Phase Transformation o o Course number 527 M

Phase Transformation o o Course number: 527 M 1700 Designation: Graduate course Instructor: Chao-Sung Lin, MSE Dept. , 33665240(office), 33662678 (lab) Office hours: 2 ~ 5 pm, Thursday

Textbook and suggested references o o o Phase Transformations in Materials [electronic resource available in NTU library], Gernot Kostorz ed. , Weinheim, New York Chichester, Wiley-VCH. (Strongly Recommended) Phase Transformations in Metals and Alloys, D. A. Porter and K. E. Easterling, 2 nd ed. , Nelson Thornes, UK (Strongly Recommended) Theory of Transformations in Metals and Alloys: Equilibrium and general Kinetic Theory, J. W. Christian, 2 nd ed. , Pergamon Press Ltd. Solidification Processing, M. C. Flemings, Mc. GRAW-HILL. Diffusion in Solids, P. G. Shewmon, Mc. GRAWHILL.

Recommended background o o o Materials Science and Engineering Physical Metallurgy Principles, R. E. Read-Hill Metallurgical Thermodynamics Materials Thermodynamics, D. R. Gaskell Chemical Thermodynamics of Materials, C. H. P. Lupis.

Topics o o o Thermodynamics and Phase diagrams (4) Solidification (3) Diffusion Kinetics (3) Second Phase Precipitation (4) Diffusionless Transformations (2) Grading o o Mid-Term (50%) Final (50%)

Thermodynamics and Phase diagrams Thermodynamic properties o o External influences n P, T, V (any two of them are required) Thermodynamics properties n U, internal energy n S, entropy n H = U + PV, enthalpy n G = H – TS, Gibbs free energy n A = U – TS, Hemholtz free energy o Useful parameters n cv, constant volume heat capacity n cp, constant pressure heat capacity

Gibbs free energy change Partial molar Gibbs energy of an ideal gas is the standard molar Gibbs energy (at 1 bar) pi is the partial pressure in bar Consider the reaction for the formation of 2 dn moles of H 2 S

Chemical equilibrium The reaction will proceed as DG < 0 When equilibrium is reached, DG = 0 Where K is the equilibrium constant Note: DG < 0 is a necessary condition for a reaction to occur, but is not a sufficient condition. - need to consider the rate of the reaction
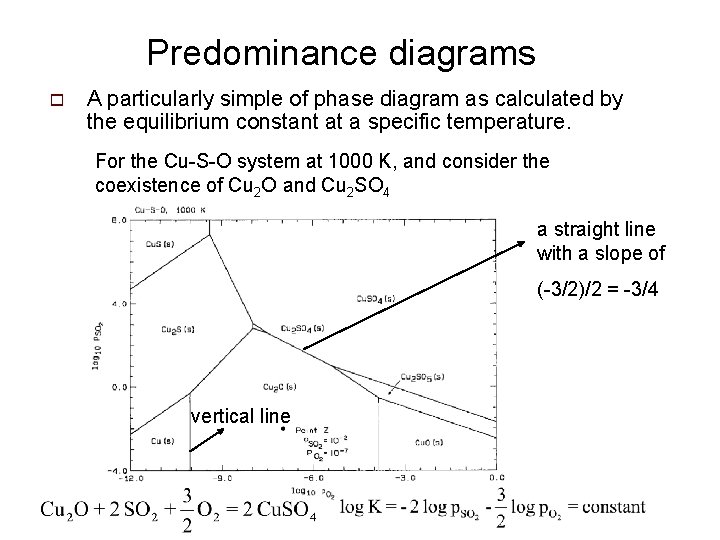
Predominance diagrams o A particularly simple of phase diagram as calculated by the equilibrium constant at a specific temperature. For the Cu-S-O system at 1000 K, and consider the coexistence of Cu 2 O and Cu 2 SO 4 a straight line with a slope of (-3/2)/2 = -3/4 vertical line

