PHITT the new international trial for hepatoblastoma and

PHITT the new international trial for hepatoblastoma and HCC Professor Bruce Morland Birmingham, UK

2 linked European efforts

Where did it all start?


And then Los Angeles COG Spring meeting 2012

The LA papers!

And then Gdansk, Poland SIOPEL Spring meeting

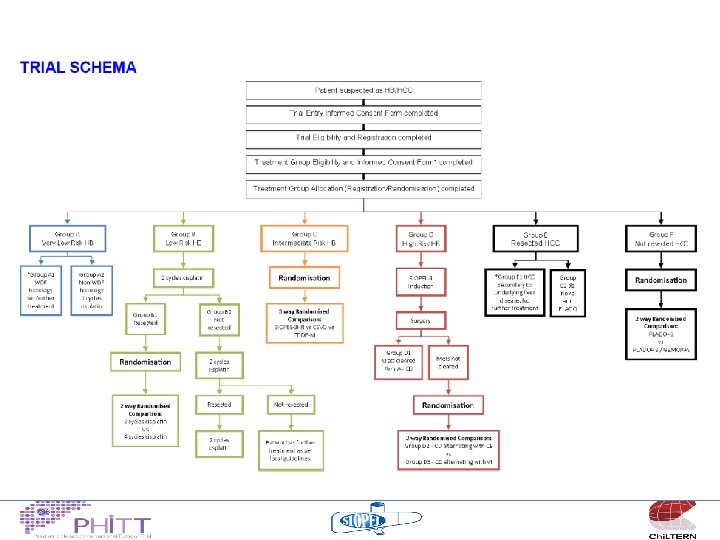
Towards a new hepatoblastoma protocol

Gdansk principles • Global study – SIOPEL, COG, JPLT, GPOH • Encompassing all liver tumours in children/young adults – HB, HCC, FLHCC, TCT…… • Common risk stratification • Integral studies – Pathology, biology, radiology, surgery, late effects • Introduce new drugs – Relapse guidelines

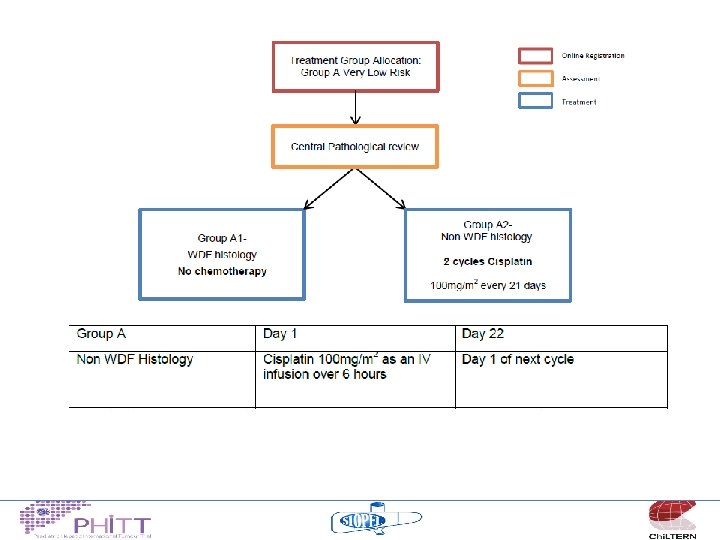
Treatment philosophy “low risk” • Continue the COG strategy for upfront resection for a selected group of tumours – Establish an agreed definition of “resectable” – No chemotherapy for well differentiated fetal histology tumours (Pure fetal) – “Minimal” chemotherapy for non-WDF tumours

