Phases of Matter Grab a piece of construction

Phases of Matter

Grab a piece of construction paper and a pair of scissors from the desk. Then discuss the following questions with a neighbor. What happens to a puddle of water on a sunny day? � What happens to ice on a warm day? � What happens to a pond in very cold temperatures? �

http: //www. shmoop. com/video /chemistry-3 -1 -the-states-ofmatter? tag=6 FDC 1073429 B 473 D 9451 D 81983782473


With your table, list some examples of solids. Then brainstorm with your group to see if you can come up with at least 3 things (properties) that all of the solids have in common.

Phases of Matter • Solids: Particles are tightly packed together and DO NOT move past each other. • They vibrate in place. © 2013 S. Coates

Phases of Matter • Solids have a definite. SHAPE • Solids have a definite. VOLUME Example—Marble Shape= Sphere Volume= can be found using water displacement © 2013 S. Coates

Phases of Matter • Examples of Solids: © 2013 S. Coates

With your table, list some examples of liquids. Then brainstorm with your group to see if you can come up with at least 3 things (properties) that all of the liquids have in common.

Phases of Matter • Liquids: Particles are still tightly packed together and they SLIDE move past each other. © 2013 S. Coates

Phases of Matter • Examples of Liquids: © 2013 S. Coates

Phases of Matter • Liquids. DO NOThave a definite. SHAPE, they take the shape of their container. Example—Orange Juice • Liquids have a definite. VOLUME Shape= None, it takes the shape of the glass. Volume= can be found using a beaker or graduated cylinder. © 2013 S. Coates

With your table, list some examples of gases. Then brainstorm with your group to see if you can come up with at least 3 things (properties) that all of the solids have in common.

Phases of Matter • Gases: Particles are not tightly packed together, and have so much energy they slip past each other quickly. © 2013 S. Coates

Phases of Matter • Examples of Gases: © 2013 S. Coates

Phases of Matter • Gases. DO NOThave a definite. SHAPE • Gases. DO NOThave a definite. VOLUME Example—Smoke Shape= Not definite. Volume= Not definite. Gases are usually always expanding. © 2013 S. Coates

Phases of Matter • Plasma: Particles are moving so quickly it is hard to see what they are actually doing. © 2013 S. Coates

Phases of Matter • Examples of Plasmaon Earth: © 2013 S. Coates
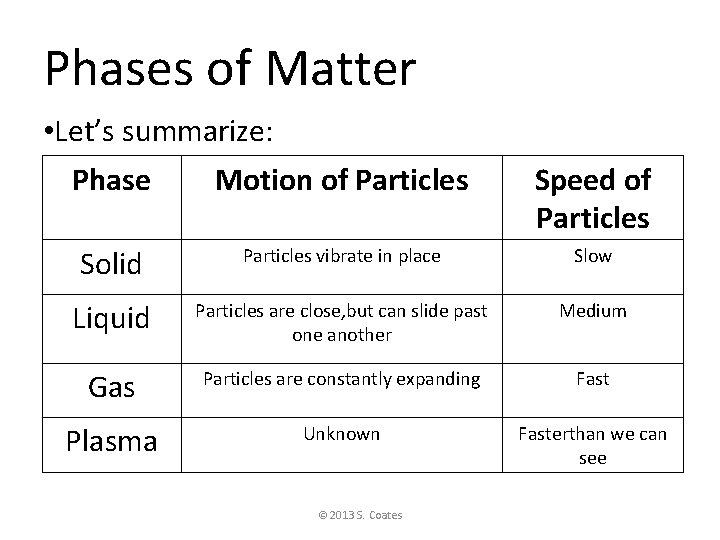
Phases of Matter • Let’s summarize: Phase Motion of Particles Speed of Particles Solid Particles vibrate in place Slow Liquid Particles are close, but can slide past one another Medium Gas Particles are constantly expanding Fast Plasma Unknown Fasterthan we can see © 2013 S. Coates
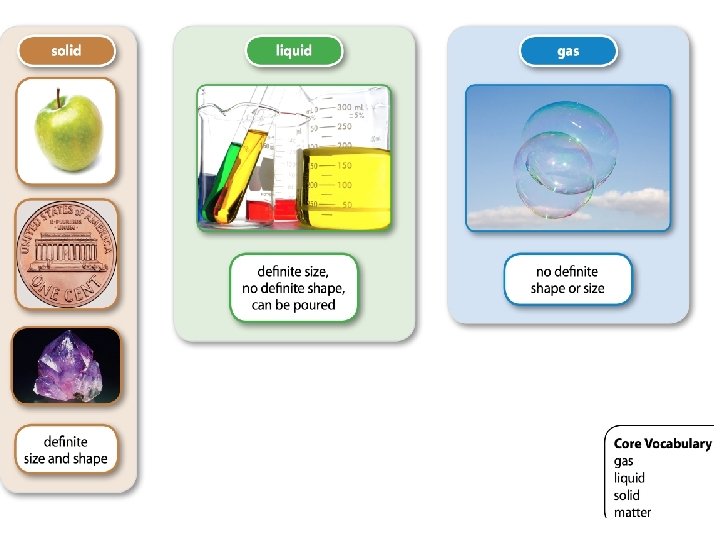

