Phase Ib Efficacy Safety of AntiCD 47 Antibody

Phase Ib Efficacy, Safety of Anti-CD 47 Antibody Magrolimab + Azacitidine in Untreated AML CCO Independent Conference Highlights of the 2020 Virtual ASH Annual Meeting, December 5 -8, 2020 *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. Supported by educational grants from Amgen; Astra. Zeneca; Bristol-Myers Squibb; Epizyme, Inc. ; Glaxo. Smith. Kline; Incyte Corporation; Janssen Biotech; Karyopharm Therapeutics Inc. ; Novartis; Pharma. Essentia Corp. ; Seattle Genetics; and Takeda Oncology.
About These Slides § Please feel free to use, update, and share some or all of these slides in your noncommercial presentations to colleagues or patients § When using our slides, please retain the source attribution: Slide credit: clinicaloptions. com § These slides may not be published, posted online, or used in commercial presentations without permission. Please contact permissions@clinicaloptions. com for details

Magrolimab + Azacitidine in Untreated AML: Background § High levels of CD 47 expression in many cancers associated with macrophage immune evasion § Magrolimab: first-in-class macrophage immune checkpoint inhibitor targeting CD 47 ‒ Triggers macrophage phagocytosis of tumor cells ‒ May synergize with azacitidine induction of pro-phagocytic signals[1] § Phase Ib study explored safety, preliminary efficacy of magrolimab + azacitidine in untreated AML or MDS[2] § This analysis reporting preliminary results in AML cohort[2] 1. Feng. ASH 2018. Abstr 616. 2. Sallman. ASH 2020. Abstr 330. Slide credit: clinicaloptions. com

