PHOTOCHEMISTRY BY Dr Depinder Kaur Associate Prof in

PHOTOCHEMISTRY BY Dr. Depinder Kaur Associate Prof. in Chemistry

INTRODUCTION • Photochemistry is defined as that branch of chemistry which deals with the study of interaction of radiation with matter resulting into a physical change or into a chemical reaction (called photochemical reaction) whose rates and mechanisms are studied after initiation by the radiant energy. The radiation of photochemical importance are those which lie in the visible and ultraviolet region from 8000 - 2000 Å (800 - 200 nm). • Thus the two main processes studied under photochemistry are : • (i) Photophysical processes • (ii) Photochemical processes or photochemical reactions.

Photophysical processes • Photophysical processes are those processes which take place in the presence of light but do not result into any chemical reaction. . If the absorbed light is emitted instanteniously, the process is called fluorescence. If the absorbed light is emitted after some time lag, the process is called phosphorescence. . Thus photophysical processes include phenomena like fluorescence , phosphresence and photoelectric effect.

• Photochemical reactions are those reactions which take place by absorption of light by the reacting substances. These reactions are generally brought about by the absorption of light radiations of the visible and ultraviolet region which lie between 8000 - 2000 Å (800 -200 nm) reaction. A large number of different types of reactions can be brought about by exposure to suitable light e. g. synthesis, decomposition, oxidation, reduction, polymerisation, isomeric change etc.

• Two well known examples of photochemical reactions are given below : • (i) Combination of hydrogen and chlorine to form hydrogen chloride • H 2(g) + cl 2(g) Light→ 2 HCl(g) • (ii) Photosynthesis of carbohydrates plants • 6 C 02 + 6 H 20 → C 6 H 1206 + 602 • From air Glucose
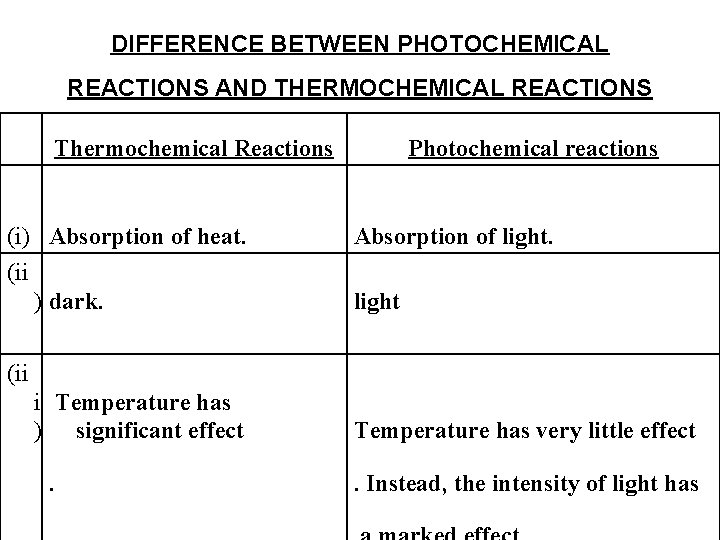
DIFFERENCE BETWEEN PHOTOCHEMICAL REACTIONS AND THERMOCHEMICAL REACTIONS Thermochemical Reactions (i) Absorption of heat. (ii ) dark. Photochemical reactions Absorption of light (ii i Temperature has ) significant effect Temperature has very little effect . Instead, the intensity of light has .

iv) (ΔG) (Δ G) is always negative. may not be negative. examples of reactions with ^G positive and still spontaneous (i)Synthesis of carbohydrates in plants. (ii)Ozonization of oxygen. iv) Activation energy arises (iii)Decomposition of HCl into H 2 and Cl 2* Activation energy is acquired by absorption from the intermolecular collisions of photons of suitable energy

LAWS GOVERNING ABSORPTION OF LIGHT • When a monochromatic light (light consisting of a single wavelength) is passed through a medium some light is absorbed by the medium and hence the intensity of the incident light decreases. The decrease of intensity on passing through a pure homogeneous medium (i. e. a pure liquid) is governed by Lambert's law and the decrease of intensity on passing through a solution is governed by Beer's law.

