Phase Transformations Thermodynamic Basis ByeongJoo Lee POSTECH MSE

Phase Transformations Thermodynamic Basis Byeong-Joo Lee POSTECH - MSE calphad@postech. ac. kr Byeong-Joo Lee cmse. postech. ac. kr
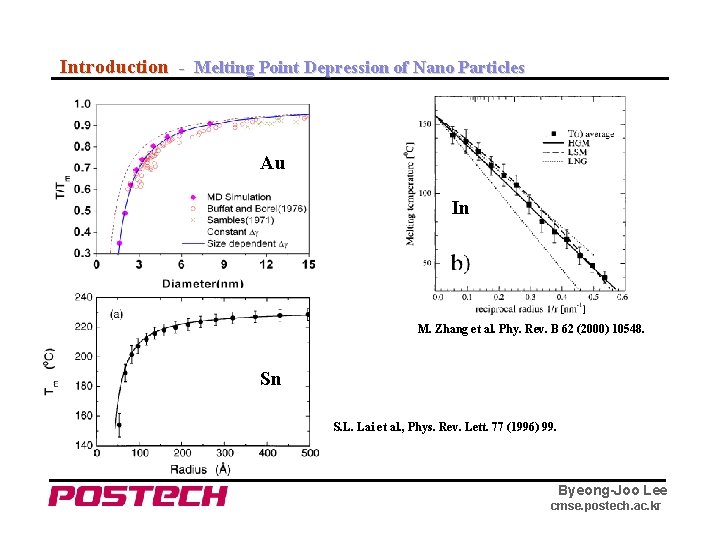
Introduction - Melting Point Depression of Nano Particles Au In M. Zhang et al. Phy. Rev. B 62 (2000) 10548. Sn S. L. Lai et al. , Phys. Rev. Lett. 77 (1996) 99. Byeong-Joo Lee cmse. postech. ac. kr
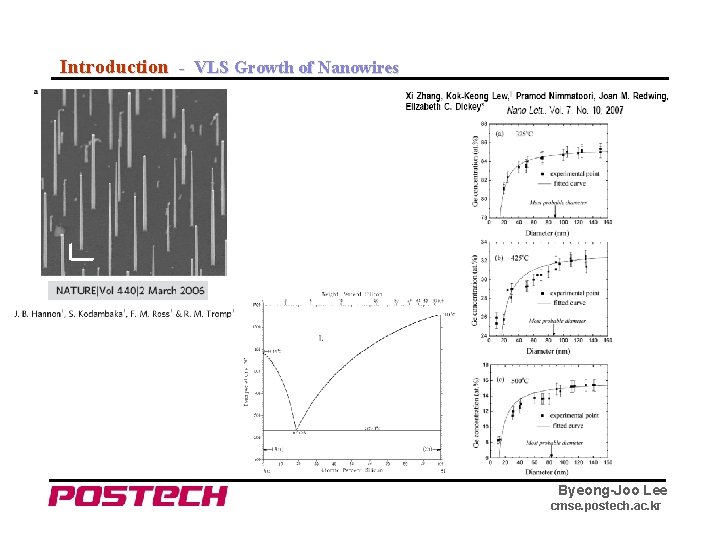
Introduction - VLS Growth of Nanowires Byeong-Joo Lee cmse. postech. ac. kr

Gibbs Energy ▷ Effect of Temperature on G ▷ Effect of Pressure on G Byeong-Joo Lee cmse. postech. ac. kr

Gibbs Energy for a Unary System - from d. G = –Sd. T + Vd. P Gibbs Energy as a function of T and P @ constant T Byeong-Joo Lee cmse. postech. ac. kr

Gibbs Energy for a Unary System - from G = H – ST Gibbs Energy as a function of T and P @ constant T Byeong-Joo Lee cmse. postech. ac. kr

Gibbs Energy for a Unary System - Temperature Dependency 서로 다른 출발점에서 유도된 위의 두 식은 같은 식인가? Empirical Representation of Heat Capacities 를 이용하여 위의 두 식이 동일한 것임을 증명하라. Byeong-Joo Lee cmse. postech. ac. kr

