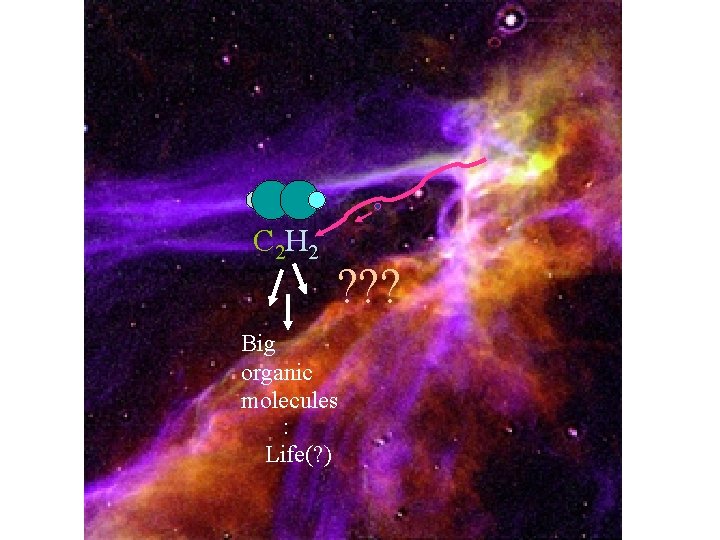
C 2 H 2 ? ? ? Big organic molecules : Life(? )
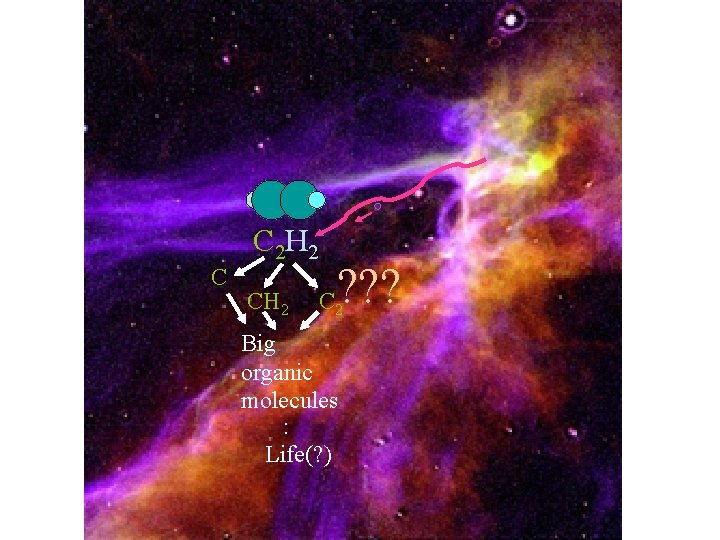
C 2 H 2 C CH 2 ? ? ? C 2 Big organic molecules : Life(? )
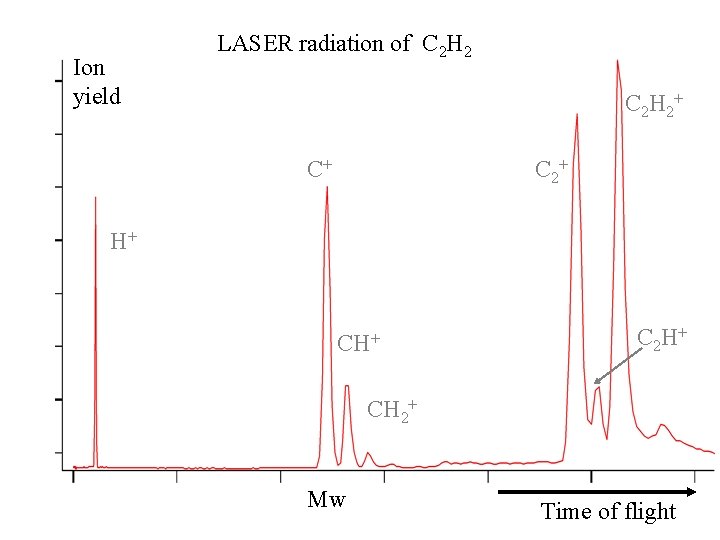
Ion yield LASER radiation of C 2 H 2+ Magn jóna C+ C 2+ H+ C 2 H + CH 2+ Mw Time of flight