Lambert's Law. • When a monochromatic light is passed through a pure homogeneous medium, the decrease in the intensity of light with thickness of the absorbing medium at any point X is proportional to the intensity of the incident light i. e. the intensity of light at the point X (i. e. just before entering into the dx). Mathematically, • -dl / dx α I or -dl / dx =k. I

• • • The constant of proportionality k is called the absorption coefficient depending upon the nature of the absorbing medium. The intensity I at any point X (at a distance x from the start of the medium), can be found in terms if the original intensity I 0 as follows : Equation (1) can be written as d. I/ I= -k dx . . . (2 When x = 0, I = I 0 x = x, I =I

Integrating equation (2) between the limits x = 0 to x and I= I 0 to I, we get This equation expresses how the original intensity I 0 is reduced to intensity I after passing through a thickness x of the medium*. The difference between I 0 and I obviously gives the intensity of the light absorbed.

• Thus • Iabs =I 0 - I = IO-I 0 e –kx • • Equation (3) can be written as 2. 303 log I/I 0 = -kx log I/I 0 = - kx/2. 303 log I/I 0 = - k ‘x I/I 0 = 10 - k ‘x (6) I = I 0 x 10 - k ‘x Where k’= k/2. 303 is called extinction coefficient of the substance ( i. e. of the absorbing medium). Now it is this quantity which is called absorption coefficient or absorptivity of the substance.

• The physical significance of the extinction coefficient or absorptivity follows more clearly from equation (6) which can be rewritten as • K’ = -1/x log I/I 0 = 1/x log I 0 /I • If log I 0 /I = 1 , K’= 1/x • But log I 0 /I = 1 means I 0 /I = 10 • i. e. I = I O/10 • Hence extinction coefficient or absorptivity may be defined as follows : • The extinction coefficient or absorptivity is the reciprocal of that layer thickness (expressed in cm) at which the intensity of the light falls to onetenth of its original value.

Beer's Law • ). It is applicable to solutions and states as : • When a monochromic light is passed through a solution, the decrease in the intensity of light with the thickness of the solution is directly proportional not only to the intensity of the incident light but also to the concentration 'c' of the solution. Thus for a solution, mathematically, we have • -dl/dx α I x c • -dl/dx = εIc (ε is pronounced as epsilon) (8) • where ε is a constant of proportionality and is called molar absorption coefficient. Its value depends upon the nature of the absorbing solute and the

• Equation (8) can be rewritten as • d. I/I = - εc dx • As before, integrating this equation between the limits x = 0 to x and I= I 0 to I , we get • ln I/IO = - εcx • Or I/I 0 =e - εcx • Or I =I 0 e - εcx • This equation shows that the intensity of a monochromatic light decreases exponentially from I 0 to I with increase in thickness x and concentration c.

• The intensity of the light absorbed by the solution when it passes through a length x of the solution will be given by • Iabs = I 0 - I 0 e- εcx = I 0(I- e- εcx). . (12) • • • 2. 303 log I/ I 0 = - εcx or log. I/I 0=- εcx/2. 303 or log. I/I 0=- έcx _(13) or I/I 0= 10 - έcx or I = I 0 x 10 - έcx where έ = ε/ 2. 303 was earlier called molar extinction coefficient of the absorbing solution. Now it is this quantity which is called molar absorption coefficient or molar absorptivity of the absorbing solution.

• The physical significance of molar extinction coefficient or molar absorptivity, follows from equation (13), which can be written as • έ = -1/cx log I/I 0 = 1/cx log I 0/I • If c=1 M and log I 0/I = 1 then έ = 1/x • As before, log I 0/I = 1 means I 0/I = 10 i. e. I = I 0/10 • Hence molar extinction coefficient or molar absorptivity may be defined as the reciprocal of that thickness of the solution layer of 1 molar concentration which reduces the intensity of the light passing through it to one-tenth of its original value. • It may be pointed out that gases follow Beer's law and not Lambert's law.