Gibbs Energy for a Unary System - Effect of Pressure Molar volume of Fe Expansivity = 7. 1 cm 3 = 0. 3 × 10 -4 K-1 ∆H(1→ 100 atm, 298) = 17 cal ※ The same enthalpy increase is obtained by heating from 298 to 301 K at 1 atm Molar volume of Al Expansivity = 10 cm 3 = 0. 69 × 10 -4 K-1 ∆H(1→ 100 atm, 298) = 23. 7 cal ※ The same enthalpy increase is obtained by heating from 298 to 302 K at 1 atm ∆S(1→ 100 atm, 298) = -0. 00052 e. u. for Fe -0. 00167 e. u. for Al ※ The same entropy decrease is obtained by lowering the temperature from 298 by 0. 27 and 0. 09 K at 1 atm. ※ The molar enthalpies and entropies of condensed phases are relatively insensitive to pressure change Byeong-Joo Lee cmse. postech. ac. kr

Gibbs Energy for a Unary System - absolute value available ? • V(T, P) based on expansivity and compressibility • Cp(T) • S 298: by integrating Cp/T from 0 to 298 K and using 3 rd law of thermodynamics (the entropy of any homogeneous substance in complete internal equilibrium may be taken as zero at 0 K) • H 298: from first principles calculations, but generally unknown ※ H 298 becomes a reference value for GT ※ Introduction of Standard State Byeong-Joo Lee cmse. postech. ac. kr

Standard States Byeong-Joo Lee cmse. postech. ac. kr

Gibbs Energy, Chemical Potential and Activity ▷ Gibbs energy of mixing vs. Gibbs energy of formation ▷ activity wrt. liquid A or B ▷ activity wrt. “ref” A or B ▷ activity wrt. [ ]i Byeong-Joo Lee cmse. postech. ac. kr

Gibbs Energy ▷ Effect of Composition on G Byeong-Joo Lee cmse. postech. ac. kr
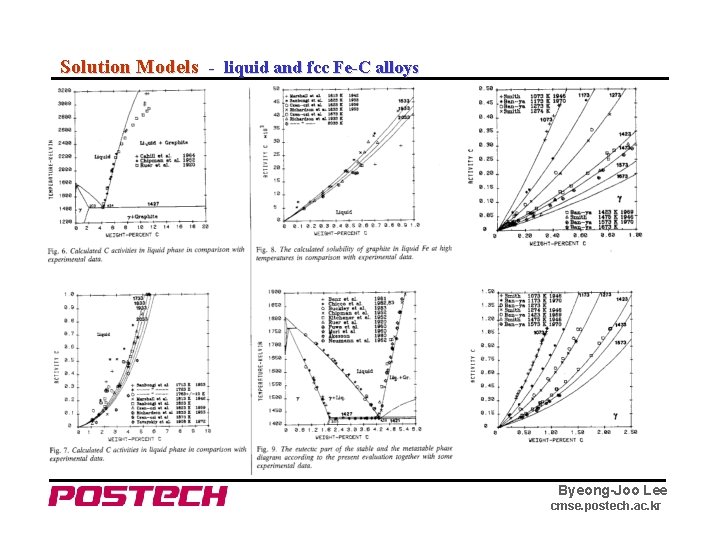
Solution Models - liquid and fcc Fe-C alloys Byeong-Joo Lee cmse. postech. ac. kr

Solution Model - liquid and fcc Fe-C alloys fcc : (Fe)1(Va, C) 1 Liquid : (Fe, C) Ternary (Fe, Mn)a(Va, C) c Byeong-Joo Lee cmse. postech. ac. kr

Thermodynamic Modeling – Gibbs Energy For a Phase with Formula Unit, (M 1, M 2, …, Mi, …)a(Va, C, N)c Byeong-Joo Lee cmse. postech. ac. kr