Magrolimab + Azacitidine in Untreated AML Cohort: Phase Ib Study Design Safety Cohort Untreated AML ineligible for induction chemotherapy (n = 64) Magrolimab 1 mg/kg priming dose, then 30 mg/kg QW + Azacitidine 75 mg/m 2 Days 1 -7 Expansion Cohort* Magrolimab 1 mg/kg priming dose, then 30 mg/kg Q 2 W + Azacitidine 75 mg/m 2 Days 1 -7 *Data presented from AML expansion cohort. § Primary endpoints: safety of magrolimab alone or with azacitidine; efficacy of combination in untreated AML/MDS § Secondary endpoints: magrolimab PK, PD, immunogenicity § Exploratory endpoints: CD 47 receptor occupancy, immune activity biomarkers, molecular profiling in AML/MDS Sallman. ASH 2020. Abstr 330. Slide credit: clinicaloptions. com
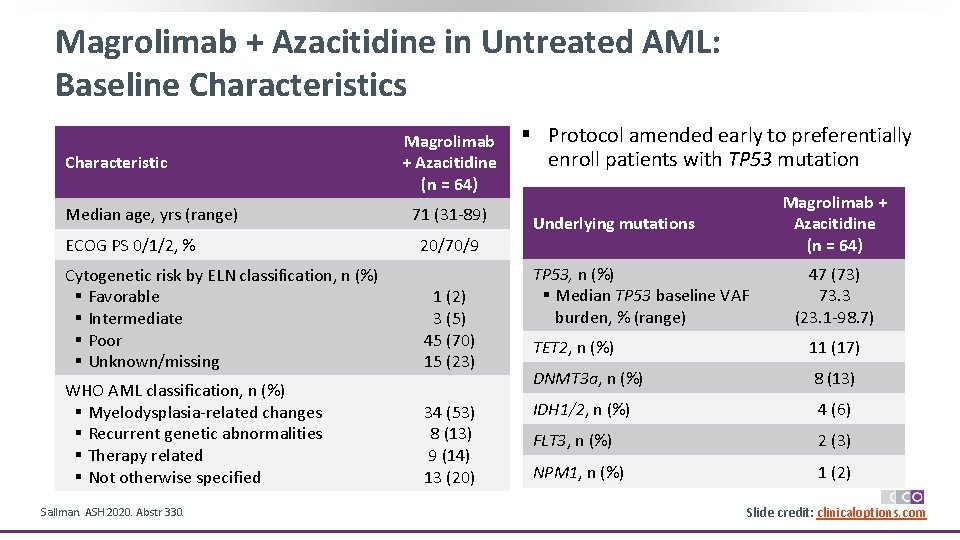
Magrolimab + Azacitidine in Untreated AML: Baseline Characteristics Characteristic Median age, yrs (range) ECOG PS 0/1/2, % Magrolimab + Azacitidine (n = 64) 71 (31 -89) 20/70/9 Cytogenetic risk by ELN classification, n (%) § Favorable § Intermediate § Poor § Unknown/missing 1 (2) 3 (5) 45 (70) 15 (23) WHO AML classification, n (%) § Myelodysplasia-related changes § Recurrent genetic abnormalities § Therapy related § Not otherwise specified 34 (53) 8 (13) 9 (14) 13 (20) Sallman. ASH 2020. Abstr 330. § Protocol amended early to preferentially enroll patients with TP 53 mutation Magrolimab + Azacitidine (n = 64) Underlying mutations TP 53, n (%) § Median TP 53 baseline VAF burden, % (range) 47 (73) 73. 3 (23. 1 -98. 7) TET 2, n (%) 11 (17) DNMT 3 a, n (%) 8 (13) IDH 1/2, n (%) 4 (6) FLT 3, n (%) 2 (3) NPM 1, n (%) 1 (2) Slide credit: clinicaloptions. com