Procedure: 1. Choose Cu as the base element. 2. Consider the formation each solid compound from one mole of the base element, Cu. Say, for Cu. O 3. Obtain DG 0 from tables of thermodynamics properties 4. DG is calculated for any given p. SO 2 and p. O 2. 5. The one with the most negative DG is the stable compound.
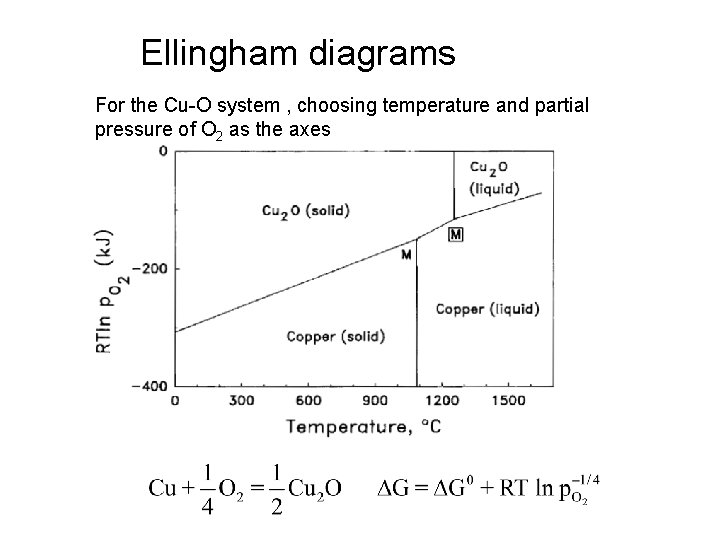
Ellingham diagrams For the Cu-O system , choosing temperature and partial pressure of O 2 as the axes
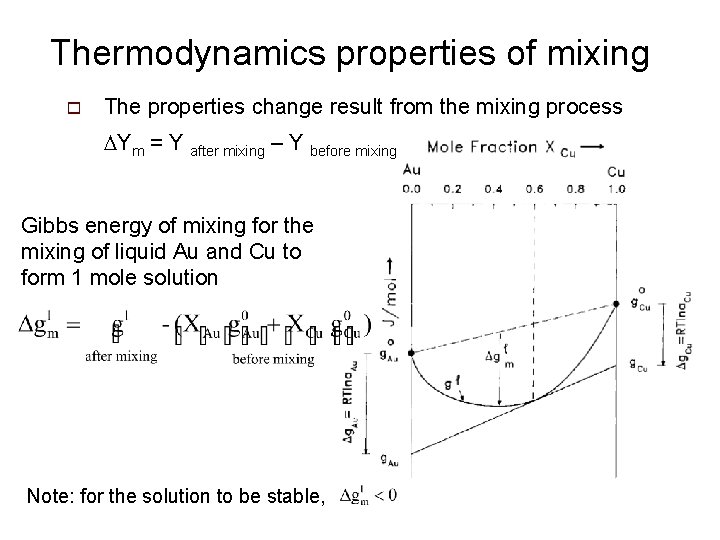
Thermodynamics properties of mixing o The properties change result from the mixing process DYm = Y after mixing – Y before mixing Gibbs energy of mixing for the mixing of liquid Au and Cu to form 1 mole solution Note: for the solution to be stable,

Chemical potential The partial molar quantity of a thermodynamics property o o The partial molar Gibbs free energy, also known as the chemical potential, is the rate of the Gibbs energy as component i is added. I II dn. Cu Note: 1. mi = mi (T, n) is an intensive function. 2. When two or more phases are in equilibrium, the chemical potential of any component is the same in all phase.
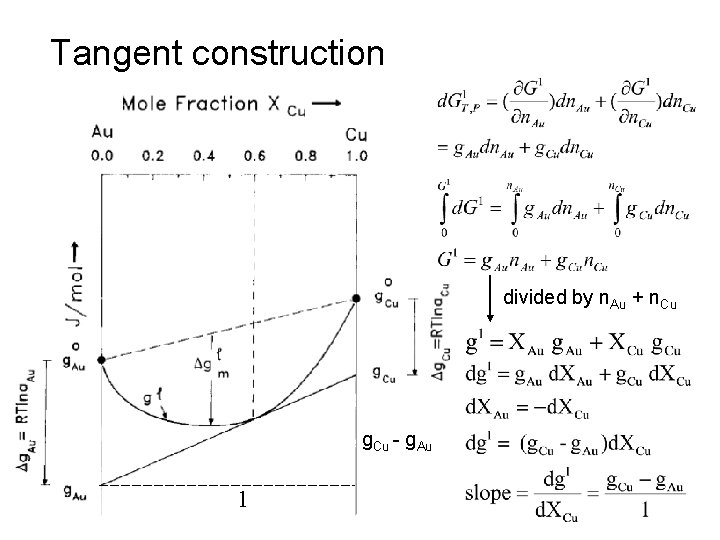
Tangent construction divided by n. Au + n. Cu g. Cu - g. Au 1

