Phases of Matter and Phase Changes HEAT n





























- Slides: 35

Phases of Matter and Phase Changes

HEAT n n Heat is one form of __________ Other forms of energy include: ¨ Light ¨ Sound ¨ Nuclear ¨ Kinetic (energy of motion) ¨ Potential (stored energy)

HEAT n n Heat is one form of ENERGY Other forms of energy include: ¨ Light ¨ Sound ¨ Nuclear ¨ Kinetic (energy of motion) ¨ Potential (stored energy)

Heat is. . . A measure of how much ______ energy matter has n Particles with high heat are moving at a ______ average speed n Particles with low heat are moving at a ______ average speed n

Heat is. . . A measure of how much kinetic energy matter has n Particles with high heat are moving at a HIGH average speed n Particles with low heat are moving at a LOW average speed n

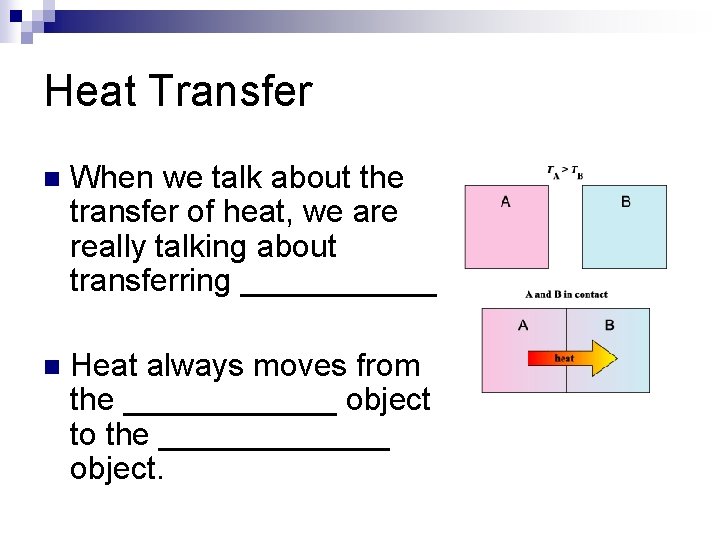
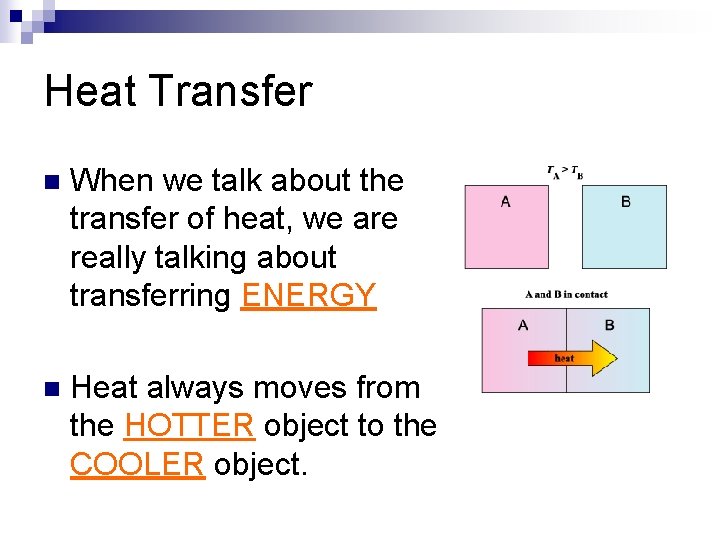
Heat Transfer n When we talk about the transfer of heat, we are really talking about transferring ______. n Heat always moves from the ______ object to the _______ object.

Heat Transfer n When we talk about the transfer of heat, we are really talking about transferring ENERGY n Heat always moves from the HOTTER object to the COOLER object.

Heat and the Motion of Molecules n When we add heat (energy to molecules), it does WHAT to their motion? Heating water animation: n http: //mutuslab. cs. uwindsor. ca/schurko/ani mations/waterphases/status_water. htm n

Heat and the Motion of Molecules n When we add heat (energy to molecules), it does WHAT to their motion? Heat increases the motion and energy level of the molecules Heating water animation: n http: //mutuslab. cs. uwindsor. ca/schurko/ani mations/waterphases/status_water. htm n

Phases of Matter n There are four main phases of matter: ¨ Solid ¨ Liquid ¨ Gas ¨ _______? n n ____ is the phase where molecules have the most energy ____ is the phase where molecules have the least energy

Phases of Matter n There are four main phases of matter: ¨ Solid ¨ Liquid ¨ Gas ¨ PLASMA. . . we will not study in depth, but technically has the most energy n n GAS is the phase where molecules have the most energy SOLID is the phase where molecules have the least energy

