Phase Ib Study Results Subgroup Efficacy Analysis of

Phase Ib Study Results: Subgroup Efficacy Analysis of Atezolizumab + Bevacizumab in Patients With Previously Untreated, Unresectable Hepatocellular Carcinoma Kyung-Hun Lee, 1 Baek-Yeol Ryoo, 2 Chih-Hung Hsu, 3 Kazushi Numata, 4 Stacey Stein, 5 Wendy Verret, 6 Steve Hack, 6 Jessica Spahn, 6 Bo Liu, 6 Heba Abdullah, 6 Aiwu Ruth He, 7 Michael S Lee 8 1 Seoul National University Hospital, Seoul, South Korea; 2 Asan Medical Center, University of Ulsan College of Medicine, Seoul, South Korea; 3 National Taiwan University Hospital, Taipei, Taiwan; 4 Yokohama City University Medical Center, Yokohama, Japan; 5 Yale School of Medicine, New Haven, CT, USA; 6 Genentech, Inc. , South San Francisco, CA, USA; 7 Georgetown University Medical Center, Washington, DC, USA; 8 Lineberger Comprehensive Cancer Center, University of North Carolina at Chapel Hill, NC, USA 13 th Annual Conference 13 th 20 ► 22 September 2019 Chicago, USA ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA https: //bit. ly/2 ZCXq. Hc 1 Lee KH. Atezo + Bev in HCC

Disclosures • Advisory board: Bayer, Ono Pharmaceutical, Samsung Bioepis, Roche, Eisai, Astra. Zeneca • Honoraria: Roche, Astra. Zeneca • This study is sponsored by F. Hoffmann-La Roche, Ltd 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA https: //bit. ly/2 ZCXq. Hc 2 Lee KH. Atezo + Bev in HCC

Background • Approximately 80% of patients with HCC present with unresectable cancer, 1 usually complicated by underlying liver disease 1, 2 • VEGFR tyrosine kinase inhibitors are the first-line systemic standard of care for unresectable or metastatic HCC 2, 3 – Associated with modest survival benefits and considerable toxicities 2, 3 – Unconfirmed ORRs per RECIST 1. 