Phase Transformations Nucleation Kinetics ByeongJoo Lee POSTECH MSE

Phase Transformations Nucleation Kinetics Byeong-Joo Lee POSTECH - MSE calphad@postech. ac. kr Byeong-Joo Lee cmse. postech. ac. kr

Motivation Byeong-Joo Lee cmse. postech. ac. kr
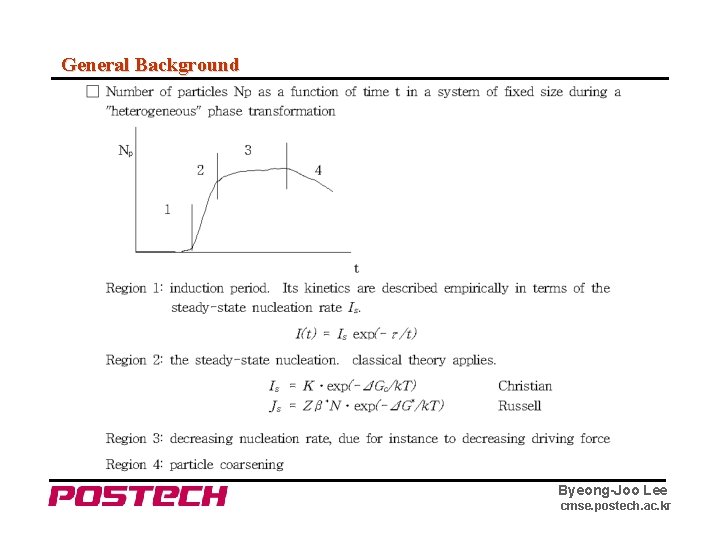
General Background Byeong-Joo Lee cmse. postech. ac. kr

Classical theory of nucleation References : 1. K. C. Russell, "Nucleation in Solids" in Phase Transformations, ASM 1970. 2. D. Turnbull, "Phase Changes" in Solid State Physics 3, 226, Academic Press, 1956. 3. J. W. Christian, Theory of Transformations in Metals and Alloys, Pergamon, 1965. □ Gibbs (1877) : activation energy ΔG*, critical nucleus size r*. ※ Understanding of the role of thermal fluctuation ← statistical thermodynamics Byeong-Joo Lee cmse. postech. ac. kr

Classical theory of nucleation □ Volmer and Weber (1925) : ▷ formation of larger particle by adding atoms to smaller particles ▷ van't Hoff's suggestion that the reaction goes in both directions p 1 ⇆ p 2 ⇆ p 3 ⇆ p 4 ⇆ p 5 etc. pi represent the particles of various sizes (number of atoms: i) @ equilibrium ni = n 1․exp (-ΔGi/k. T) ni : equil. number of particles of size i for the formation of water droplets in a supersaturated vapor calculation of the rates of individual reactions ← calculation of the number of water molecules in the vapor which hit a droplet per unit time using kinetic gas theory, neglecting reverse reaction the number of nuclei which grow above the critical size per unit time I = z. A*n* = z· 4π(r*)2·n 1·exp (-ΔG*/k. T) z : the collision frequency, according to the kinetic gas theory z = p/(2πmk. T)1/2 p : vapor pressure m : mass of molecules Byeong-Joo Lee cmse. postech. ac. kr

Classical theory of nucleation □ Becker and Döring (1935) : ▷ Improved the treatment considering accommodation factor and reverse reaction 0 < α < 1 accommodation factor β: correction for reverse reaction I = α βZA*n* ※ "No one can prove" H. Reiss, J. Chem. Phys. 20, 1216 (1952) Byeong-Joo Lee cmse. postech. ac. kr

Nucleation Rate in Solids Collision frequency : → rate by which an atom will jump across the phase interface : diffusion energy barrier term, exp (-Qdiff/k. T), should be appended. Russell : Js = Zβ*No* exp (-ΔGn*/k. T) : steady state nucleation rate Zeldovich factor No* : number of nucleation site ("per mole" or "per volume") J = Js exp (-τ/t) : time-dependent nucleation rate incubation (induction) time Byeong-Joo Lee cmse. postech. ac. kr

