Phase III ADAURA Adjuvant Osimertinib vs Placebo After

Phase III ADAURA: Adjuvant Osimertinib vs Placebo After Complete Resection in Patients With Stage IBIIIA EGFR-Mutated NSCLC CCO Independent Conference Coverage* Highlights of the 2020 ASCO Virtual Scientific Meeting, May 29 -31, 2020 *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. This activity is provided by Clinical Care Options, LLC Supported by educational grants from Astra. Zeneca; Daiichi Sankyo, Inc. ; Ipsen Pharma; Jazz Pharmaceuticals, Inc. ; and Merck Sharp & Dohme Corp.
About These Slides § Please feel free to use, update, and share some or all of these slides in your noncommercial presentations to colleagues or patients § When using our slides, please retain the source attribution: Slide credit: clinicaloptions. com § These slides may not be published, posted online, or used in commercial presentations without permission. Please contact permissions@clinicaloptions. com for details

ADAURA: Background § ~ 30% of patients with NSCLC have resectable disease at diagnosis[1, 2] ‒ Standard of care for early-stage NSCLC consists of surgery followed by adjuvant cisplatin-based CT in patients with stage II-IIIA disease and in some patients with stage IB disease [3 -5] § Despite adjuvant CT improving OS and DFS in stage II-III NSCLC, recurrence rates remain high at ~ 30% to 70%[6] § Osimertinib: third-generation EGFR TKI approved by FDA for treatment of EGFR-mutated metastatic NCSLC in first-line setting and, if EGFR T 790 M+, in those with progression on/after EGFR TKI tx[7] ‒ Activity and safety profile in later-stage disease suggests potential efficacy in early-stage disease[8, 9] § Current unplanned interim analysis of phase III ADAURA trial compared adjuvant osimertinib vs placebo after complete tumor resection ± adjuvant CT in patients with stage IB-IIIA EGFR-mutated NSCLC[10] 1. Le Chevalier. Ann Oncol. 2010; 21(suppl 7): vii 196. 2. Cagle. Arch Pathol Lab Med. 2013; 137: 1191. 3. Postmus. Ann Oncol. 2017; 28(suppl 4): iv 1. 4. Kris. JCO. 2017; 35: 2960. 5. Chansky. JTO. 2017; 12: 1109. 6. Pignon. JCO. 2008; 26: 3552. 7. Osimertinib PI. 8. Shen. J Thorac Dis. 2018; 10: 1364. 9. Soria. NEJM. 2018; 378: 113. 10. Herbst. ASCO 2020. Abstr LBA 5. Slide credit: clinicaloptions. com

ADAURA: Study Design § International, randomized, double-blind phase III trial (data cutoff for interim analysis: 1/17/2020) ‒ IDMC recommended early unblinding due to efficacy; at time of unblinding, trial had completed enrollment and all patients had ≥ 1 yr of follow-up Patients with completely resected stage IB/II/IIIA NSCLC with negative margins; primary nonsquamous NSCLC with EGFR ex 19 del or L 858 R*; aged ≥ 18 yrs (≥ 20 yrs in Japan/Taiwan); WHO PS 0/1; brain imaging done; adj CT permitted; maximum time from surgery to randomization: 10 wks without adj CT, 26 wks with adj CT (N = 682) Herbst. ASCO 2020. Abstr LBA 5. § Primary endpoint: investigator-assessed DFS in patients with stage II/ IIIA disease Stratified by stage (IB vs IIIA), EGFR mutation (ex 19 del vs L 858 R), race (Asian vs non-Asian) Osimertinib 80 mg QD (n = 339) Placebo QD (n = 343) Until 3 yrs, recurrence, or d/c criterion met† *Confirmed centrally in tissue. †Follow-up: until recurrence, Wks 12 and 24, then Q 24 W to 5 yrs, then yearly; after recurrence, Q 24 W for 5 yrs, then yearly. ‒ Trial designed to test superiority with assumed DFS HR of 0. 70 § Secondary endpoints: DFS in overall population; landmark DFS rates at Yrs 2, 3, 4, and 5; OS; HRQo. L; safety Slide credit: clinicaloptions. com

ADAURA: Baseline Characteristics Characteristic, % Osimertinib (n = 339) Placebo (n = 343) 68 72 64 (30 -86) 62 (31 -82) Smoker/nonsmoker* 32/68 25/75 Asian/non-Asian 64/36 WHO PS 0/1 64/36 31 35 34 31 34 35 Adenocarcinoma/other histology† 95/5 96/4 EGFR ex 19 del/L 858 R 55/45 56/44 55 56 Female Median age, yrs (range) AJCC staging at diagnosis (7 th edition) § IB § IIIA Prior adjuvant CT *Former smoker: osimertinib, n = 104; placebo, n = 83; current smoker: osimertinib, n = 4; placebo, n = 3; never smoker: osimertinib, n = 231; placebo, n = 257. †Includes bronchial gland carcinoma (NOS): osimertinib, n = 1; placebo, n = 2; malignant adenosquamous carcinoma: osimertinib, n = 4; placebo, n = 5; other: osimertinib, n = 11; placebo, n = 7. †Centrally assessed at randomization. Slide credit: clinicaloptions. com Herbst. ASCO 2020. Abstr LBA 5.