1 were 19% with lenvatinib and 7% with sorafenib 3 • Single-agent PD-L 1/PD-1 immune checkpoint inhibitors have shown clinical activity against HCC, but not yet superiority over standards of care, in randomised studies 4, 5 • Many HCC tumors are hypervascularised and overexpress VEGF and PD-L 16, 7 – The anti-VEGF monoclonal antibody bevacizumab showed modest single-agent activity in HCC 8 – In addition to anti-angiogenic activity, bevacizumab’s immunomodulatory effects alter the tumour microenvironment and may boost anti–PD-L 1 -mediated anti-tumour response 9 -11 – Dual blockade of PD-L 1 and VEGF has shown clinical benefit in other tumour types 12 -14 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA 1. Lau WY, et al. An Surg. 2001; 2. Villanueva A, et al. NEJM. 2019; 3. Kudo M, et al. Lancet. 2018; 4. Finn RS, et al. JCO. 2019 (abstract 4004); 5. BMS press release. https: //www. clinicaltrialsarena. com/news/bms-checkmate-459 -trial/. 2019; 6. Morse MA, et al. CCR. 2019; 7. Gao Q, et al. CCR. 2009; 8. Wattenberg MM, et al. Cancer Med. 2019; 9. Motz GT, et al. Nat Med. 2014; 10. Roland CL, et al. Mol Can Ther. 2009; 11. Voron T, et al. J Exp Med. 2015; 12. Reck M, et al. Lancet Respir Med. 2019; 13. Wallin JJ, et al. Nat Commun. 2016; 14. Mc. Dermott DF, et al. JCO 2016. https: //bit. ly/2 ZCXq. Hc 3 Lee KH. Atezo + Bev in HCC
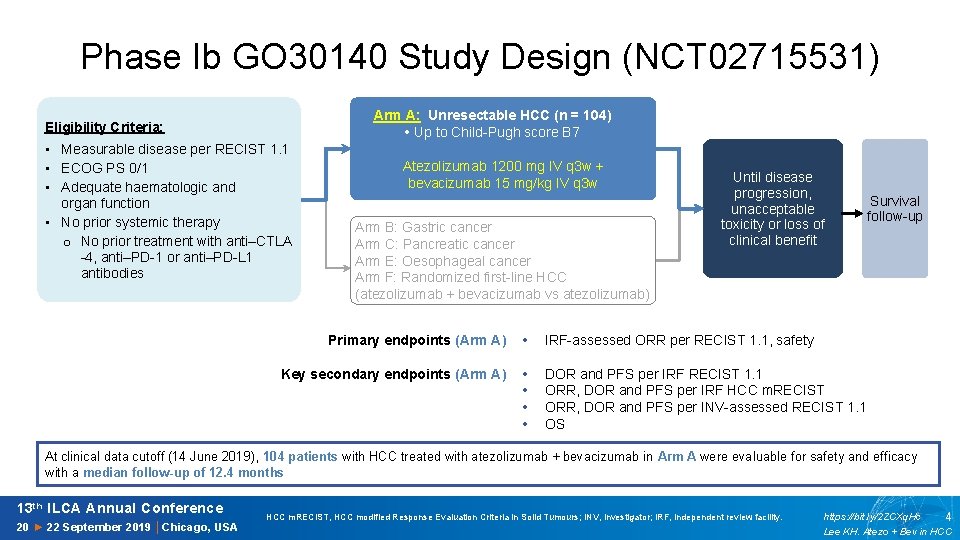
Phase Ib GO 30140 Study Design (NCT 02715531) Arm A: Unresectable HCC (n = 104) • Up to Child-Pugh score B 7 Eligibility Criteria: • Measurable disease per RECIST 1. 