Phases of Matter: Solid Particles have little motion/energy; they are packed/joined together tightly n Particles are very organized n Do NOT move to fit the shape of their container and do not fill the container (they keep their original shape) n It is hard to compress or change the shape of a solid n

Phases of Matter: Liquid Particles have more motion/energy; joined together more loosely n Particles WILL take on the shape of their container but will not fill it entirely n Molecules of a liquid like to stick together. Liquids are hard to compress. n

Phases of Matter: Gases Particles have a great deal of energy and do not stick together closely; they bounce around a lot! n WILL spread out to fill entire container and fit the shape of the container they are in n Gases=vapor n Gases are easily compressed n

Phases of Matter: Plasma Similar to gases, but made up of ions and electrons of an element n Examples: Northern lights, neon lights, stars n Require a special environment to exist n

Phase Changes A phase change is when we change from _____ state of matter to ___________. Example: ____________

Phase Changes A phase change is when we change from one state of matter to another Example: A solid changing to a liquid

Phases of Matter In the boxes below draw the three phases of matter. Use 20 molecules for each phase. SOLID LIQUID GAS

Phases of Matter http: //mutuslab. cs. uwindsor. ca/schurko/animations/phasesofmatter. html

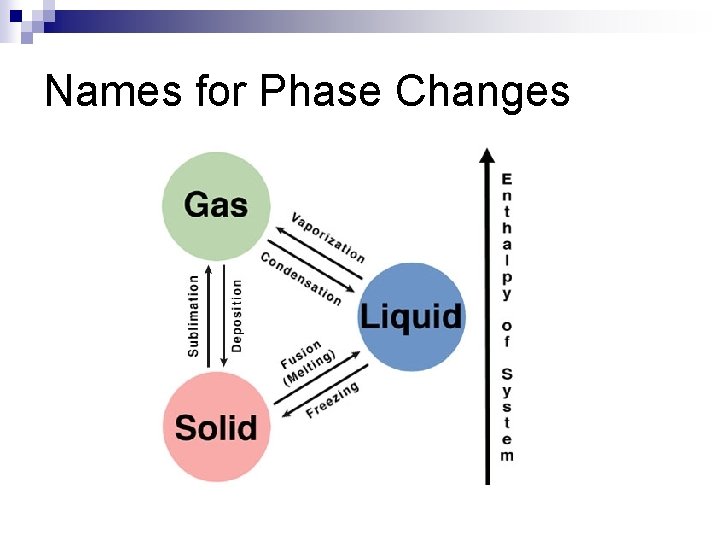
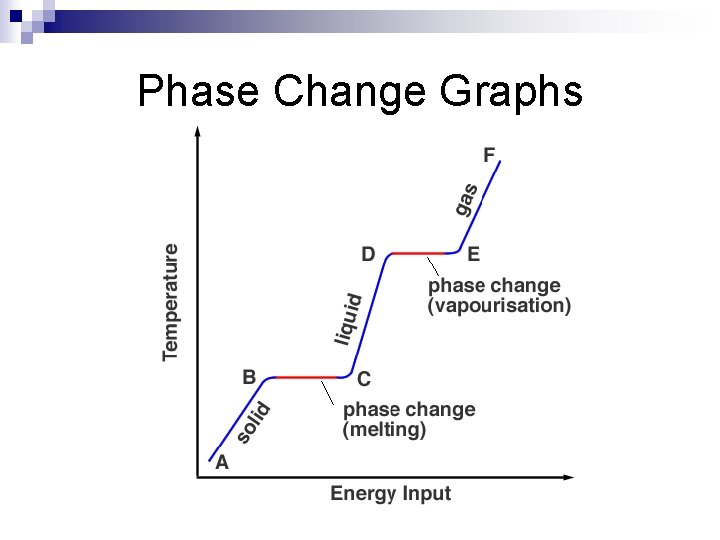
Names for Phase Changes Solid to liquid: n Liquid to gas: n Solid to gas: n Gas to liquid: n Liquid to solid: n Gas to solid: n

Names for Phase Changes Solid to liquid: MELTING n Liquid to gas: VAPORIZATION/BOILING n Solid to gas: SUBLIMATION n Gas to liquid: CONDENSATION n Liquid to solid: FREEZING n Gas to solid: DEPOSITION n