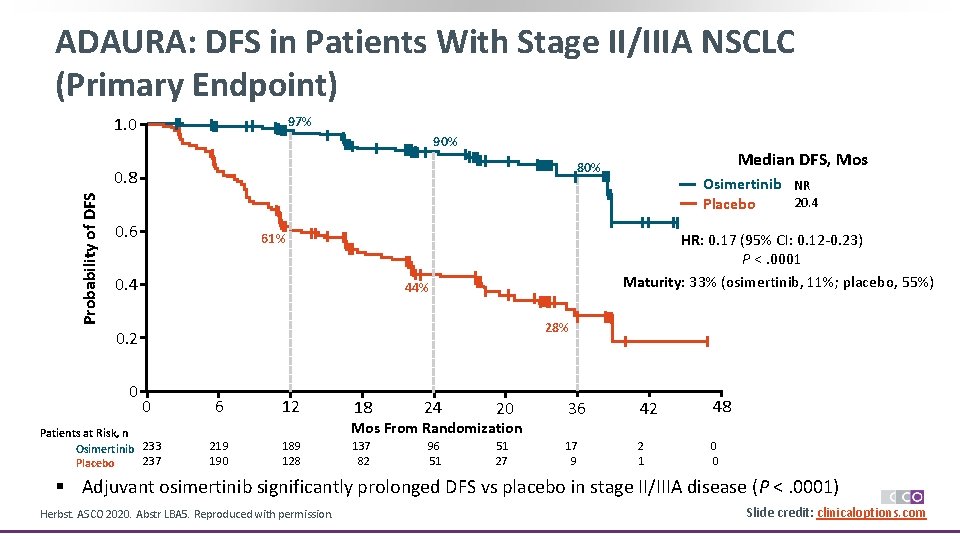
ADAURA: DFS in Patients With Stage II/IIIA NSCLC (Primary Endpoint) 1. 0 97% 90% Probability of DFS 0. 8 0. 6 Osimertinib NR 20. 4 Placebo HR: 0. 17 (95% CI: 0. 12 -0. 23) P <. 0001 Maturity: 33% (osimertinib, 11%; placebo, 55%) 61% 0. 4 44% 28% 0. 2 0 Median DFS, Mos 80% 0 Patients at Risk, n Osimertinib 233 237 Placebo 6 12 18 24 20 36 42 48 137 82 96 51 51 27 17 9 2 1 0 0 Mos From Randomization 219 190 189 128 § Adjuvant osimertinib significantly prolonged DFS vs placebo in stage II/IIIA disease (P <. 0001) Herbst. ASCO 2020. Abstr LBA 5. Reproduced with permission. Slide credit: clinicaloptions. com
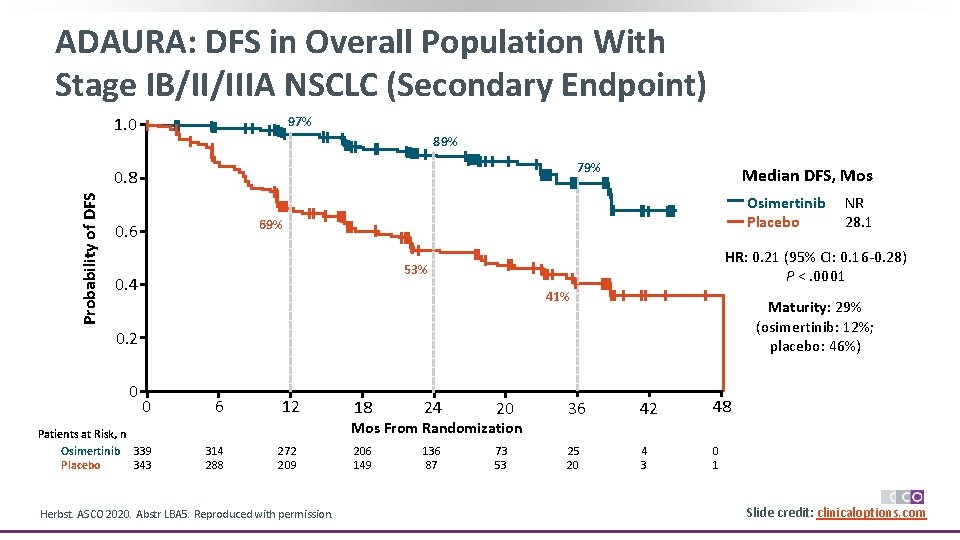
ADAURA: DFS in Overall Population With Stage IB/II/IIIA NSCLC (Secondary Endpoint) 1. 0 97% 89% 79% Probability of DFS 0. 8 Median DFS, Mos Osimertinib Placebo 69% 0. 6 HR: 0. 21 (95% CI: 0. 16 -0. 28) P <. 0001 53% 0. 4 41% Maturity: 29% (osimertinib: 12%; placebo: 46%) 0. 2 0 0 Patients at Risk, n Osimertinib 339 343 Placebo 6 12 18 24 20 36 42 48 206 149 136 87 73 53 25 20 4 3 0 1 Mos From Randomization 314 288 272 209 Herbst. ASCO 2020. Abstr LBA 5. Reproduced with permission. NR 28. 1 Slide credit: clinicaloptions. com
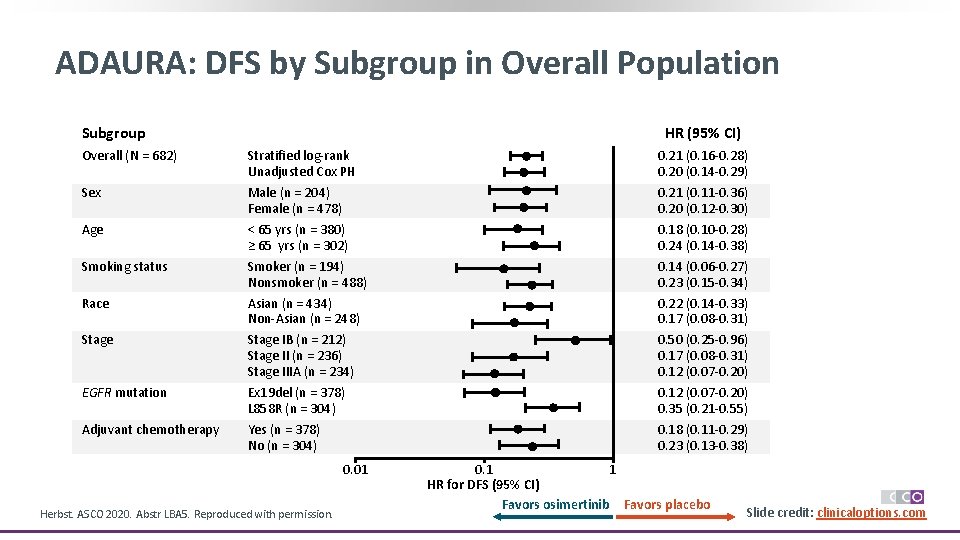