1 • ECOG PS 0/1 • Adequate haematologic and organ function • No prior systemic therapy o No prior treatment with anti–CTLA -4, anti–PD-1 or anti–PD-L 1 antibodies Atezolizumab 1200 mg IV q 3 w + bevacizumab 15 mg/kg IV q 3 w Arm B: Gastric cancer Arm C: Pancreatic cancer Arm E: Oesophageal cancer Arm F: Randomized first-line HCC (atezolizumab + bevacizumab vs atezolizumab) Until disease progression, unacceptable toxicity or loss of clinical benefit Primary endpoints (Arm A) • IRF-assessed ORR per RECIST 1. 1, safety Key secondary endpoints (Arm A) • • DOR and PFS per IRF RECIST 1. 1 ORR, DOR and PFS per IRF HCC m. RECIST ORR, DOR and PFS per INV-assessed RECIST 1. 1 OS Survival follow-up At clinical data cutoff (14 June 2019), 104 patients with HCC treated with atezolizumab + bevacizumab in Arm A were evaluable for safety and efficacy with a median follow-up of 12. 4 months 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA HCC m. RECIST, HCC modified Response Evaluation Criteria in Solid Tumours; INV, investigator; IRF, independent review facility. https: //bit. ly/2 ZCXq. Hc 4 Lee KH. Atezo + Bev in HCC
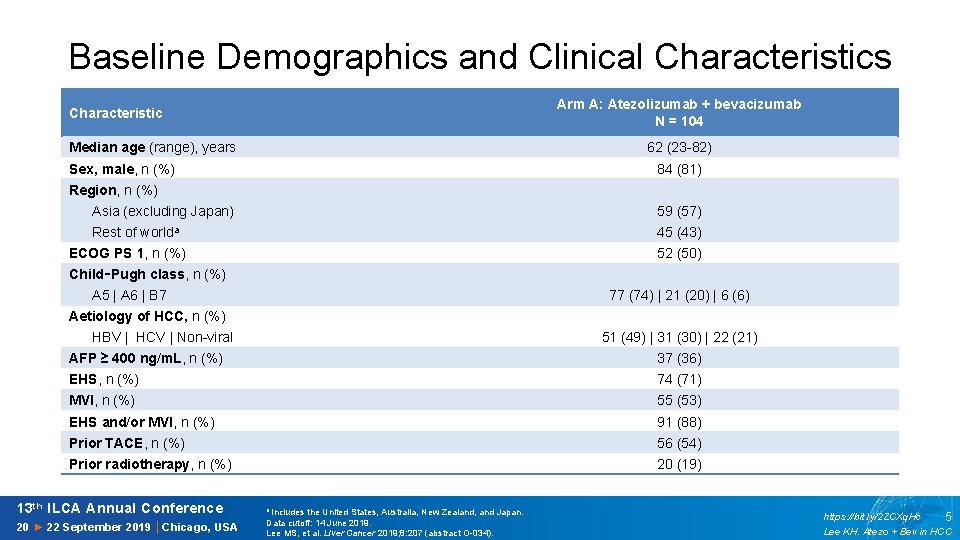
Baseline Demographics and Clinical Characteristics Arm A: Atezolizumab + bevacizumab N = 104 Characteristic Median age (range), years 62 (23 -82) Sex, male, n (%) 84 (81) Region, n (%) Asia (excluding Japan) 59 (57) Rest of worlda 45 (43) ECOG PS 1, n (%) 52 (50) Child‑Pugh class, n (%) A 5 | A 6 | B 7 77 (74) | 21 (20) | 6 (6) Aetiology of HCC, n (%) HBV | HCV | Non-viral 51 (49) | 31 (30) | 22 (21) AFP ≥ 400 ng/m. L, n (%) 37 (36) EHS, n (%) 74 (71) MVI, n (%) 55 (53) EHS and/or MVI, n (%) 91 (88) Prior TACE, n (%) 56 (54) Prior radiotherapy, n (%) 20 (19) 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA a Includes the United States, Australia, New Zealand, and Japan. Data cutoff: 14 June 2019. Lee MS, et al. Liver Cancer 2019; 8: 207 (abstract O-034). https: //bit. ly/2 ZCXq. Hc 5 Lee KH. Atezo + Bev in HCC
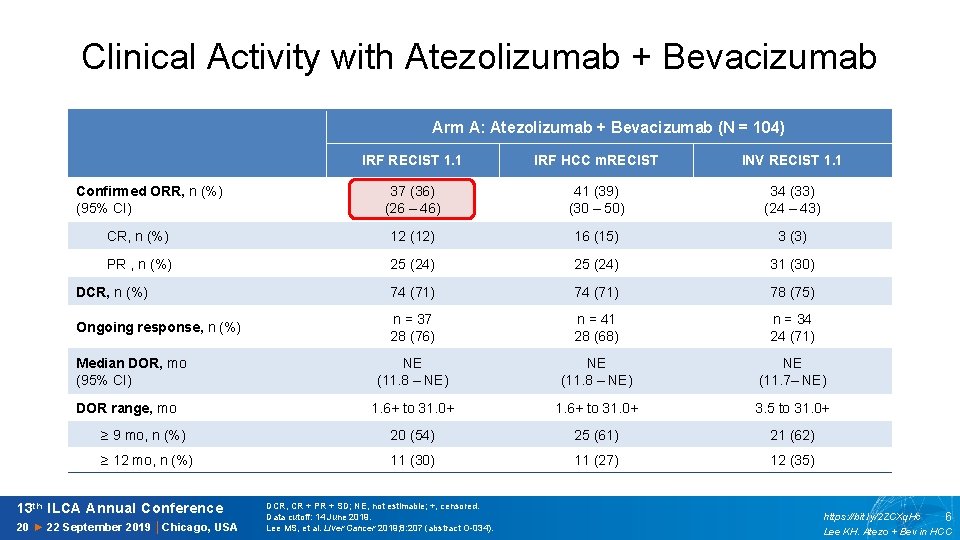