Names for Phase Changes

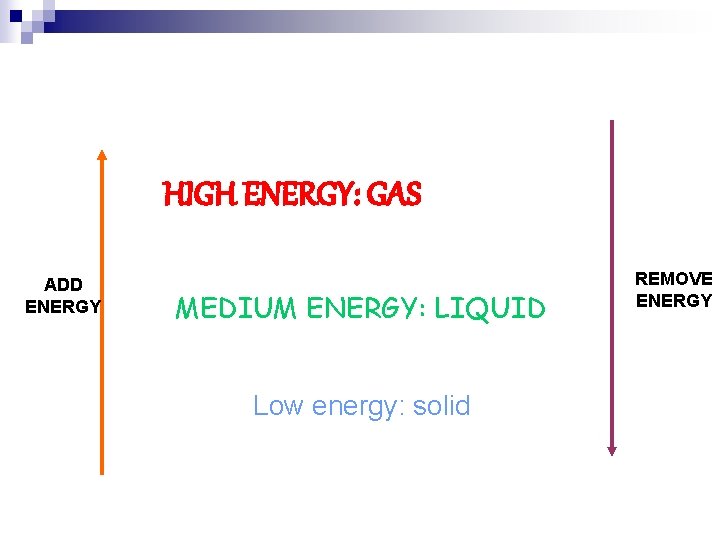
HIGH ENERGY: GAS ADD ENERGY MEDIUM ENERGY: LIQUID Low energy: solid REMOVE ENERGY

Phase Changes n To go to a state of matter involving MORE energy, we must ______ energy. We can do this by _____ the substance. n Examples: If we heat ice (____), it will turn into water (______), and will eventually boil and turn into water vapor (_____)

Phase Changes n To go to a state of matter involving MORE energy, we must ADD energy. We can do this by HEATING the substance. n Examples: If we heat ice (solid), it will turn into water (liquid), and will eventually boil and turn into water vapor (gas)

Phase Changes n To go to a state of matter involving LESS energy, we must ______ energy. We can do this by _____ the substance. n Examples: Water vapor (______) from your hot shower condenses on a cold mirror (____). If you leave a window open, it might turn into ice droplets (____).

Phase Changes n To go to a state of matter involving LESS energy, we must REMOVE energy. We can do this by COOLING the substance. n Examples: Water vapor (gas) from your hot shower condenses on a cold mirror (liquid). If you leave a window open, it might turn into ice droplets (solid).

Check your understanding n n n What are three main states of matter? ___________ List these three states of matter from the state with the LEAST amount of energy to the state with the MOST energy: ___________ To go from a gas to a liquid, would you need to ADD energy (heat) or RELEASE energy (cool)? __________

Check your understanding n What are three main states of matter? ___GAS___ ___LIQUID___ ___SOLID___ n List these three states of matter from the state with the LEAST amount of energy to the state with the MOST energy: ___SOLID___ ___LIQUID___ ___GAS___ n To go from a gas to a liquid, would you need to ADD energy (heat) or RELEASE energy (cool)? ___RELEASE___

Melting Points and Boiling Points are Properties of Matter We observed that water _____ at 0 degrees Celsius n We observed that water ____ at 100 degrees Celsius n Water’s natural state at room temperature (25 degrees Celsius) is as a _____ n

Melting Points and Boiling Points are Properties of Matter We observed that water MELTS/FREEZES at 0 degrees Celsius n We observed that water BOILS/CONDENSES at 100 degrees Celsius n Water’s natural state at room temperature (25 degrees Celsius) is as a LIQUID n

Different Substances will have Different Boiling Points and Melting Points What is the natural state of oxygen at room temperature? n Could we condense nitrogen? Could we freeze nitrogen? n Can you name three substances that are solids at room temperature? n

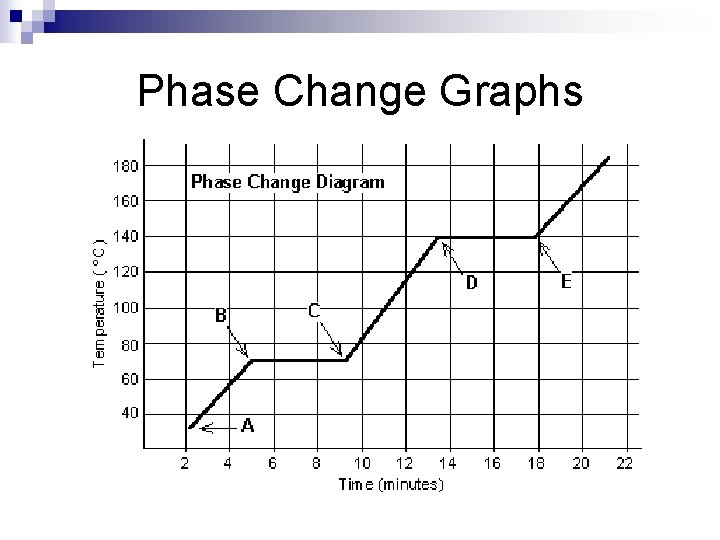
Check your Understanding Why are melting points and boiling points considered to be properties of matter? n Can you look at a Phase Change Graph and tell at which temperature the substance begins melting and begins boiling? n

Phase Change Graphs

Phase Change Graphs