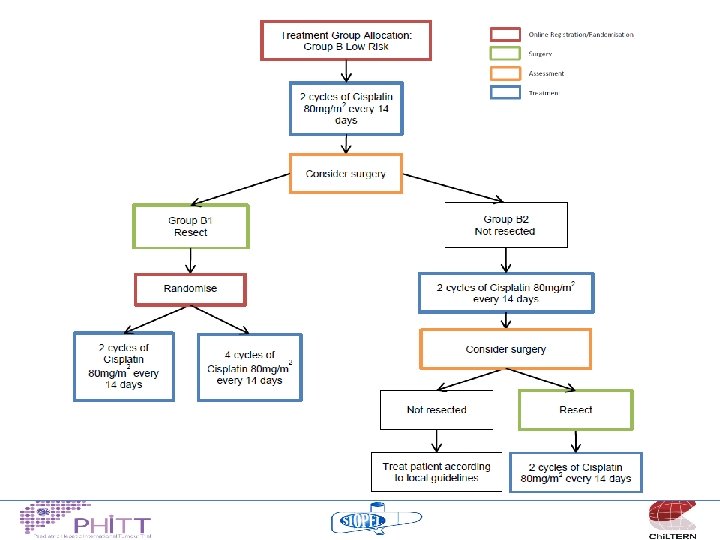
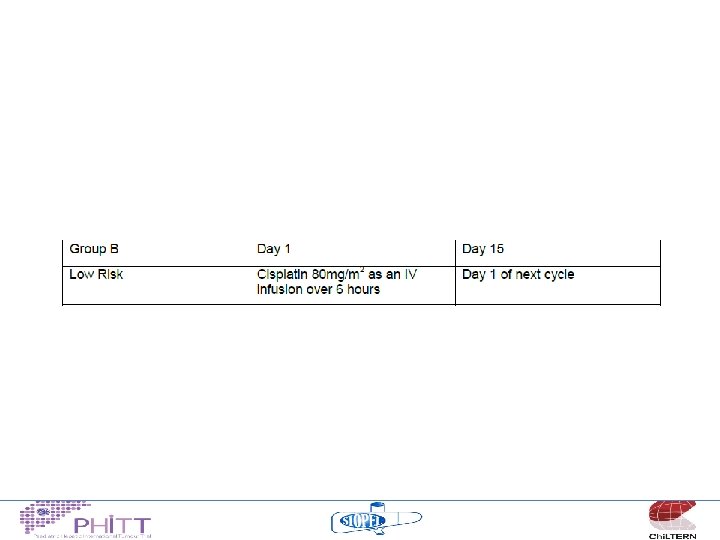
Treatment philosophy “standard risk” • Non metastatic, no adverse features – Build on the published SIOPEL experience (SIOPEL 3) cisplatin monotherapy 95% 3 y OS – Attempt to give less cisplatin 4 v 6 cycles

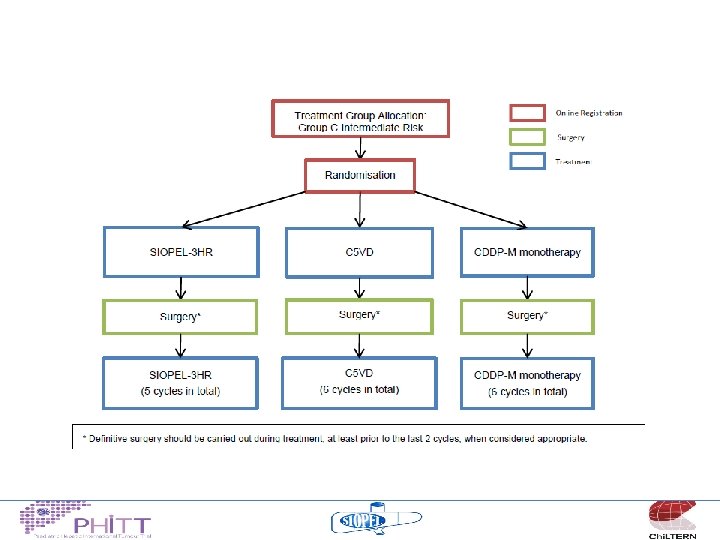
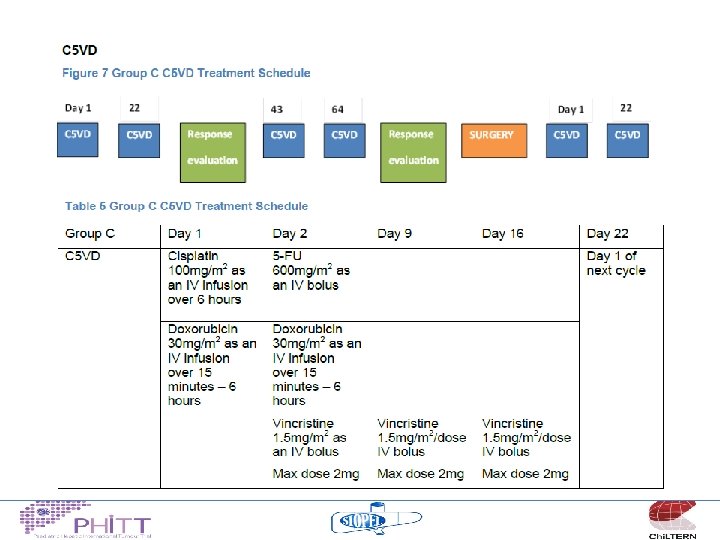
Treatment philosophy “high risk” • Non-metastatic, Pretext 4 tumours or other adverse features – These patients have challenging SURGICAL problems – 3 chemotherapy regimens appear to yield similar results (C 5 VD, SIOPEL 4 and SIOPEL 3 HR) – ? randomise between these 3