• Units of molar absorptivity or absorption coefficient • έ = - 1 /cx log I/I 0 • log I/I 0 is a dimensionless quantity. • In SI units, x is in metres (m) and c is in mol m -3. Hence, we have • έ=(m 3 mol -1)(m -1)=m 2 mol -1

LAWS GOVERNING PHOTOCHEMICAL REACTIONS • There are two important laws of photochemistry : • 1. Grotthus-Draper Law. This law is also called First law of photochemistry. When light falls on a body, a part of it is reflected, a part of it is transmitted and the rest of it is absorbed. It is only the absorbed light which is effective in bringing about a chemical reaction.

• The absorbed light must not always result into chemical reaction. It may simply bring about fluorescence, phosphorescence etc. , it may be simply converted into thermal energy e. g. in case of potassium permanganate solution, . Further in some cases, it is observed that light energy may not be absorbed by the reacting substance directly as in photo-sensitization'. Example is photosynthesis in plants where chlorophyll acts as a photosensitizer.

• 2. Stark-Einstein's Law of Photochemical Equivalence. • This law is also called second law of photochemistry. Every atom or molecule that takes part in a photochemical reaction absorbs one quantum of the radiation to which the substance is exposed. • If v is the frequency of the absorbed radiation , then energy absorbed bv one mole of the reacting molecules will then be given by • E = Nhv • (where N is Avogadro's number) • Putting v =c/λ We can write E = Nh c/λ where c is the velocity of light and λ is the wavelength of the absorbed radiation

• The energy possessed by one mole of photons (or the energy absorbed by one mole of the reacting molecules) is called one einstein. • In CGS units • E = 2. 86 x 105 / λ (Å) kcal per mole • In SI units • E= 11. 97 x 10 -5 / λ(m) k. J/mol • Thus the energy per Einstein is inversely proportional to the wavelength of the radiation. Shorter the wavelength of a radiation , greater is the energy per Einstein. For example the order of wavelength of different radiations is • X rays < Ultraviolet < Visible < Infrared • Hence the energies per Einstein of these radiations are in the order • X rays > Ultraviolet >Visible > Infrared

INTERPRETATION OF EINSTEIN'S LAW -'QUANTUM EFFICIENCY' • If Einstein's law is correct, every reacting molecule will absorb one quantum of radiation. . However, it is found that in a number of cases, a small amount of the light absorbed can bring about a large amount of reaction, whereas in some other cases, large amount of the light absorbed can bring about only a small amount of reaction. This was explained on the basis that there are two steps. The first stage (involving the absorption of light) is called the primary process. The reactions involved in the second stage are called secondary processes.

• The number of quanta absorbed with the number of reacting molecules, a term called quantum efficiency or quantum yield (φ) has been introduced. • φ = Number of molecules reacting in a given time • Number of quanta of light absorbed in the same time • = Number of moles reacting in a given time • Number of einsteins of light absorbed in the same time. • Hence quantum efficiency may be defined as the number of moles reacting per einstein. light absorbed. • If Stark-Einstein's law is strictly obeyed in the form already stated, φ should be equal to unity. However, studies have shown that the law is applicable only to the primary processes. Hence Each molecule that gets activated to initiate the reaction absorbs one

EXPERIMENTAL DETERMINATION OF QUANTUM YIELD OF A PHOTOCHEMICAL REACTION • • • φ = Number of moles reacting per sec Number of quanta absorbed per sec. = Rate of chemical reaction Quanta absorbed per sec Thus to determine the quantum yield, we have to measure (i) the rate of chemical reaction • (ii) the quanta absorbed per sec.