Each card has with either the name of the category, a definition, an example, or a supporting fact and they will need to wander around the room trying to connect with the people who are in the same state of matter.

Hot and Cold Simulation

Phases of Matter • Two “laws” about gases… 1. Charles’ Law • • • T= V Volume (of gas) and Temperature When temperature goes up, volume goes up When temperature goes down, volume goes down © 2013 S. Coates

Gas + Heat = Expansion! © 2013 S. Coates http: //www. coloradoguy. com/balloona-vista/hotairballoons-buenavista-co. htm http: //www. usaballoon. com/fly. htm

Phases of Matter • Two “laws” about gases… 2. Boyles’ Law V= P • Volume (of gas) and Pressure • When pressure goes up, volume goes down • When pressure goes down, volume goes up © 2013 S. Coates

The amount of water pressure determines the size of bubbles in the water. © 2013 S. http: //www. gettyimages. com/detail/91300130/Photographers-Choice Coates http: //gallery. photo. net/photo/9734756 -lg. jpg
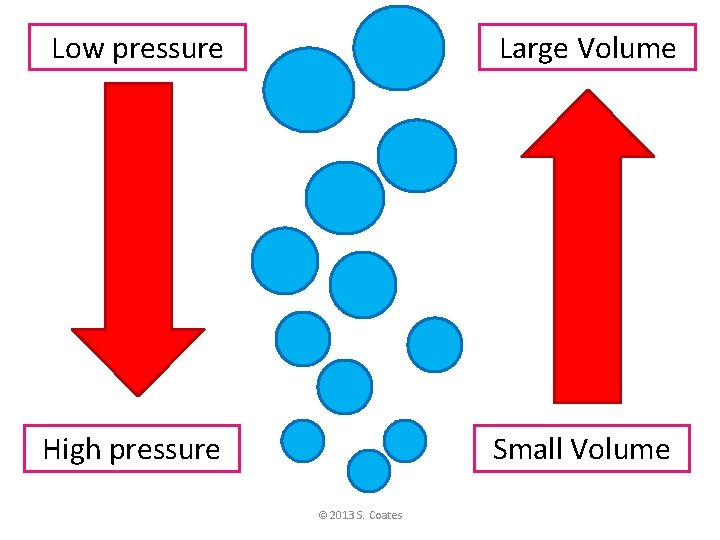
Low pressure Large Volume High pressure Small Volume © 2013 S. Coates