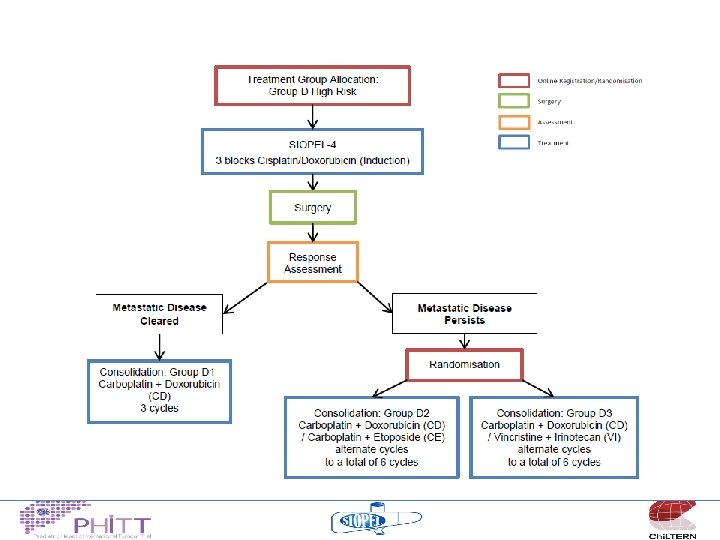
Treatment philosophy “very high risk” • Metastatic patients – Build on the encouraging results from SIOPEL 4 19/20 survivors if cleared lung mets with “induction” chemo – Intensify therapy for patients who don’t clear metastases • what with? • VCR/irinotecan, carboplatin/etoposide, ICE, sorafenib….

SIOPEL Spring 2013 Bologna: PHITT is born!




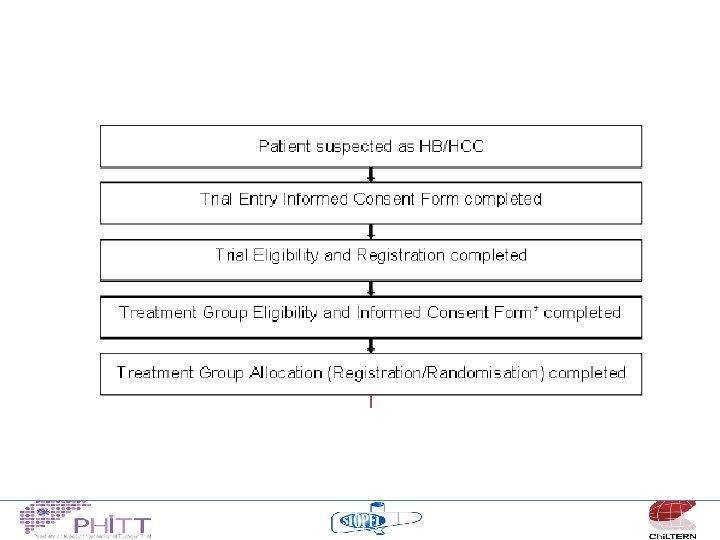
Consent/Registration “Screening” • Consent for screening PRIOR to biopsy/surgery to allow full spectrum of biological samples to be collected • Allow trial entry if biopsy/surgery has already taken place • Protocol/study set up will promote/encourage pre-biopsy/surgery consent