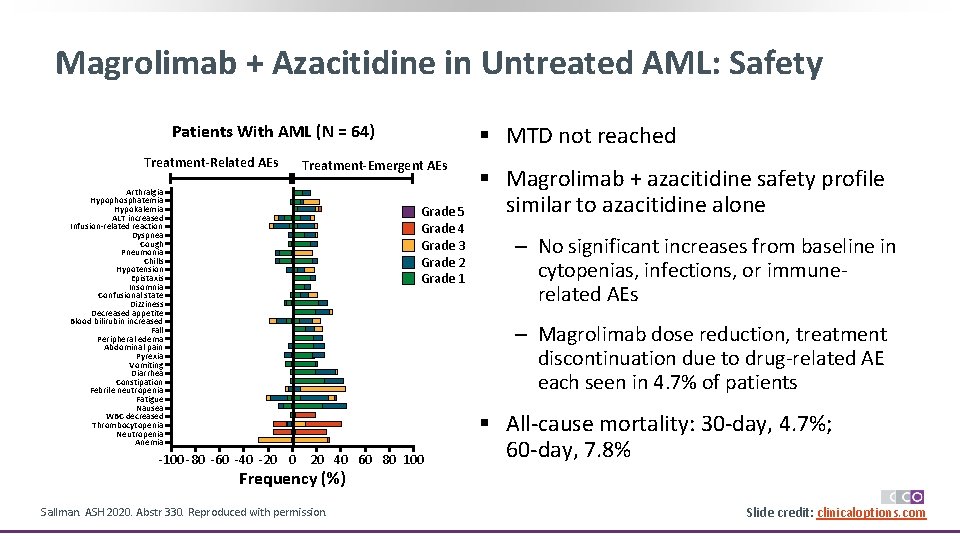
Magrolimab + Azacitidine in Untreated AML: Safety Patients With AML (N = 64) Treatment-Related AEs § MTD not reached Treatment-Emergent AEs Arthralgia Hypophosphatemia Hypokalemia ALT increased Infusion-related reaction Dyspnea Cough Pneumonia Chills Hypotension Epistaxis Insomnia Confusional state Dizziness Decreased appetite Blood bilirubin increased Fall Peripheral edema Abdominal pain Pyrexia Vomiting Diarrhea Constipation Febrile neutropenia Fatigue Nausea WBC decreased Thrombocytopenia Neutropenia Anemia Grade 5 Grade 4 Grade 3 Grade 2 Grade 1 § Magrolimab + azacitidine safety profile similar to azacitidine alone ‒ No significant increases from baseline in cytopenias, infections, or immunerelated AEs ‒ Magrolimab dose reduction, treatment discontinuation due to drug-related AE each seen in 4. 7% of patients -100 -80 -60 -40 -20 0 20 40 60 80 100 § All-cause mortality: 30 -day, 4. 7%; 60 -day, 7. 8% Frequency (%) Sallman. ASH 2020. Abstr 330. Reproduced with permission. Slide credit: clinicaloptions. com
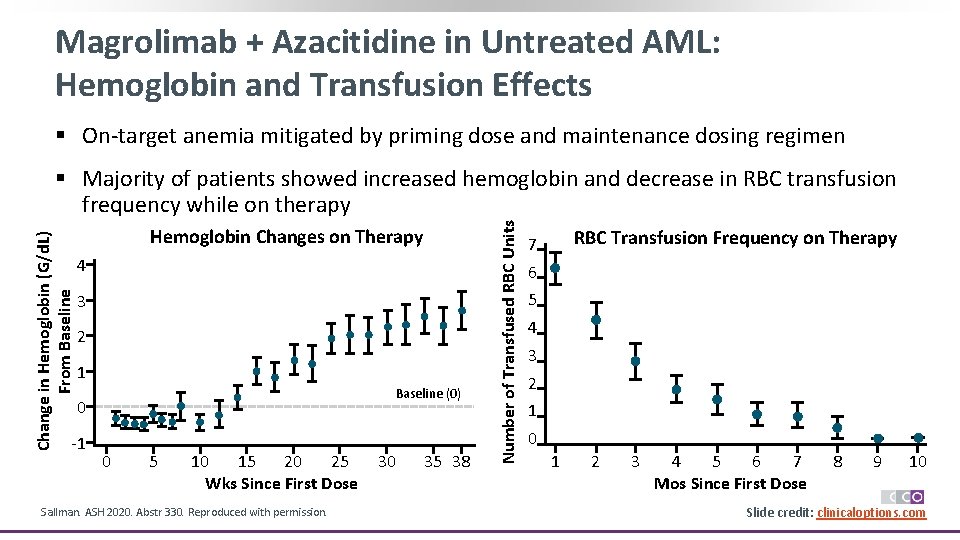