Nucleation Rate in Solids Christian : rate at which atom number of embryos will transfer to of critical size critical embryo per unit volume and make it grow : a jump (attempt) frequency ΔGa : activation energy for diffusion Nv : number of possible nucleation sites per unit volume ∵ "Observable rate" of 106/m 3․sec requires ΔGc ≲ 70 k. T Byeong-Joo Lee cmse. postech. ac. kr
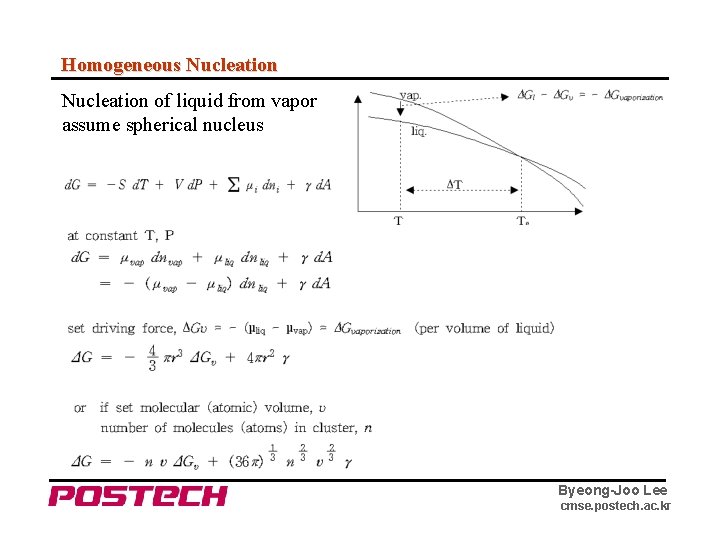
Homogeneous Nucleation of liquid from vapor assume spherical nucleus Byeong-Joo Lee cmse. postech. ac. kr

Homogeneous Nucleation Byeong-Joo Lee cmse. postech. ac. kr

Heterogeneous Nucleation of solid between liquid and solid mould ※ Physical meaning of f(θ) ※ Application of the concept of f(θ) to non-spherical nuclei ※ Heterogeneous nucleation in wall crack Byeong-Joo Lee cmse. postech. ac. kr

Nucleation in Solids Byeong-Joo Lee cmse. postech. ac. kr
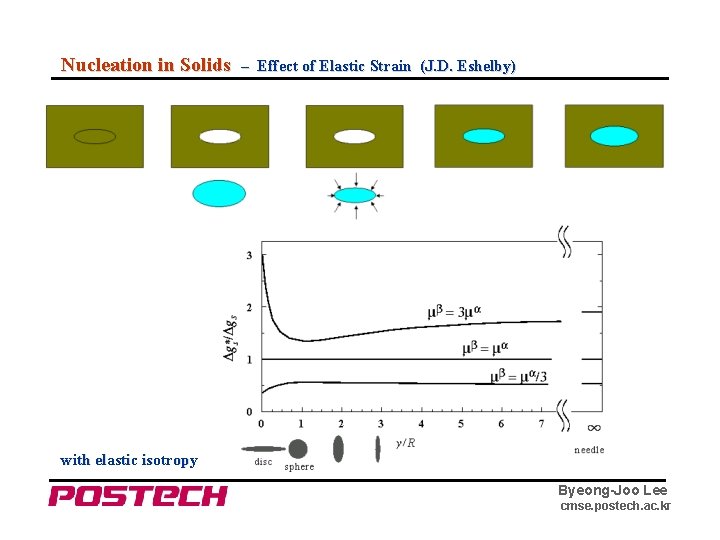
Nucleation in Solids – Effect of Elastic Strain (J. D. Eshelby) with elastic isotropy Byeong-Joo Lee cmse. postech. ac. kr
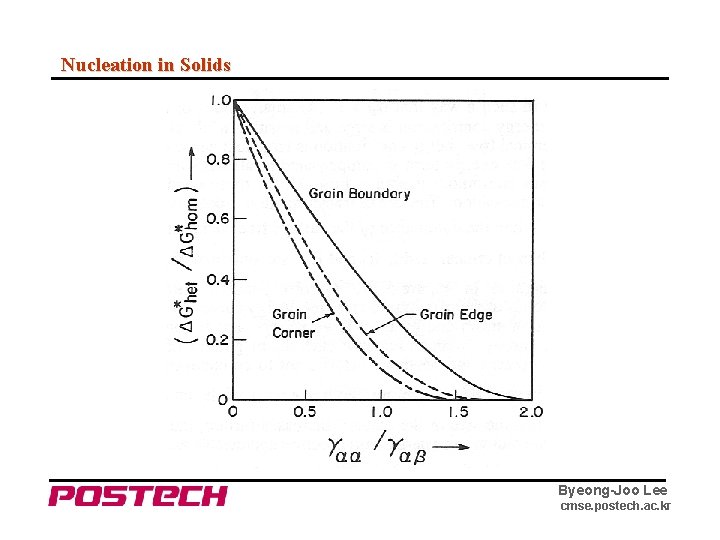
Nucleation in Solids Byeong-Joo Lee cmse. postech. ac. kr