Consent/Registration “Trial entry” • Consent for trial entry once – Histological confirmation HB/HCC – Treatment arm assignment confirmed – Eligibility criteria met

• Risk-stratified staging in paediatric hepatoblastoma: a unified analysis from the Children's Hepatic tumors International Collaboration • • Prof Rebecka L Meyers, MD†, Rudolf Maibach, Ph. D†, Prof Eiso Hiyama, MD†, Beate Häberle, MD†, Mark Krailo, Ph. D†, Prof Arun Rangaswami, MD†, Daniel C Aronson, MD, Prof Marcio H Malogolowkin, MD, Prof Giorgio Perilongo, MD, Prof Dietrich von Schweinitz, MD, Prof Marc Ansari, MD, Prof Dolores Lopez-Terrada, MD, Yukichi Tanaka, MD, Prof Rita Alaggio, MD, Prof Ivo Leuschner, MD, Tomoro Hishiki, MD, Irene Schmid, MD, Kenichiro Watanabe, MD, Kenichi Yoshimura, Ph. D, Yurong Feng, MSe, Eugenia Rinaldi, Davide Saraceno, Marisa Derosa, Prof Piotr Czauderna, MD† †These authors contributed equally • Published: Lancet Oncology, 21 November 2016

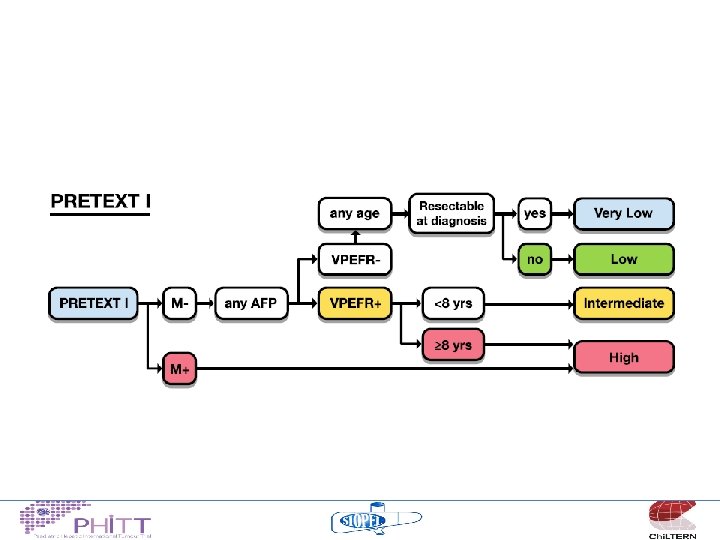
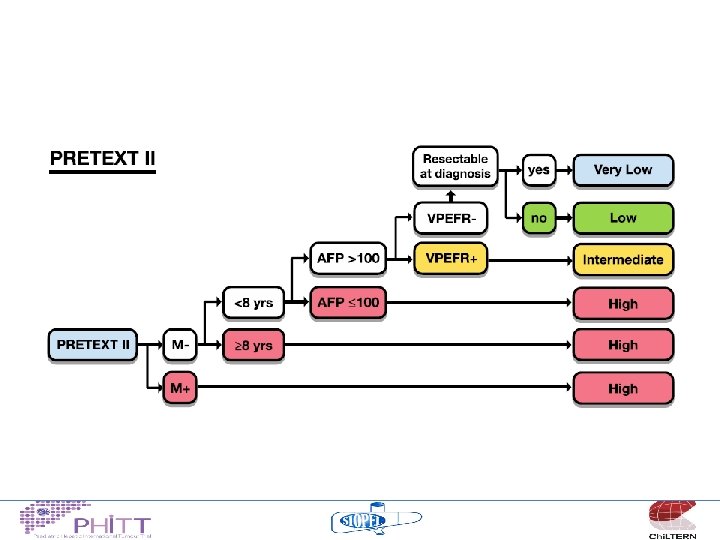
Risk Stratification • • • PRETEXT VPEFR factors Metastases Age AFP
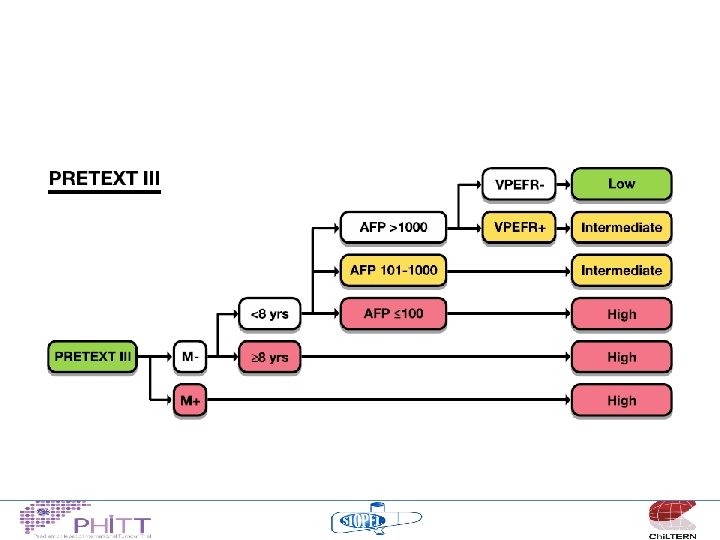
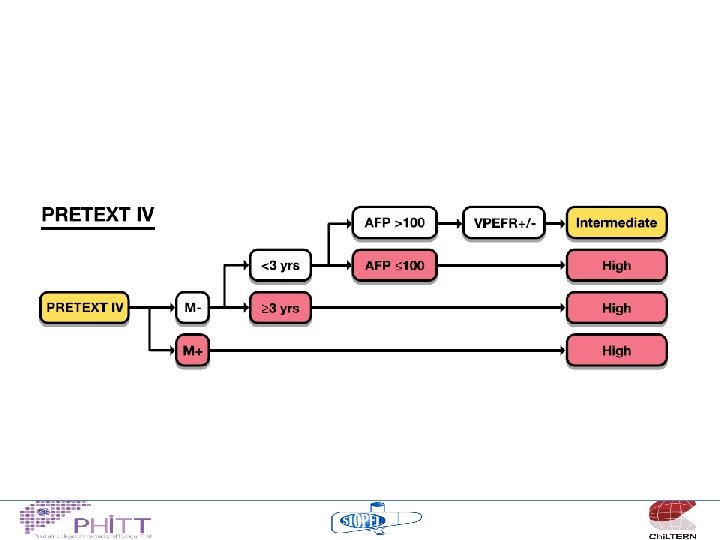