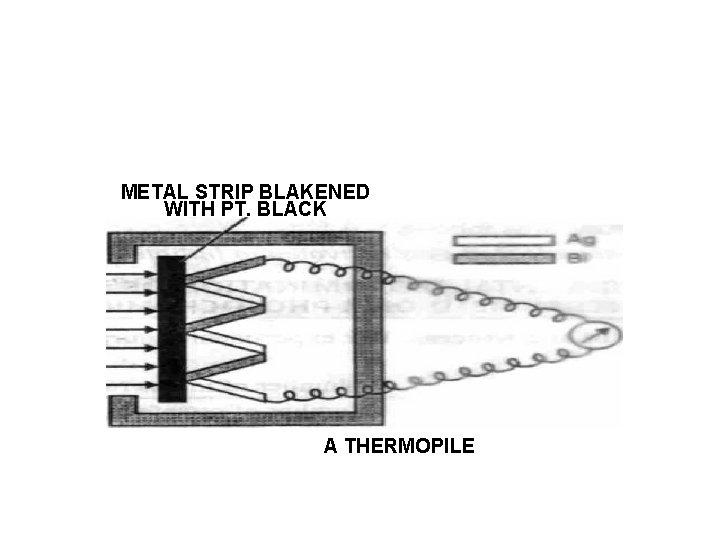
• • Apparatus. : (i) A source of radiation (ii) Lens of suitable focal length. (iii) Monochromator or filter (iv) Slit. (v) A cell (vi) Recorder. It is usually a thermopile or an actinometer which is used to measure the intensity of radiation.

• A thermopile is a set of thermocouples. Rods of two different metals (e. g. Ag and Bi) are joined alternately. (If only two rods are connected, it constitutes a thermocouple). Radiations falling on this are almost completely absorbed by it. As a result, this set of junctions becomes hot. The temperature difference of the hot end and the cold end produces a current which can be measured by connecting a milliammeter to thermopile. By calibrating thermopile with radiations of known intensity, the intensity of the desired radiations can be measured.
METAL STRIP BLAKENED WITH PT. BLACK A THERMOPILE

• A chemical actinometer is a device in which gas mixture or solutions sensitive to light are used The working of this device is based upon the fact that a definite amount of the radiation absorbed brings about a definite amount of chemical reaction. uranyl oxalate actinometer which consists of 0. 05 M oxalic acid and 0. 01 M uranyl sulphate (U 02 S 04) solution in water. : • Uranyl sulphate acts as photosensitizer. The extent of reaction is measured by titrating oxalic acid solution with potassium permanganate solution. The actinometer is first calibrated against a thermopile. This actinometer can be used for measuring only the intensity of the radiation lying in the range 2000 - 5000 Å.

Procedure • (i)Measurement of the intensity of light absorbed. First, the empty cell or the cell filled with solvent and is placed in thermostat. . The reading is taken on the recorder. From this we get the energy of the incident radiation. Now the reactants are taken in the cell , the radiations are allowed to pass through it for definite time and the reading is taken again. The difference between the two readings gives the total energy absorbed by the reactants in a given time. From this the intensity of the radiation absorbed Iabs is calculated : • Iabs = Total energy absorbed • Volume of the reaction mixture x Time in sec

• (ii) Measurement of the rate of reaction. The rate of the reaction can be studied as usual. For this purpose, either the change in some physical property is followed or samples of the reaction mixture are removed periodically from the cell and analysed. • Knowing the rate of reaction and the intensity of light absorbed, the quantum yield of the reaction can be calculated.

PHOTOCHEMICAL REACTIONS • A photochemical reaction takes place in two steps • 1. Primary process : Activation of some molecules by absorption of light. • AB + hv → AB* • Or AB + hv → A + B* • 2. Secondary process : Reaction of activated molecules of primary process with other molecules resulting into activation or deactivation •

• On the basis of above processes photochemical reactions are of three types • 1. Quantum yield is a small integer. • H 2 + Br 2 → 2 HBr φ=1 • 2. Quantum yield is very large. • H 2 + Cl 2 → 2 HCl φ=10 6 • 3. Quantum yield is very small. • NH 3 + hv → N 2 + H 2 φ=0. 25

Photosynthesis of HBr i. e. Photochemical combination of Hydrogen and Bromine to form HBr. • The reaction may be represented as • H 2 + Br 2 → 2 HBr • The quantum efficiency of this reaction is about 0. 01. This is explained by following mechanism )Primary process ; • Br 2 + hv → 2 Br • (b)Secondary Process : • (i) Br + H 2 endo→ HBr + H • (ii) H + Br 2 → HBr + Br • (iii)H + HBr → H 2 + Br • (iv) Br + Br → Br 2