Magrolimab + Azacitidine in Untreated AML: Hemoglobin and Transfusion Effects § On-target anemia mitigated by priming dose and maintenance dosing regimen Hemoglobin Changes on Therapy 4 3 2 1 Baseline (0) 0 -1 0 5 10 15 20 25 Wks Since First Dose Sallman. ASH 2020. Abstr 330. Reproduced with permission. 30 35 38 Number of Transfused RBC Units Change in Hemoglobin (G/d. L) From Baseline § Majority of patients showed increased hemoglobin and decrease in RBC transfusion frequency while on therapy RBC Transfusion Frequency on Therapy 7 6 5 4 3 2 1 0 1 2 3 4 5 6 7 Mos Since First Dose 8 9 10 Slide credit: clinicaloptions. com
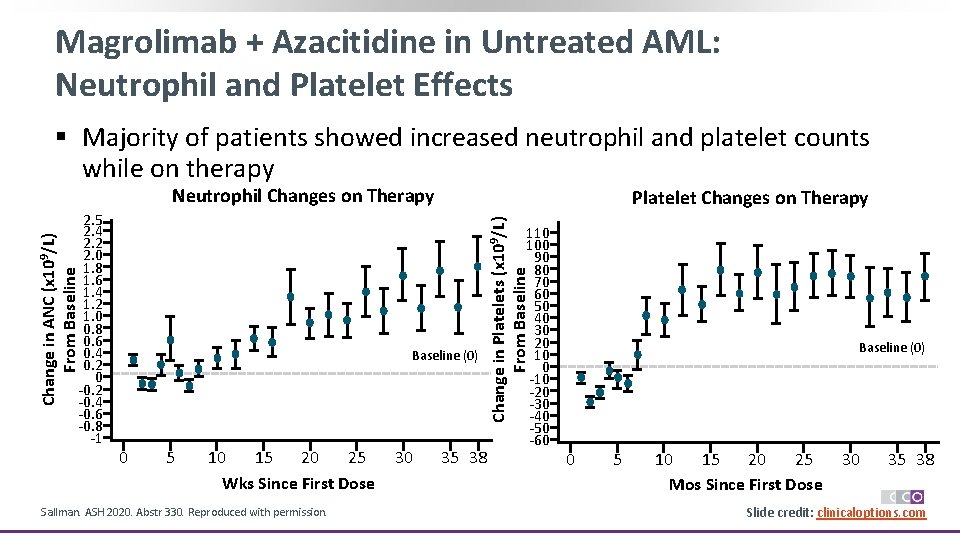
Magrolimab + Azacitidine in Untreated AML: Neutrophil and Platelet Effects § Majority of patients showed increased neutrophil and platelet counts while on therapy Neutrophil Changes on Therapy Baseline (0) 0 5 10 15 20 25 Wks Since First Dose Sallman. ASH 2020. Abstr 330. Reproduced with permission. 30 Change in Platelets (x 109/L) From Baseline Change in ANC (x 109/L) From Baseline 2. 5 2. 4 2. 2 2. 0 1. 8 1. 6 1. 4 1. 2 1. 0 0. 8 0. 6 0. 4 0. 2 0 -0. 2 -0. 4 -0. 6 -0. 8 -1 Platelet Changes on Therapy 35 38 110 100 90 80 70 60 50 40 30 20 10 0 -10 -20 -30 -40 -50 -60 Baseline (0) 0 5 10 15 20 25 Mos Since First Dose 30 35 38 Slide credit: clinicaloptions. com
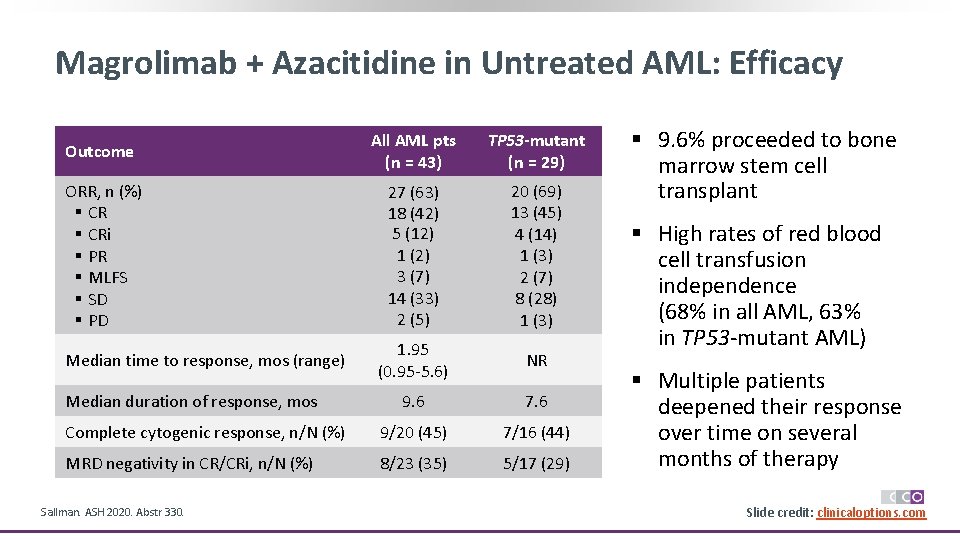
Magrolimab + Azacitidine in Untreated AML: Efficacy Outcome All AML pts (n = 43) TP 53 -mutant (n = 29) ORR, n (%) § CRi § PR § MLFS § SD § PD 27 (63) 18 (42) 5 (12) 1 (2) 3 (7) 14 (33) 2 (5) 20 (69) 13 (45) 4 (14) 1 (3) 2 (7) 8 (28) 1 (3) 1. 95 (0. 95 -5. 6) NR 9. 6 7. 