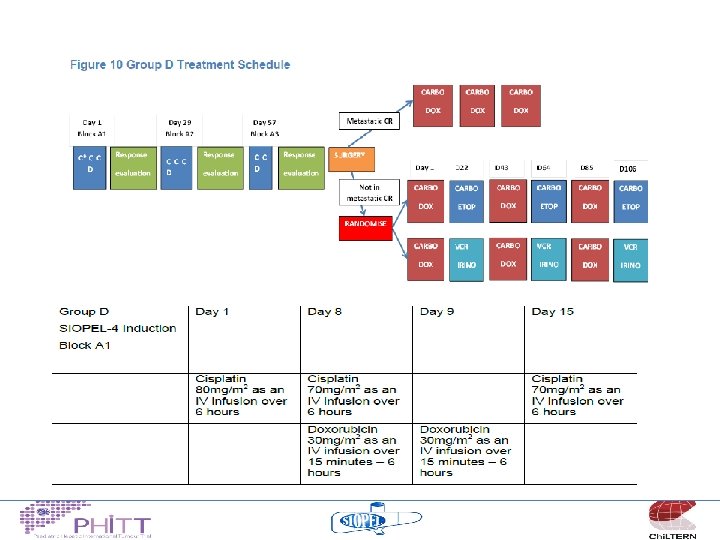
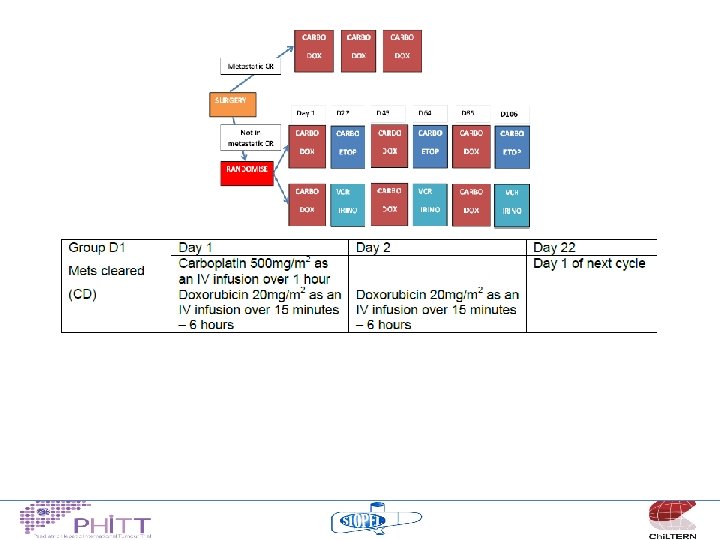
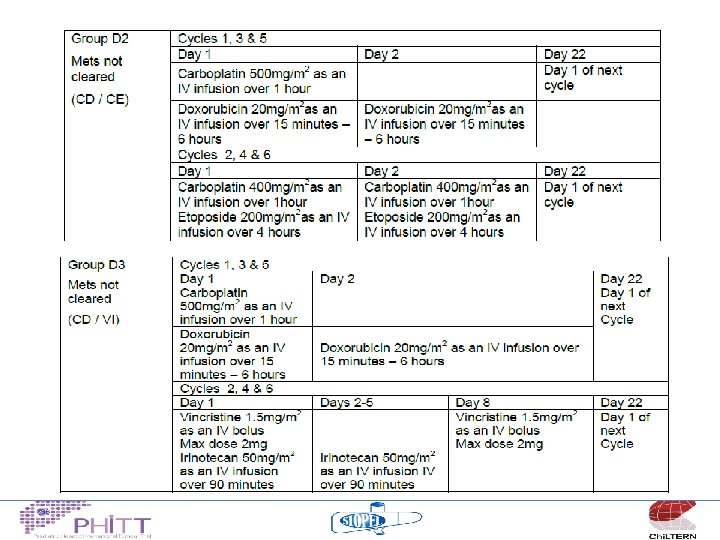
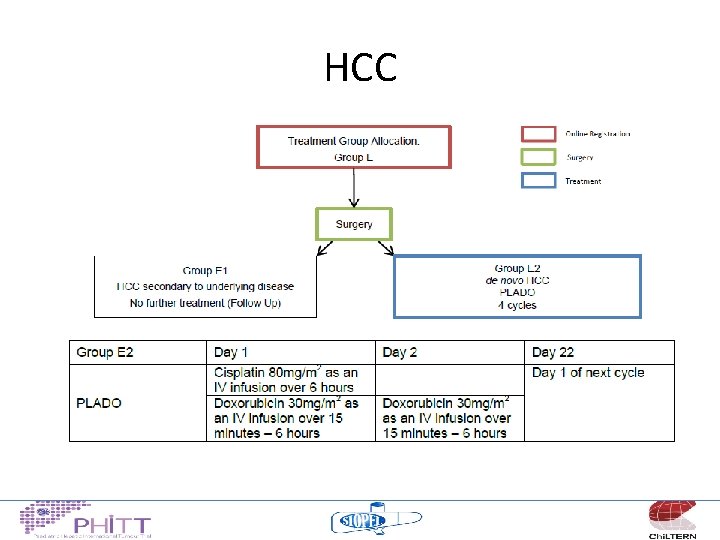