Photosynthesis of HCl from H 2 and Cl 2 • Its quantum yeild is very high i. e. 10 4 to 10 6 it may be represented as, • H 2 + Cl 2 → 2 HCl • The different steps involved are as follows : • (a) Primary process : • Cl 2 + hv → 2 Cl • (b) Secondary process : • (i) CI + H 2 exo--→ HCl + H • (ii) H + Cl 2 → HCl+ CI • (iii) CI + CI → Cl 2

• The mechanism of the above reaction is similar to HBr, yet the quantum yield of this reaction is very high whereas that of HBr is very low. The difference is explained on the basis of the reaction (i) of the secondary processes. In the present reaction, the reaction (i) of the secondary processes is exothermic and therefore, takes place very easily and consequently the chain reaction is set up very easily. In the photosynthesis of HBr, the reaction (i) of the secondary process is endothermic and thus has very little tendency to take place.

• The quantum yield of the above reaction decreases if the reaction vessel contains small traces of oxygen. • H + 02 → H 02 • CI + 02 + HCl → H 02 + Cl 2 (i) The reaction can be brought-about without exposing the reaction mixture to light but by simply adding some CI or H atoms into the reaction mixture. • (ii) The quantum yield of the reaction decreases if the reaction is carried out in capillary tubes.

Photolysis of Ammonia. • The Q Y is 0. 25. The probable mechanism is : • (a) Primary process: • NH 3 + hv → NH 2 + H • (b) Secondary processes. ;

• (i) NH 2 + NH 2 → N 2+ 2 H 2 • • (ii) NH 2 + NH 2 → N 2 H 4 (iii) H + H → H 2 (iv) NH 2 + H → NH 3 Since the quantum yield of the reaction is very low, the reaction (i) to (iii) take place only to a very small extent. The important secondary reaction is the reaction (iv) only which involves the reversal of the primary process.

PHOTO- PHYSICAL PROCESSES • Fluorescence. There are certain substances which when exposed to light or certain other radiations absorb the energy and then immediately or instantaneously start re-emitting the energy. Such substances are called fluorescent substances and the phenomenon is called fluorescence. The word 'instantaneous' used above implies that the time between the absorption of this energy and the re-emission of the absorbed energy is not more 10 -8 sec. Thus fluorescence starts as soon as the substance is exposed to light and the fluorescence stops as soon as the light is cut off.

• Examples are : • (i) fluorite, Ca. F 2 (from which the name of the phenomenon is derived) • ( ii) certain organic dyes such as eosin, fluorescein etc • (iii) vapour of sodium, mercury, iodine etc. • (iv) certain inorganic compounds such as uranyl sulphate, • In fluorescence, the wavelength of emitted light is usually greater than that of the absorbed light. This is because the energy absorbed raises the electron to a sufficiently higher level but the return of the excited electron to the original level takes place in steps, through the

• The energy thus produced in every jump is smaller and hence the wavelength of the light emitted is large (as E = hv = hc/λ i. e. E α 1/λ) than that of the absorbed light. • In case of mercury vapour, the emitted light has the same wavelength as that of the absorbed light. This phenomenon is called resonance fluorescence. In certain cases, light of shorter wavelength (i. e. of higher energy) is emitted. This is explained by suggesting that the emitted light contains the absorbed energy as well as some energy present in the system before absorption.

• Phosphorescence. In case of fluorescent materials, the florescence stops as soon as the external light is cut off. However, there are certain substances which continue to glow for some time even after the external light is cut off. Such substances are called phosphors or phosphoresce substances and the phenomenon is called phosphorescence. Thus in a way, phosphorescence is a slow fluorescence. It is found in solid, the molecules have least freedom of motion. .

• The best examples of the phosphorescence include zinc sulphide and sulphide of the alkaline earth metals. fluorescent substances becomes phosphorescent if fixed by suitable methods, e. g. by fusion with other substances etc. For example, many dyes (which show fluorescence) when dissolved in fused boric acid or glycerol and then cooled to get rigid mass become phosphorescent.