Thermodynamic Modeling – Sublattice Model LIQUID BCC_A 2 FCC_A 1 HCP_A 3 M 6 C M 23 C 6 M 7 C 3 CEMENTITE SIGMA CHI_A 12 LAVES_PHASE MU_PHASE P_PHASE R_PHASE KSI_CARBIDE (C, Cr, Fe, Mn, Mo, N, Ni, Si)1 (Cr, Fe, Mn, Mo, Ni, Si)1(C, N, Va)3 (Cr, Fe, Mn, Mo, Ni, Si)1(C, N, Va)1 (Cr, Fe, Mn, Mo, Ni, Si)1(C, N, Va). 5 (Fe, Ni)2(Mo)2(Cr, Fe, Mo, Ni)2(C)1 (Cr, Fe, Mn, Ni)20(Cr, Fe, Mn, Mo, Ni)3(C)6 (Cr, Fe, Mn, Mo, Ni)7(C)3 (Cr, Fe, Mn, Mo, Ni)3(C, N)1 (Fe, Mn, Ni)8(Cr, Mo)4(Cr, Fe, Mn, Mo, Ni, Si)18 (Cr, Fe, Ni)24(Cr, Mo)10(Cr, Fe, Mo, Ni)24 (Cr, Fe, Mn, Mo, Si)2(Cr, Fe, Mn, Mo, Si)1 (Cr, Fe, Mn, Ni)7(Mo)2(Cr, Fe, Mo, Ni)4 (Cr, Fe, Ni)24(Cr, Fe, Mo, Ni)20(Mo)12 (Cr, Fe, Mn, Ni)27(Mo)14(Cr, Fe, Mn, Mo, Ni)12 (Cr, Fe, Mo)3(C)1 Byeong-Joo Lee cmse. postech. ac. kr

Phase Equilibria Byeong-Joo Lee cmse. postech. ac. kr

Interface Energy - Curvature effect Byeong-Joo Lee cmse. postech. ac. kr

Curvature Effect – Capillary Pressure System condition T = constant Vα = Vβ = V = constant @ equilibrium Byeong-Joo Lee cmse. postech. ac. kr

Curvature Effect – Capillary Pressure Effect on Vapor Pressure and Solubility Vapor Pressure Solubility of pure B phase in a dilute solution Byeong-Joo Lee cmse. postech. ac. kr
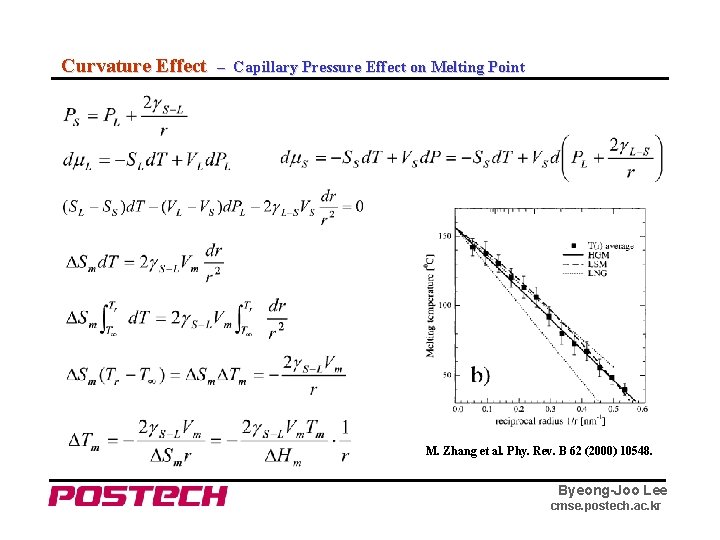
Curvature Effect – Capillary Pressure Effect on Melting Point M. Zhang et al. Phy. Rev. B 62 (2000) 10548. Byeong-Joo Lee cmse. postech. ac. kr