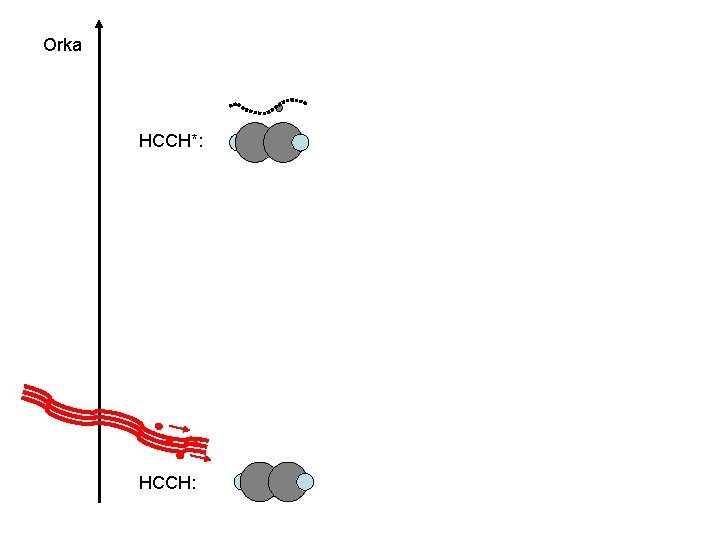
Orka HCCH*: HCCH:
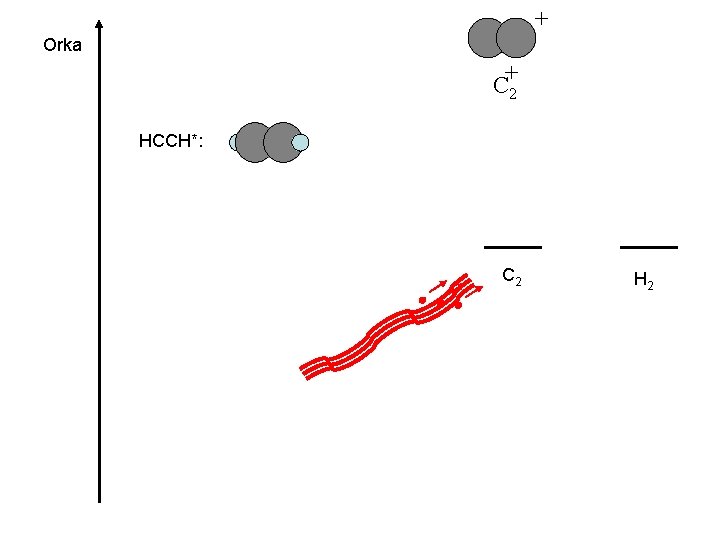
+ Orka C+2 HCCH*: C 2 H 2
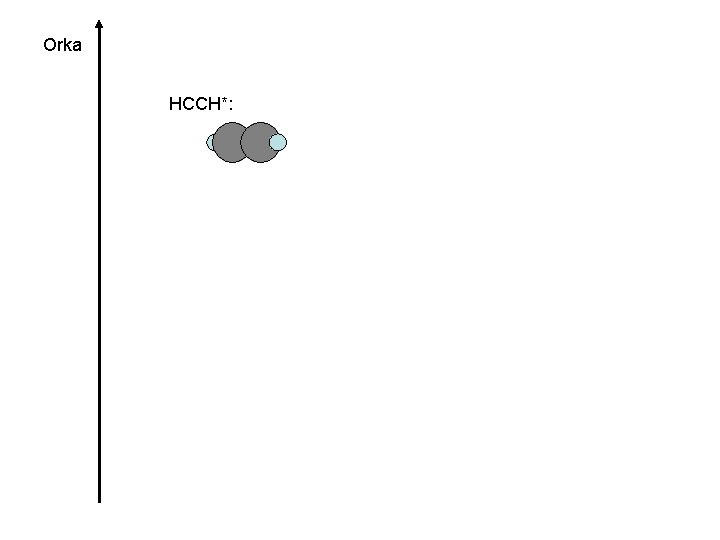
Orka HCCH*:
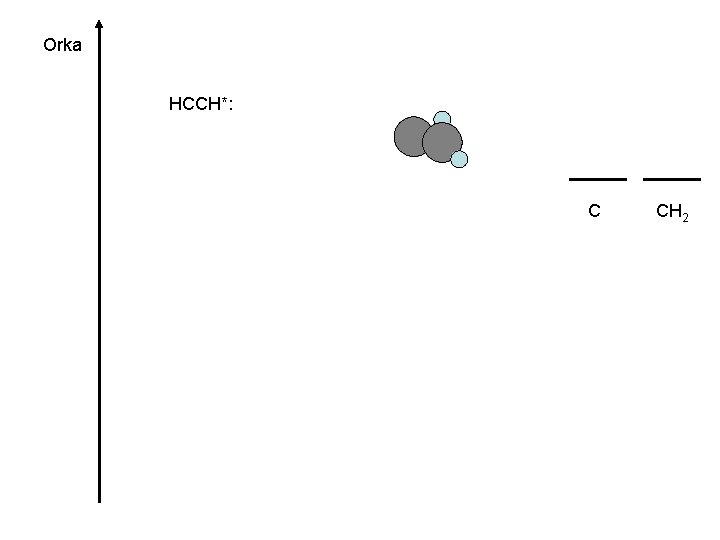
Orka HCCH*: C CH 2
- Slides: 35