Clinical Activity with Atezolizumab + Bevacizumab Arm A: Atezolizumab + Bevacizumab (N = 104) IRF RECIST 1. 1 IRF HCC m. RECIST INV RECIST 1. 1 37 (36) (26 – 46) 41 (39) (30 – 50) 34 (33) (24 – 43) CR, n (%) 12 (12) 16 (15) 3 (3) PR , n (%) 25 (24) 31 (30) DCR, n (%) 74 (71) 78 (75) Ongoing response, n (%) n = 37 28 (76) n = 41 28 (68) n = 34 24 (71) NE (11. 8 – NE) NE (11. 7– NE) DOR range, mo 1. 6+ to 31. 0+ 3. 5 to 31. 0+ ≥ 9 mo, n (%) 20 (54) 25 (61) 21 (62) ≥ 12 mo, n (%) 11 (30) 11 (27) 12 (35) Confirmed ORR, n (%) (95% CI) Median DOR, mo (95% CI) 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA DCR, CR + PR + SD; NE, not estimable; +, censored. Data cutoff: 14 June 2019. Lee MS, et al. Liver Cancer 2019; 8: 207 (abstract O-034). https: //bit. ly/2 ZCXq. Hc 6 Lee KH. Atezo + Bev in HCC

Progression Free Survival and Overall Survival Summary Arm A: Atezolizumab + Bevacizumab (N = 104) Progression Free Survival IRF RECIST 1. 1 IRF HCC m. RECIST INV RECIST 1. 1 69 (66) 75 (72) 7. 3 (5. 4 – 9. 9) 7. 4 (5. 6 – 10. 7) 6 -month PFS rate (%) 54 55 56 12 -month PFS rate (%) 35 35 38 No. of events (%) Median PFS (95% CI), mo Overall Survival No. of deaths (%) 47 (45) Median OS (95% CI), mo 17. 1 (13. 8 – NE) 6 -month OS rate (%) 82 12 -month OS rate (%) 63 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA Data cutoff: 14 June 2019. https: //bit. ly/2 ZCXq. Hc 7 Lee KH. Atezo + Bev in HCC
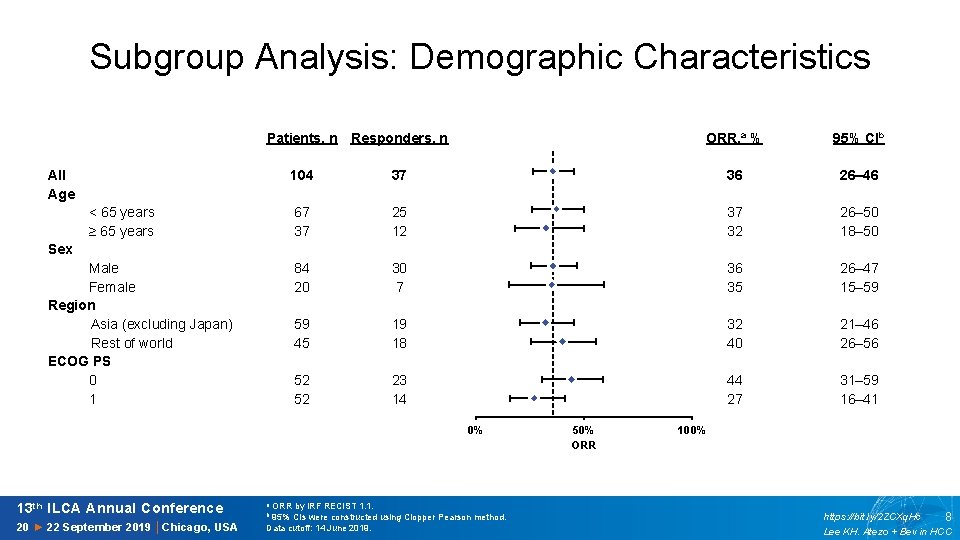
Subgroup Analysis: Demographic Characteristics Patients, n Responders, n ORR, a % 95% CIb 36 26– 46 37 32 26– 50 18– 50 36 35 26– 47 15– 59 32 40 21– 46 26– 56 44 27 31– 59 16– 41 14 All Age 104 13 37 12 < 65 years ≥ 65 years 67 37 11 25 12 10 9 Sex Male Female Region Asia (excluding Japan) Rest of world ECOG PS 0 1 84 20 8 30 7 7 6 59 45 5 19 18 4 3 52 52 23 14 2 1 0 0% 13 th ILCA Annual Conference a ORR by IRF RECIST 1. 