ADAURA: DFS by Subgroup in Overall Population HR (95% CI) Subgroup Overall (N = 682) Sex Age Smoking status Race Stage EGFR mutation Adjuvant chemotherapy Stratified log-rank Unadjusted Cox PH Male (n = 204) Female (n = 478) < 65 yrs (n = 380) ≥ 65 yrs (n = 302) Smoker (n = 194) Nonsmoker (n = 488) Asian (n = 434) Non-Asian (n = 248) Stage IB (n = 212) Stage II (n = 236) Stage IIIA (n = 234) Ex 19 del (n = 378) L 858 R (n = 304) Yes (n = 378) No (n = 304) 0. 01 Herbst. ASCO 2020. Abstr LBA 5. Reproduced with permission. 0. 21 (0. 16 -0. 28) 0. 20 (0. 14 -0. 29) 0. 21 (0. 11 -0. 36) 0. 20 (0. 12 -0. 30) 0. 18 (0. 10 -0. 28) 0. 24 (0. 14 -0. 38) 0. 14 (0. 06 -0. 27) 0. 23 (0. 15 -0. 34) 0. 22 (0. 14 -0. 33) 0. 17 (0. 08 -0. 31) 0. 50 (0. 25 -0. 96) 0. 17 (0. 08 -0. 31) 0. 12 (0. 07 -0. 20) 0. 35 (0. 21 -0. 55) 0. 18 (0. 11 -0. 29) 0. 23 (0. 13 -0. 38) 0. 1 1 HR for DFS (95% CI) Favors osimertinib Favors placebo Slide credit: clinicaloptions. com
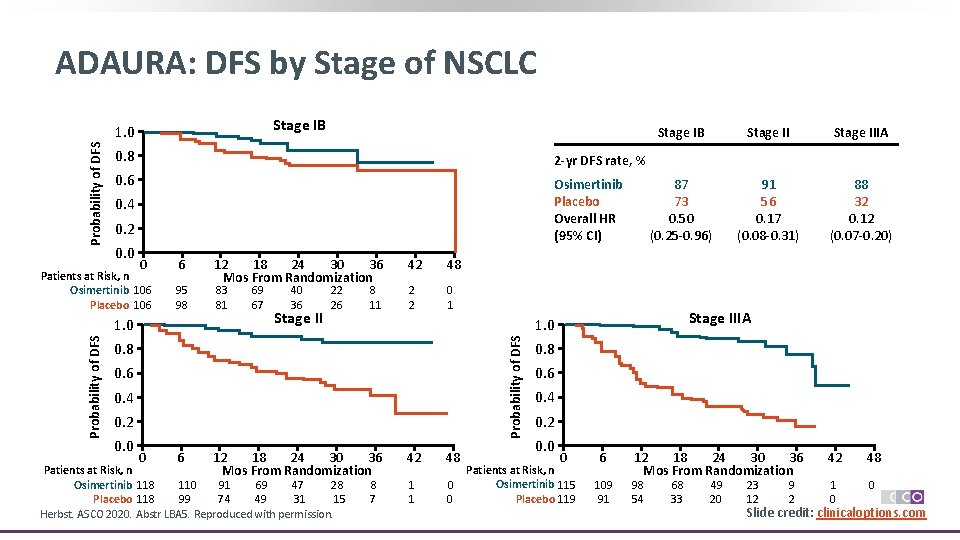
ADAURA: DFS by Stage of NSCLC Stage IB Probability of DFS 1. 0 0. 8 2 -yr DFS rate, % 0. 6 Osimertinib Placebo Overall HR (95% CI) 0. 4 0. 2 0. 0 0 Patients at Risk, n Osimertinib 106 Placebo 106 6 95 98 83 81 69 67 40 36 Stage II 22 26 8 11 42 48 2 2 0 1 0. 8 0. 6 0. 4 0. 2 0. 