Phases of Matter • Let’s summarize: Phase Solid Definite Shape? YES Definite Volume? YES Liquid NO YES Gas NO NO Plasma http: //www. harcourtschool. com/activity/st ates_of_matter/
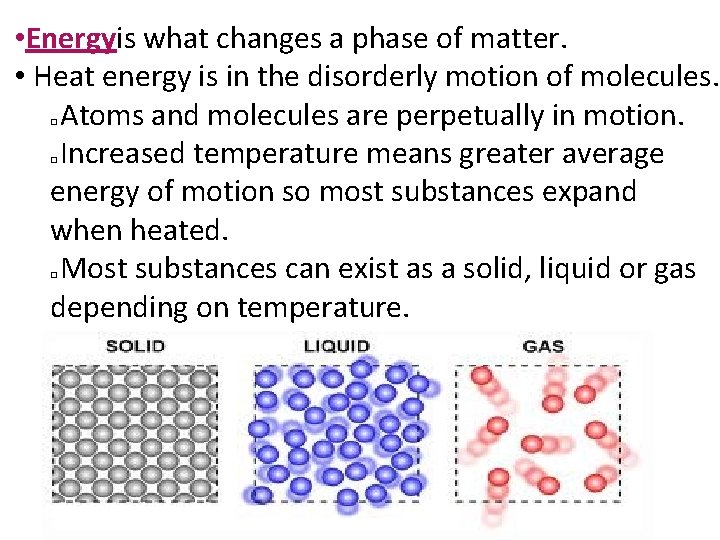
• Energyis what changes a phase of matter. • Heat energy is in the disorderly motion of molecules. Atoms and molecules are perpetually in motion. Increased temperature means greater average energy of motion so most substances expand when heated. Most substances can exist as a solid, liquid or gas depending on temperature. � � � © 2013 S. Coates

Phases of Matter ADDED • Is ENERGY being ADDED or TAKEN AWAY in this phase change: The added energy has caused the chocolate particles to speed up. Before they were vibrating in place, now they are moving fast enough to slip past one another. Solid Liquid © 2013 S. Coates

Phases of Matter • Is ENERGY being. ADDEDor. TAKEN AWAYin this phase change: ADDED The added energy has caused the water particles tospeed up. Before they were moving fast enough to slip past one another, now they have enough energy to break away from one another and expand. Liquid Gas © 2013 S. Coates

Phases of Matter • Is ENERGY being. ADDEDor. TAKEN AWAYin this phase change: Taken Away Taking away energy from a rain drop slows the water molecules down so that they no longer slide past one another. Liquid Solid © 2013 S. Coates

Please grab the two sheet from the desk. • Write your name on the Bill Nye worksheet and then set it aside. • Begin filling in what you know on the states of matter handout.

Phases Change simulation

Study Jams http: //www. shmoop. com/video/chemistry-3 -1 -the-states-ofmatter? tag=6 FDC 1073429 B 473 D 9451 D 81983782473 http: //studyjams. scholastic. com/studyjams/science/matter/solids-liquidsgases. htm

Grab the sheet from the front and write your name on it. Match the phase change with the correct description: melting freezing evaporation condensation 1. temperature increase, liquid to gas 2. temperature increase, solid to liquid 3. temperature decrease, gas to liquid 4. temperature decrease, liquid to solid 2. What is plasma and what is an example of plasma?

Read over the lab for today. Be ready to start the lab soon! BYOD – but face down and not on 12/15/2021

http: //phet. colorado. edu/en/simulation/states-of-matter http: //www. brainpop. com/games/mattersorter/ https: //www. youtube. com/watch? v=Fj 1 Id. Odm. Oj. Y&feature=youtu. be

Take the next couple of minutes to look over your notes to prepare for the quiz. Vocabulary on quiz: • Solid, liquid, gas • Melting, freezing, boiling, evaporation, condensation, sublimation, deposition • Know when heat is added and when heat is removed for phase changes Ex: State of Matter + o r- heat = process Ending state of matter GAS -- HEAT = DEPOSITION SOLID

Pick your group of 4 but do not sit with them yet. We will be completing a lab today. Grab the lab sheet from the front and begin reading over it.
- Slides: 40