1. 20 ► 22 September 2019 │Chicago, USA Data cutoff: 14 June 2019. b 95% CIs were constructed using Clopper Pearson method. 50% ORR 100% https: //bit. ly/2 ZCXq. Hc 8 Lee KH. Atezo + Bev in HCC
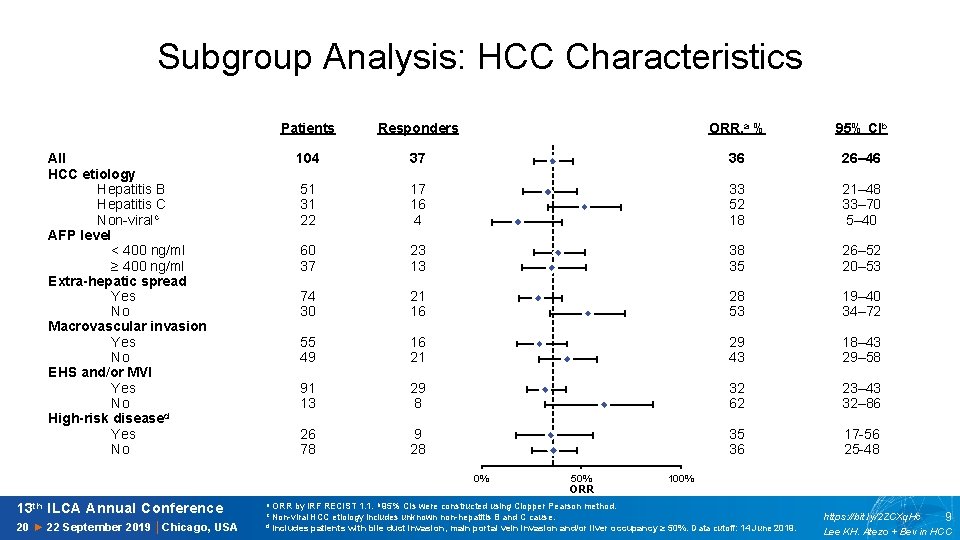
Subgroup Analysis: HCC Characteristics All HCC etiology Hepatitis B Hepatitis C Non-viralc AFP level < 400 ng/ml ≥ 400 ng/ml Extra-hepatic spread Yes No Macrovascular invasion Yes No EHS and/or MVI Yes No High-risk diseased Yes No Patients Responders 104 37 51 31 22 17 16 4 60 37 23 13 74 30 21 16 55 49 16 21 91 13 29 8 26 78 9 28 21 20 19 18 17 16 15 14 13 12 11 10 9 8 7 6 5 4 3 2 1 0 0% 50% ORR 13 th ILCA Annual Conference a ORR by IRF RECIST 1. 1. b 95% CIs were constructed using Clopper Pearson method. 20 ► 22 September 2019 │Chicago, USA d ORR, a % 95% CIb 36 26– 46 33 52 18 21– 48 33– 70 5– 40 38 35 26– 52 20– 53 28 53 19– 40 34– 72 29 43 18– 43 29– 58 32 62 23– 43 32– 86 35 36 17 -56 25 -48 100% c Non-viral HCC etiology includes unknown non-hepatitis B and C cause. Includes patients with bile duct invasion, main portal vein invasion and/or liver occupancy ≥ 50%. Data cutoff: 14 June 2019. https: //bit. ly/2 ZCXq. Hc 9 Lee KH. Atezo + Bev in HCC
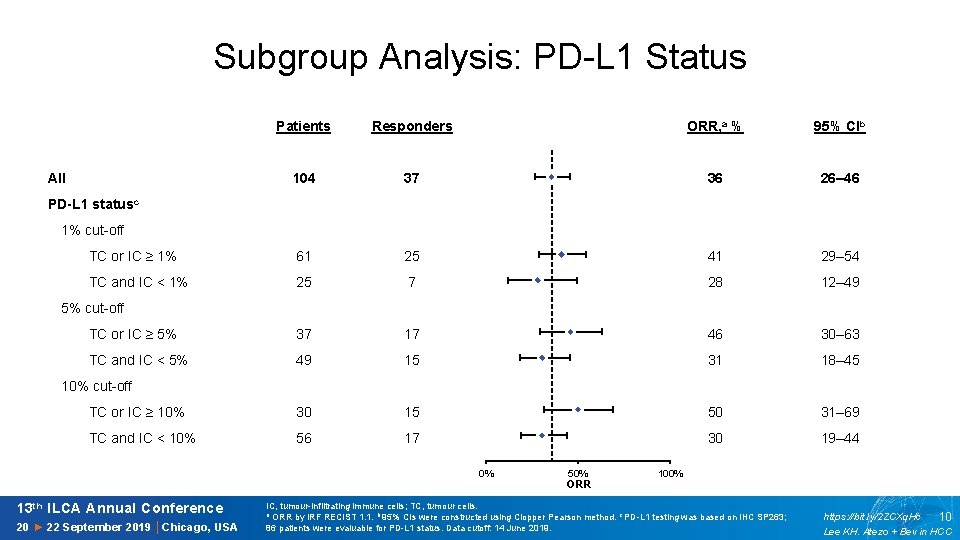
Subgroup Analysis: PD-L 1 Status Patients Responders ORR, a % 95% CIb 36 26– 46 12 All 104 11 37 PD-L 1 statusc 10 1% cut-off 9 TC or IC ≥ 1% 61 25 8 41 29– 54 TC and IC < 1% 25 7 7 28 12– 49 6 5% cut-off TC or IC ≥ 5% 37 17 5 46 30– 63 TC and IC < 5% 49 15 4 31 18– 45 3 10% cut-off TC or IC ≥ 10% 30 15 2 50 31– 69 TC and IC < 10% 56 17 1 30 19– 44 0% 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA 0 50% ORR 100% IC, tumour-infiltrating immune cells; TC, tumour cells. a ORR by IRF RECIST 1. 