0 0 6 12 18 24 30 36 Mos From Randomization Patients at Risk, n Osimertinib 118 110 91 69 47 28 Placebo 118 99 74 49 31 15 Herbst. ASCO 2020. Abstr LBA 5. Reproduced with permission. Stage IIIA 87 73 0. 50 (0. 25 -0. 96) 91 56 0. 17 (0. 08 -0. 31) 88 32 0. 12 (0. 07 -0. 20) Stage IIIA 1. 0 Probability of DFS 1. 0 12 18 24 30 36 Mos From Randomization Stage IB 8 7 42 48 1 1 0 0 0. 8 0. 6 0. 4 0. 2 0. 0 0 Patients at Risk, n Osimertinib 115 Placebo 119 6 109 91 12 18 24 30 36 Mos From Randomization 98 54 68 33 49 20 23 12 9 2 42 48 1 0 0 Slide credit: clinicaloptions. com
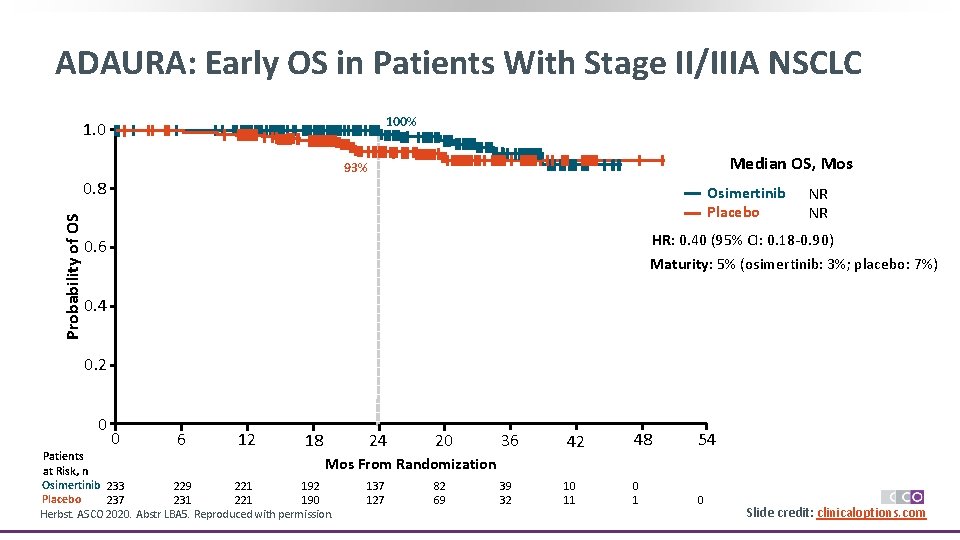
ADAURA: Early OS in Patients With Stage II/IIIA NSCLC 100% 1. 0 Median OS, Mos 93% Probability of OS 0. 8 Osimertinib Placebo NR NR HR: 0. 40 (95% CI: 0. 18 -0. 90) 0. 6 Maturity: 5% (osimertinib: 3%; placebo: 7%) 0. 4 0. 2 0 0 6 12 18 Patients Mos at Risk, n Osimertinib 233 229 221 192 Placebo 237 231 221 190 Herbst. ASCO 2020. Abstr LBA 5. Reproduced with permission. 24 20 36 42 48 54 39 32 10 11 0 From Randomization 137 127 82 69 Slide credit: clinicaloptions. com
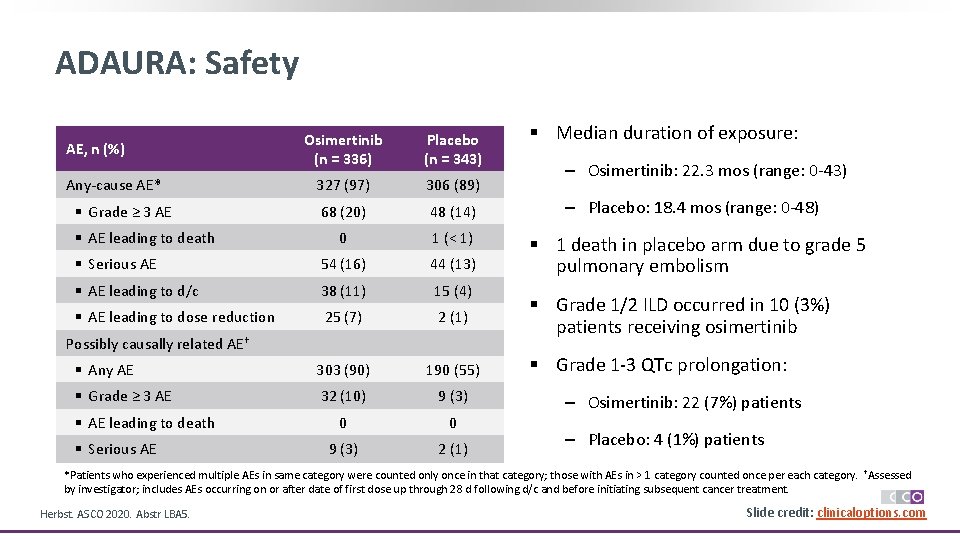
ADAURA: Safety Osimertinib (n = 336) Placebo (n = 343) 327 (97) 306 (89) 68 (20) 48 (14) 0 1 (< 1) § Serious AE 54 (16) 44 (13) § AE leading to d/c 38 (11) 15 (4) § AE leading to dose reduction 25 (7) 2 (1) § Any AE 