HCC
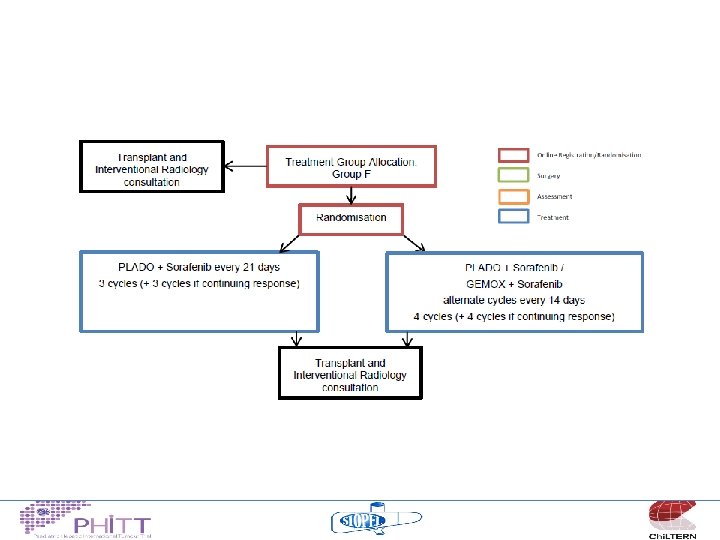
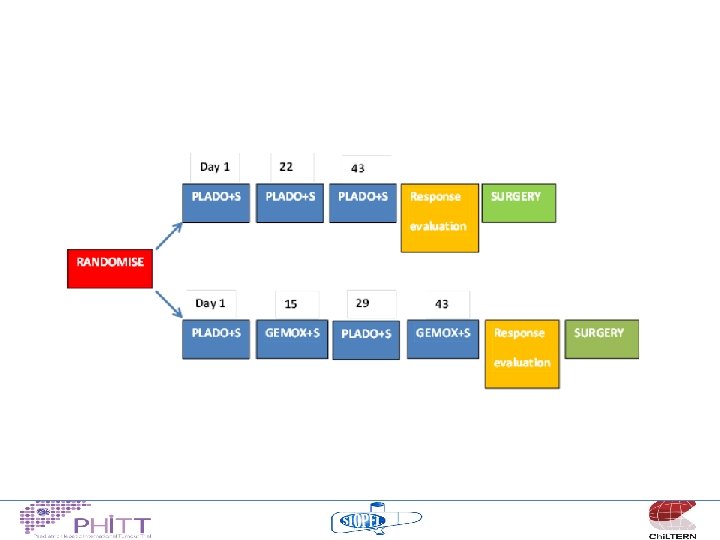
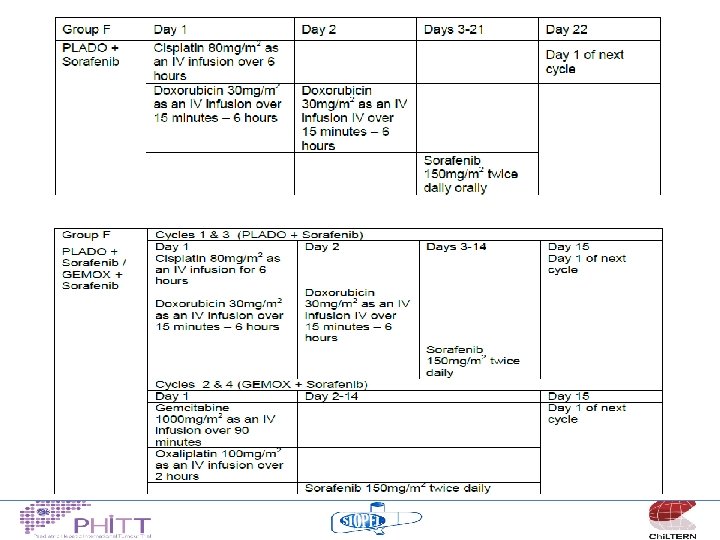

