Phases of Matter Phases of Matter can take











- Slides: 14

Phases of Matter

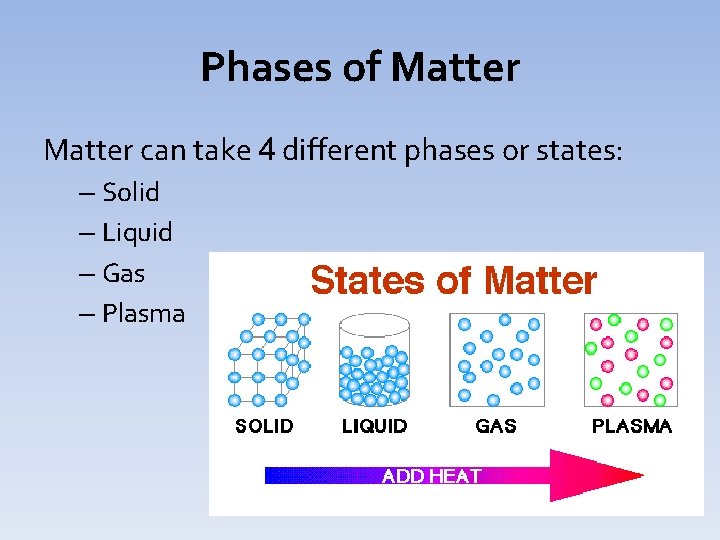
Phases of Matter can take 4 different phases or states: – Solid – Liquid – Gas – Plasma

Solids • Solids are substances that have a definite shape and a definite volume. • In other words, the shape of a solid never changes, nor does the space it takes up (volume. ) • The particles in a solid are packed tightly together so this is why its shape and volume don’t change.

2 Types of Solids 1. Crystalline Solids- particles are arranged in a repeating pattern. - These substances do NOT lose shape when heated. - Examples- sugar, sand, salt

2. Amorphous Solids- particles are NOT arranged in a repeating pattern. - These substances lose their shape when heated. - Examples- plastics, rubber, glass

Liquids • Liquids are substances that have a definite volume but no definite shape. • In other words, the shape of a liquid can change but the space it takes up (volume) cannot. • The particles in a liquid are packed almost as tightly as a solid but can move around one another so this is why its shape can change.

Gases • Gases are substances that have no definite shape and no definite volume. • In other words, the shape of a gas can change and so can the space it takes up (volume). • The particles in a gas are spread very far apart, filling all the space available to them.

Plasma • Plasma is a state of matter that occurs at very high temperatures. • It is too cold on Earth for most matter to reach the plasma state. • Our sun and small stars are made of plasma. • Lighting strikes are plasma and plasma glows when it conducts electricity in neon signs and fluorescent bulbs. • The hottest candle flame is plasma.

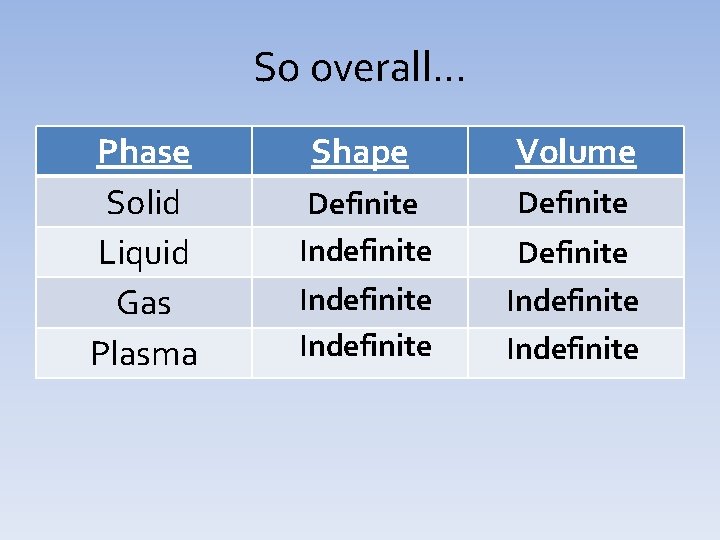
So overall… Phase Solid Liquid Gas Plasma Shape Volume Definite Indefinite Definite Indefinite

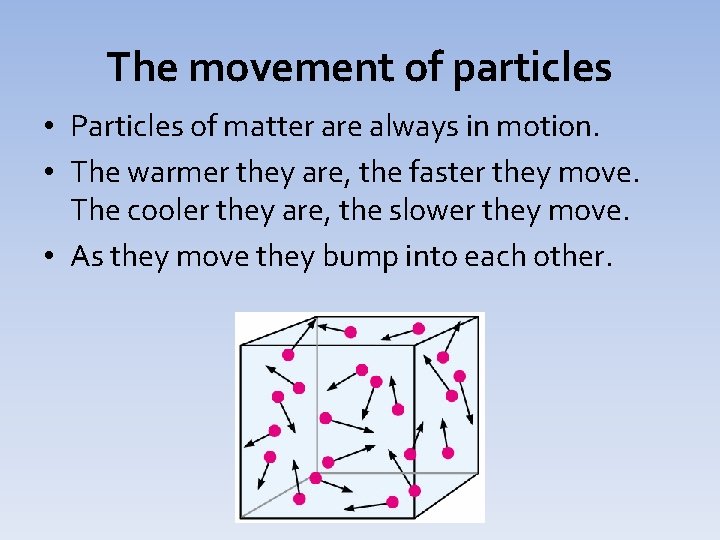
The movement of particles • Particles of matter are always in motion. • The warmer they are, the faster they move. The cooler they are, the slower they move. • As they move they bump into each other.

Phase Changes • As a substance gets warmer, its particles bump into each other more causing them to spread apart. • This causes solids to melt into liquids and liquids to evaporate into gases.

Phase Changes • As a substance gets cooler, its particles bump into each other less causing them to get closer together. • This causes gases to condense into liquids and liquids to freeze into solids.

S L Melting L S Freezing L G Evaporating G L Condensing

Sublimation • When a substance changes from a solid straight to a gas (skipping liquid), it sublimes. • S G • Only some substances can do this like dry ice and iodine.