303 (90) 190 (55) § Grade ≥ 3 AE 32 (10) 9 (3) 0 0 9 (3) 2 (1) AE, n (%) Any-cause AE* § Grade ≥ 3 AE § AE leading to death Possibly causally related AE† § AE leading to death § Serious AE § Median duration of exposure: ‒ Osimertinib: 22. 3 mos (range: 0 -43) ‒ Placebo: 18. 4 mos (range: 0 -48) § 1 death in placebo arm due to grade 5 pulmonary embolism § Grade 1/2 ILD occurred in 10 (3%) patients receiving osimertinib § Grade 1 -3 QTc prolongation: ‒ Osimertinib: 22 (7%) patients ‒ Placebo: 4 (1%) patients *Patients who experienced multiple AEs in same category were counted only once in that category; those with AEs in > 1 category counted once per each category. †Assessed by investigator; includes AEs occurring on or after date of first dose up through 28 d following d/c and before initiating subsequent cancer treatment. Herbst. ASCO 2020. Abstr LBA 5. Slide credit: clinicaloptions. com
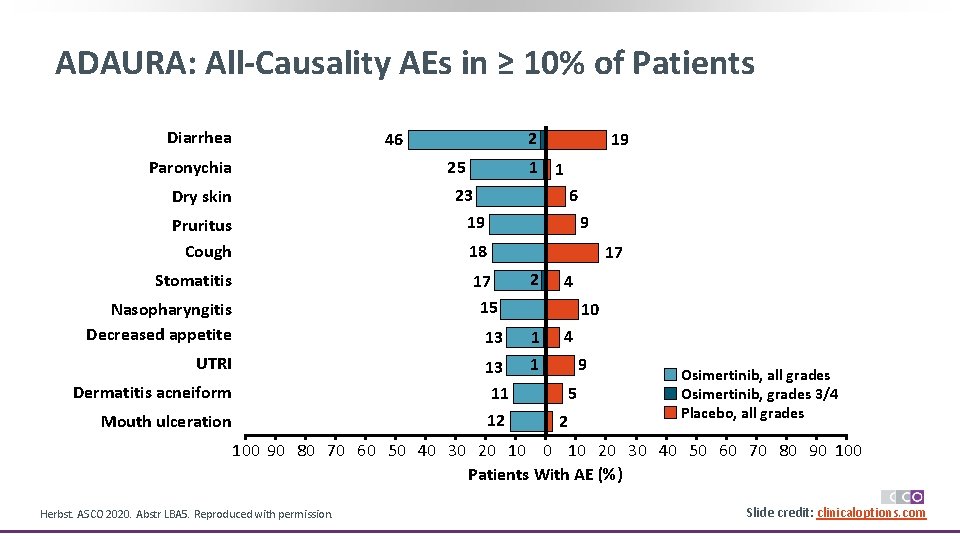
ADAURA: All-Causality AEs in ≥ 10% of Patients Diarrhea 2 46 Paronychia 25 19 1 1 6 23 19 Dry skin Pruritus Cough 9 18 Stomatitis 17 15 Nasopharyngitis Decreased appetite 13 UTRI 13 11 12 Dermatitis acneiform Mouth ulceration 17 2 4 10 1 1 4 9 5 2 Osimertinib, all grades Osimertinib, grades 3/4 Placebo, all grades 100 90 80 70 60 50 40 30 20 10 20 30 40 50 60 70 80 90 100 Patients With AE (%) Herbst. ASCO 2020. Abstr LBA 5. Reproduced with permission. Slide credit: clinicaloptions. com

ADAURA: Conclusions § In unplanned interim analysis, adjuvant osimertinib significantly prolonged DFS vs placebo after complete resection in patients with stage IB/II/IIIA EGFR+ NSCLC ‒ 83% reduction in risk of recurrence or death with osimertinib in stage II/IIIA disease (primary endpoint; HR: 0. 17; P <. 0001) ‒ 79% reduction in the risk of recurrence or death with osimertinib in the overall population (HR: 0. 21; P <. 0001) ‒ DFS prolonged with osimertinib across subgroups, including those who received prior adjuvant chemotherapy § No new safety signals observed with osimertinib § Investigators concluded that adjuvant osimertinib should be incorporated into standard practice for treating patients with stage IB/II/IIIA EGFR-mutated NSCLC following complete resection Herbst. ASCO 2020. Abstr LBA 5. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASCO 2020! Short slideset summaries and additional CME-certified analyses with expert faculty commentary on key studies in: § Breast cancer § Gynecologic cancers § Gastrointestinal cancers § Hematologic malignancies § Genitourinary cancers § Lung cancer clinicaloptions. com/oncology
- Slides: 14