1. b 95% CIs were constructed using Clopper Pearson method. c PD-L 1 testing was based on IHC SP 263; 86 patients were evaluable for PD-L 1 status. Data cutoff: 14 June 2019. https: //bit. ly/2 ZCXq. Hc 10 Lee KH. Atezo + Bev in HCC

Baseline Characteristics of Complete Respondersa (n = 12) Characteristic Median age (range), years 62 (51 -78) Age ≥ 65 years, n (%) 4 (33) Race, n (%) Asian = 8 (67); White = 2 (17); Other = 2 (17) Region, n (%) Asia (excluding Japan) = 7 (58); Rest of World = 5 (42) Baseline ECOG PS 1, n (%) 5 (42) Child‑Pugh class, n (%) A 5 = 11 (92); A 6 = 1 (8) Etiology of HCC, n (%) HBV = 7 (58); HCV = 5 (42) BCLC stage at study entry, n (%) B = 2 (17); C = 10 (83) AFP ≥ 400 ng/m. L, n (%) 4 (33) EHS present, n (%) 9 (75) MVI present, n (%) 5 (42) EHS and/or MVI present, n (%) 10 (83) Prior HCC local therapy (ie TACE, RFA or TARE), n (%) 7 (58) 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA a Per IRF-assessed RECIST 1. 1. . Data cutoff: 14 June 2019. https: //bit. ly/2 ZCXq. Hc 11 Lee KH. Atezo + Bev in HCC
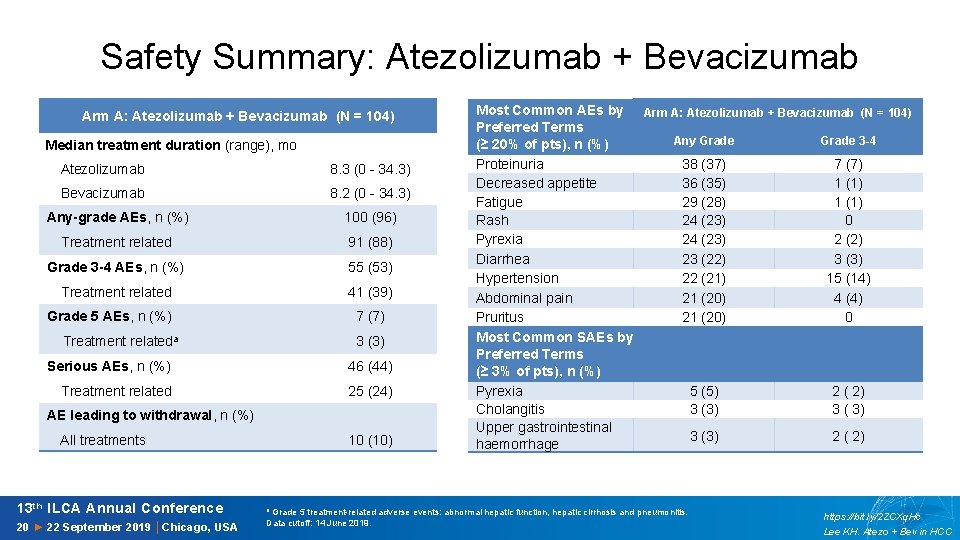
Safety Summary: Atezolizumab + Bevacizumab Arm A: Atezolizumab + Bevacizumab (N = 104) Median treatment duration (range), mo Atezolizumab 8. 3 (0 - 34. 3) Bevacizumab 8. 2 (0 - 34. 3) Any-grade AEs, n (%) 100 (96) Treatment related 91 (88) Grade 3 -4 AEs, n (%) 55 (53) Treatment related 41 (39) Grade 5 AEs, n (%) 7 (7) Treatment relateda 3 (3) Serious AEs, n (%) 46 (44) Treatment related 25 (24) AE leading to withdrawal, n (%) All treatments 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA 10 (10) Most Common AEs by Arm A: Atezolizumab + Bevacizumab (N = 104) Preferred Terms Any Grade 3 -4 (≥ 20% of pts), n (%) Proteinuria 38 (37) 7 (7) Decreased appetite 36 (35) 1 (1) Fatigue 29 (28) 1 (1) Rash 24 (23) 0 Pyrexia 24 (23) 2 (2) Diarrhea 23 (22) 3 (3) Hypertension 22 (21) 15 (14) Abdominal pain 21 (20) 4 (4) Pruritus 21 (20) 0 Most Common SAEs by Preferred Terms (≥ 3% of pts), n (%) Pyrexia 5 (5) 2 ( 2) Cholangitis 3 (3) 3 ( 3) Upper gastrointestinal 3 (3) 2 ( 2) haemorrhage a Grade 5 treatment-related adverse events: abnormal hepatic function, hepatic cirrhosis and pneumonitis. Data cutoff: 14 June 2019. https: //bit. ly/2 ZCXq. Hc Lee KH. Atezo + Bev in HCC
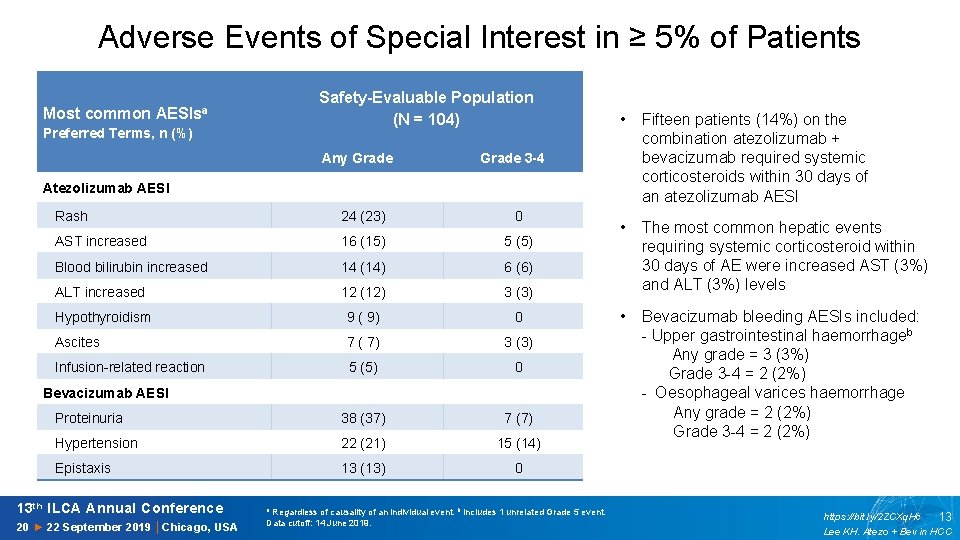
Adverse Events of Special Interest in ≥ 5% of Patients Most common AESIsa Preferred Terms, n (%) Safety-Evaluable Population (N = 104) Any Grade • Fifteen patients (14%) on the combination atezolizumab + bevacizumab required systemic corticosteroids within 30 days of an atezolizumab AESI • The most common hepatic events requiring systemic corticosteroid within 30 days of AE were increased AST (3%) and ALT (3%) levels Grade 3 -4 Atezolizumab AESI Rash 24 (23) 0 AST increased 16 (15) 5 (5) Blood bilirubin increased 14 (14) 6 (6) ALT increased 12 (12) 3 (3) Hypothyroidism 9 ( 9) Ascites 7 ( 7) 3 (3) Infusion-related reaction 5 (5) 0 Proteinuria 38 (37) 7 (7) Hypertension 22 (21) 15 (14) Epistaxis 13 (13) 0 • Bevacizumab bleeding AESIs included: 0 Bevacizumab AESI 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA a Regardless of causality of an individual event. b Includes 1 unrelated Grade 5 event. Data cutoff: 14 June 2019. - Upper gastrointestinal haemorrhageb Any grade = 3 (3%) Grade 3 -4 = 2 (2%) - Oesophageal varices haemorrhage Any grade = 2 (2%) Grade 3 -4 = 2 (2%) https: //bit. ly/2 ZCXq. Hc 13 Lee KH. Atezo + Bev in HCC