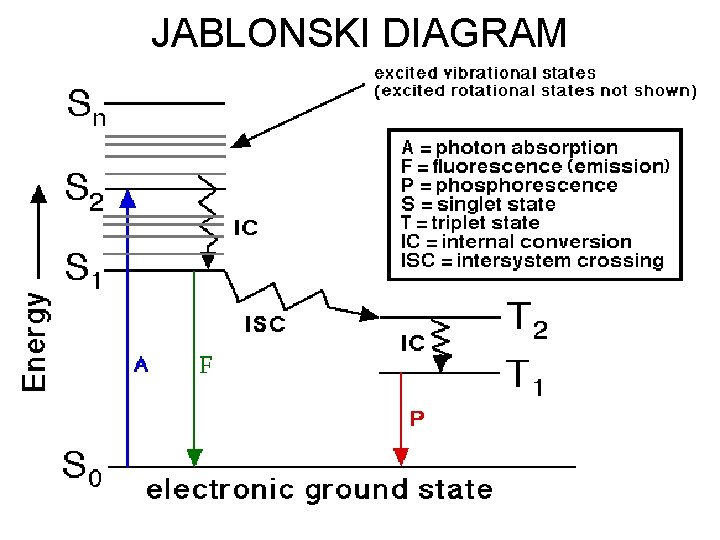
FLUORESCENCE AND PHOSPHORESCENCE IN TERMS OF EXCITATION OF ELECTRONS (JABLONSKI DIAGRAM)
JABLONSKI DIAGRAM

• Path I. The molecule may lose rest of the energy also in the form of heat so that the complete path is non-radiative. • Path II. The molecule may release the energy in the form of light or ultraviolet radiations in returning to the ground state from the first excited singlet state (shown by straight line in the diagram). Hence it is a radiative transition. This is called fluorescence. It occurs for about 10 -8 second after absorption so that a substance fluoresces only in the presence of absorbed radiation. •

• Path III. Some energy may first be lost in transition from first excited singlet state to the first excited triplet state i. e. between states of different multiplicity i. e. S 1 to T 1(or similarly. S 2 to T 2. S 3 to T 3 etc. , in the form of heat so that it is again a radiationless transition. It is also called intersystem crossing (ISC). Such transitions are normally forbidden but whenever they occur, their rate is very slow. In fur transition from first excited triplet state to the ground state, the rest of the energy may again be lost in the form of heat due to molecular collisions. Thus the complete path is non-radiative. • Further, the energy is lost not only as heat but also in undergoing a chemical reaction. As singlet excited states return quickly to the ground state, there is no chance for a chemical reaction However, if the molecules are in the triplet excited states, they return to the ground state slowly and thus get sufficient time to undergo a chemical reaction. Thus molecules undergoing chemical reaction are those which are first in the excited triplet state.

• Path IV. After intersystem crossing, the molecule may lose the rest of the energy- in the form of light in going from the excited triplet state to the ground state. This is called phosphorescence. As triplet states have much longer life times than singlet states, some as long as an hour, phosphorescence persists even after the removal of absorbed radiation. This may also be attributed to the fact that the transitions from excited triplet states to ground singlet state are forbidden. • Note that fluorescence involves a radiative transition between two states of the same multiplicity (usually two singlets) whereas phosphorescence arises due to a radiative transition between two states of different multiplicity (usually triplet to singlet). It may be observed that the energy emitted during fluorescence as well as phosphorescence is less than the energy absorbed during excitation, as a part of the energy is dissipated as heat in the internal conversion and intersystem crossing. Hence the frequencies emitted during fluorescence and phosphorescence are smaller or wavelengths are longer than those of the light absorbed for excitation. •

• The paths followed as explained above may be compared with a two-floor building having steps, ramps and windows. First step of all paths is like a ball falling from second floor to the first floor by bouncing on steps going from second floor to first floor. If the ball now falls on the ramp going from first floor to ground floor, it is like path I. If the ball after coming to first floor falls out of the window, it is like path II. If after coming to first floor, the ball moves on some steps and then falls in the ramp going from first floor to ground floor, it is like path III. If the ball after having come to first floor, moves on some steps going from first floor to ground floor and then jumps out of the window, it is like path IV.

THANKS
- Slides: 51