Gibbs-Duhem equation In general,

Relative partial properties o o The difference between the partial Gibbs energy of a component in solution and that in a stand state. The activity ai of the component relative to the chosen standard state is defined in terms of the relative partial Gibbs energy as For Au-Cu binary system where Dhm is the enthalpy of mixing Dsm is the entropy of mixing

Ideal Raoultian solution o o For ideal solution Need to consider configuration entropy only, Boltzmann’s equation Appling Stirling’s approximation

Excess properties o y. E = y - yideal

o An equation obtained using tangent constructions o The Gibbs-Duhem equation of excess properties Note: 1. Excess Gibbs energy markedly influences the form of the phase diagram. 2. If g. E < 0, the solution is thermodynamically more stable than an ideal solution. 3. g. E > 0 positive deviation, the same atoms dislike each other 4. g. E < 0 negative deviation, different atoms like each other o Activity coefficient
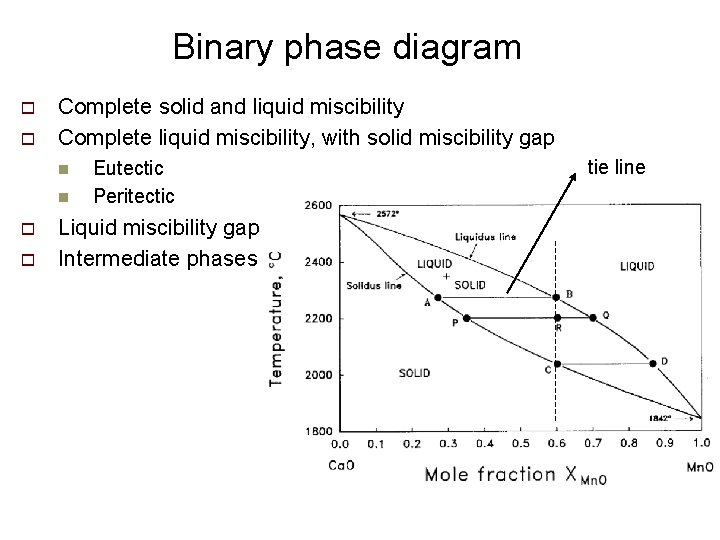
Binary phase diagram o o Complete solid and liquid miscibility Complete liquid miscibility, with solid miscibility gap n n o o Eutectic Peritectic Liquid miscibility gap Intermediate phases tie line
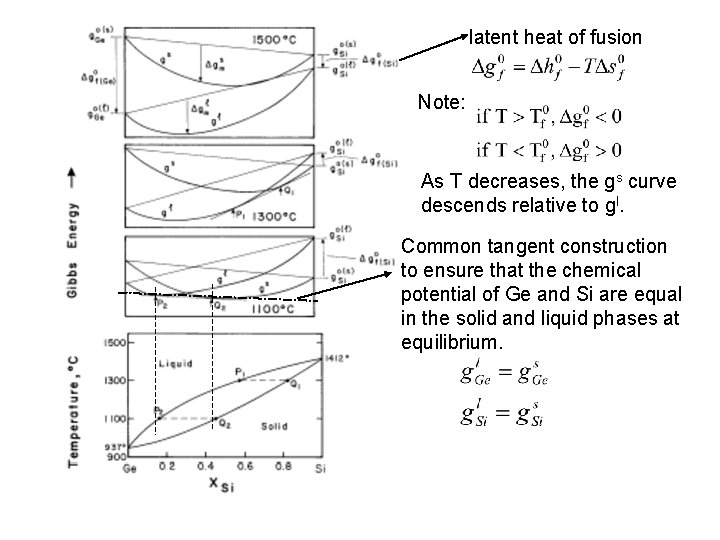
latent heat of fusion Note: As T decreases, the gs curve descends relative to gl. Common tangent construction to ensure that the chemical potential of Ge and Si are equal in the solid and liquid phases at equilibrium.
- Slides: 20