Summary • Clinically meaningful and durable objective responses were observed – Confirmed ORR with atezolizumab + bevacizumab was 36% per IRF-assessed RECIST 1. 1 – 76% of responses per IRF-assessed RECIST 1. 1 are ongoing and the median duration of response was not reached – 12% of patients achieved CR per IRF-assessed RECIST 1. 1 – Results were consistent across RECIST assessments • Benefit of atezolizumab + bevacizumab was generally consistent among subgroups analysed – Clinical activity was observed irrespective of PD-L 1 status • Combination of atezolizumab + bevacizumab was generally well tolerated and toxicities were manageable – No new safety signals were identified beyond the established single-agent safety profiles • Atezolizumab + bevacizumab may become a promising treatment option for patients with unresectable HCC – This combination is being evaluated further in the IMbrave 150 Phase III study (NCT 03434379) 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA Data cutoff: 14 June 2019. https: //bit. ly/2 ZCXq. Hc 14 Lee KH. Atezo + Bev in HCC

Acknowledgements and Study Investigators • • The patients and their families and caregivers Participating study investigators and clinical sites Y Bang J Bendell Y Chao J Chen H Chung S Davis A Dev E Gane B George • • R He H Hochstera C Hsu M Ikeda J Lee M Lee A Mahipal G Manji M Morimoto K Numata M Pishvaiana S Qin D Ryan B Ryoo N Sasahira S Stein J Strickler N Tebbutt This study is sponsored by F. Hoffmann-La Roche, Ltd. Medical writing assistance for this oral presentation was provided by Samantha Santangelo, Ph. D, of Health Interactions and funded by F. Hoffmann-La Roche, Ltd. 13 th ILCA Annual Conference 20 ► 22 September 2019 │Chicago, USA a Previous investigators. https: //bit. ly/2 ZCXq. Hc 15 Lee KH. Atezo + Bev in HCC
- Slides: 15