2014/15

Chi. LTERN Children’s Liver Tumour European Research Network We will aim to cure more children with liver cancer, expose fewer children to toxic chemotherapy and ensure their surgery is both effective and safe 7. 94 m EURO This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 668596

The Chi. LTERN Project • Built around the PHITT trial – Run, manage, coordinate the PHITT trial – Undertake biology • Biobanking across Europe • Confirm existing molecular biomarkers • Seek new biomarkers (risk stratification and new therapy – Clinical risk stratification • Validate existing CHIC analysis • Develop new stratification – Toxicity biomarkers • Ototoxicity, renal, cardiac – Validate Surgical planning tool • Software planning of resection margins/tumour volume/liver volume
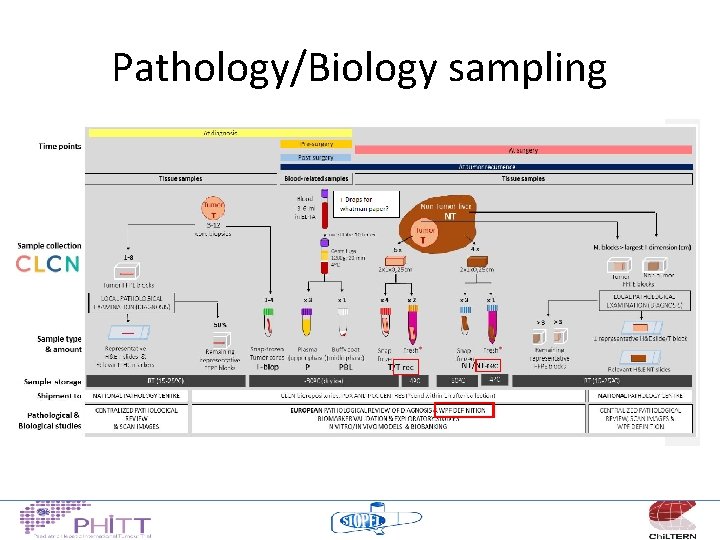
Pathology/Biology sampling


Toxicity biomarkers • Hearing (Liverpool) – SNP array • Kidney (Cincinnati) – Urine: Cystatin C, KIM-1, NGAL • Heart (Cincinnati) – Blood: Troponin, NT-pro. BNP • PK/PD (Newcastle) – Correlation with toxicity biomarkers (Cmax, AUC)

Highlights from last 6 months • ECCO 2016 – Amsterdam – SIOPE presentation. EU Commission present • “PHITT is my new favourite study, it’s my baby, I love it” • NCI Pediatric Steering Committee – Approval in February 2017 • UK regulatory submissions – MHRA – approved with minor fertility clarification – National ethics • “This is one of the best applications this committee has ever seen……. . ” • First draft of COG protocol submitted to Scientific Council • First UK site initiation visit May 25 th

Next steps • • • Roll out of EU sites CRF generation Agree surgical guidelines Agree radiology guidelines Agree pathology guidelines – EU path meeting 19 th May Utrecht • “Merger” of COG/JPLT/EU protocols • 1 st patients summer 2017

Allow me to anticipate one of your questions! How can my centre participate in PHITT?

Acknowledgements PHITT • • • Greg Tiao – COG Jim Geller – COG Eiso Hiyama – JPLT All my SIOPEL colleagues Jennifer Laidler, Veronica Moroz, Sam Munoz Vincente, Nicola Fenwick, Keith Wheatley Birmingham CRCTU Chi. LTERN • • • Carolina Armengol – Barcelona Beate Haeberle – Munich Steven Warmann – Tuebingen Gareth Veal – Newcastle Louise Woodall, Neil Werrett, Steve Baker, Poonam Patel, Keith Wheatley – Birmingham CRCTU

- Slides: 53