Curvature Effect – Capillary Pressure Effect on Melting Point of Nano Particle Byeong-Joo Lee cmse. postech. ac. kr
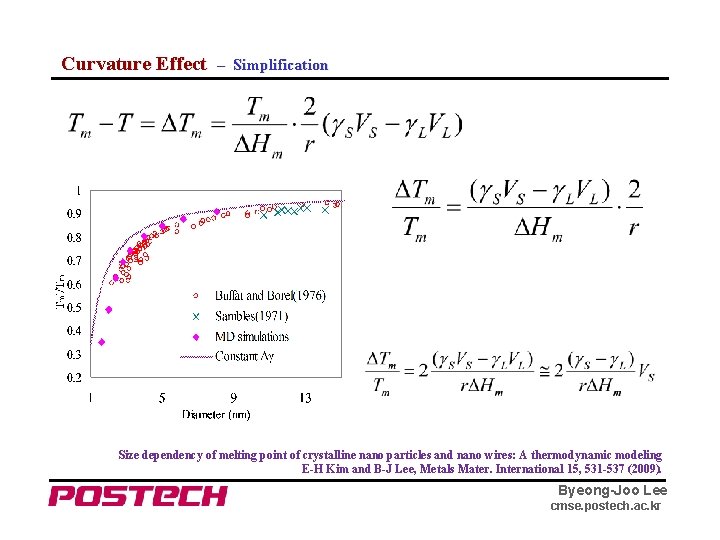
Curvature Effect – Simplification Size dependency of melting point of crystalline nano particles and nano wires: A thermodynamic modeling E-H Kim and B-J Lee, Metals Mater. International 15, 531 -537 (2009). Byeong-Joo Lee cmse. postech. ac. kr

Gibbs energy of ternary alloys Byeong-Joo Lee cmse. postech. ac. kr
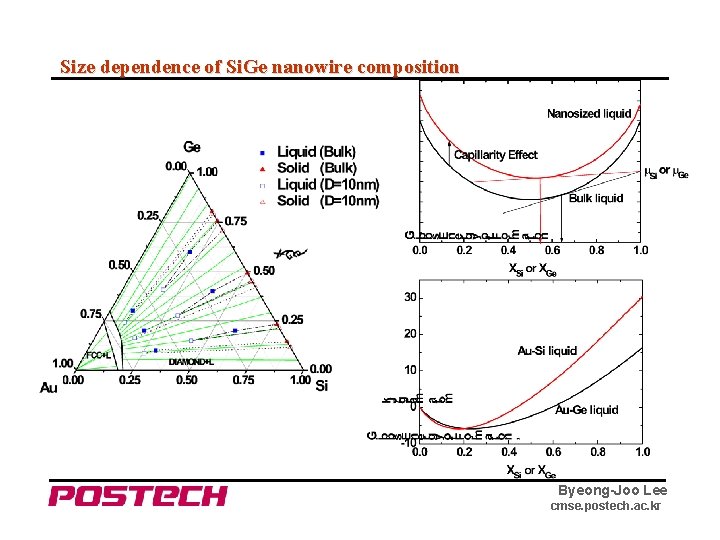
Size dependence of Si. Ge nanowire composition Byeong-Joo Lee cmse. postech. ac. kr
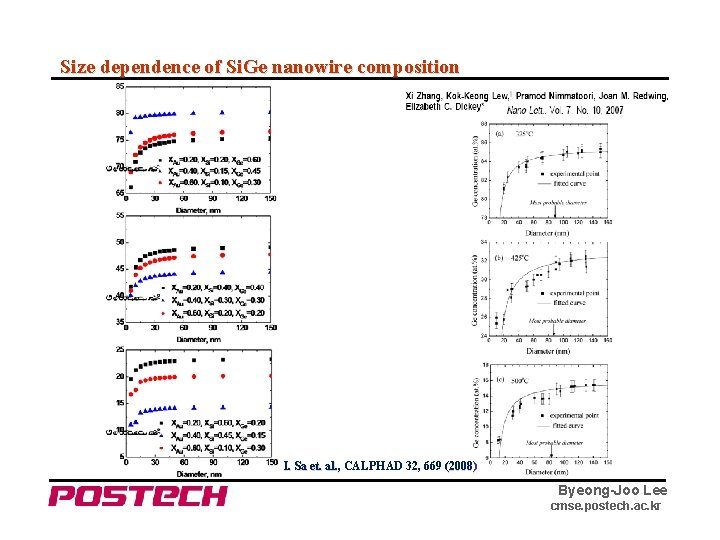
Size dependence of Si. Ge nanowire composition I. Sa et. al. , CALPHAD 32, 669 (2008) Byeong-Joo Lee cmse. postech. ac. kr
- Slides: 26