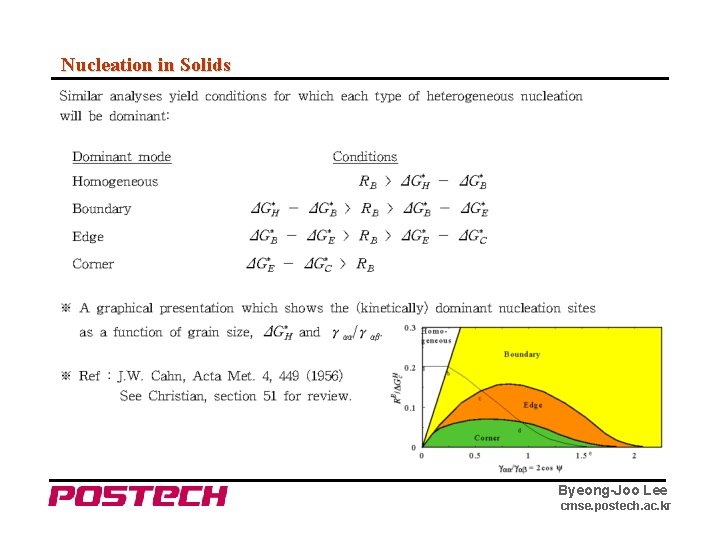
Nucleation in Solids ※ Similar relations for grain boundary edge and corner nucleation can be worked out. ⇒ for a given θ, ΔG* decreases as the "dimensionality" of the site decreases. (d = 0, 1, 2, 3 for C, E, B, H respectively) But although ΔG* decreases, the number of sites available for nucleation also decreases as dimensionality decreases. Set L : average grain diameter δ : grain boundary thickness Nv : number of atoms per unit volume ⇒ Nv. B = Nv (δ/L) # of boundary sites per volume Nv. E = Nv (δ/L)2 # of edge sites per volume Nv. C = Nv (δ/L)3 # of corner sites per volume Byeong-Joo Lee cmse. postech. ac. kr

Nucleation in Solids Substituting into general expression for I : Byeong-Joo Lee cmse. postech. ac. kr
Nucleation in Solids Byeong-Joo Lee cmse. postech. ac. kr

Nucleation in Solids - nucleation in dislocations (incoherent) Refs. : J. W. Cahn, Acta Met. 5, 169 (1957). - incoherent nucleation F. C. Larché, "Nucleation and Precipitation on Dislocations, " in Dislocations in Metallurgy, Chap. 14 of Vol. III of Dislocations in Solids, F. R. N. Nabarro (1979) - coherent nucleation Byeong-Joo Lee cmse. postech. ac. kr
- Slides: 18