6 Complete cytogenic response, n/N (%) 9/20 (45) 7/16 (44) MRD negativity in CR/CRi, n/N (%) 8/23 (35) 5/17 (29) Median time to response, mos (range) Median duration of response, mos Sallman. ASH 2020. Abstr 330. § 9. 6% proceeded to bone marrow stem cell transplant § High rates of red blood cell transfusion independence (68% in all AML, 63% in TP 53 -mutant AML) § Multiple patients deepened their response over time on several months of therapy Slide credit: clinicaloptions. com
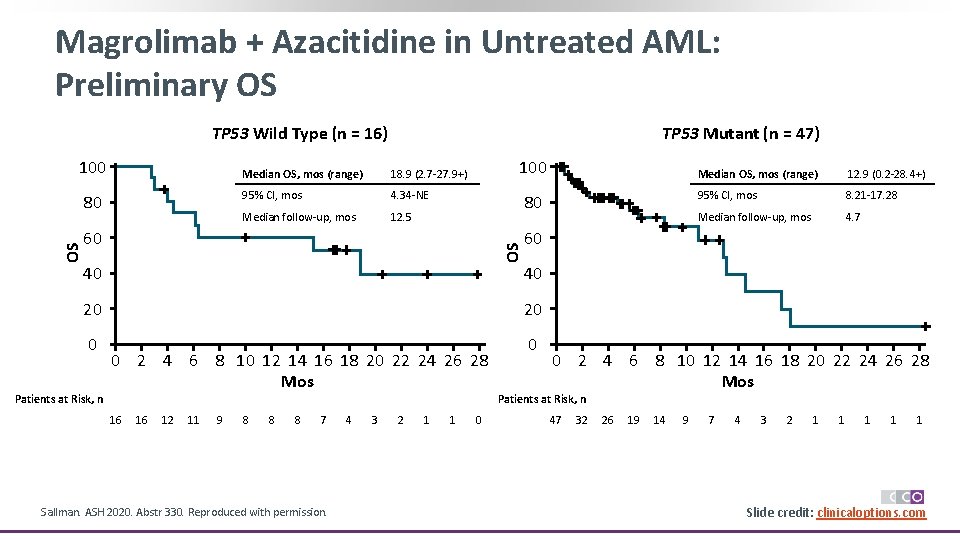
Magrolimab + Azacitidine in Untreated AML: Preliminary OS TP 53 Wild Type (n = 16) 100 Median OS, mos (range) 18. 9 (2. 7 -27. 9+) 95% CI, mos 4. 34 -NE Median follow-up, mos 12. 5 100 80 60 OS OS 80 TP 53 Mutant (n = 47) 40 0 0 16 16 12 11 9 8 8 8 7 Sallman. ASH 2020. Abstr 330. Reproduced with permission. 4 3 2 1 1 0 95% CI, mos 8. 21 -17. 28 Median follow-up, mos 4. 7 40 20 Patients at Risk, n 12. 9 (0. 2 -28. 4+) 60 20 0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 Mos Median OS, mos (range) 0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 Mos Patients at Risk, n 47 32 26 19 14 9 7 4 3 2 1 1 1 Slide credit: clinicaloptions. com

Magrolimab + Azacitidine in Untreated AML: Investigator Conclusions § Magrolimab + azacitidine combination induced high response rates in untreated AML patients ineligible for intensive chemotherapy[1] ‒ Combination associated with durable responses that deepened in many patients over time on therapy ‒ More rapid responses, higher CR rate than previously reported with azacitidine monotherapy[2, 3] ‒ ORR, CR rates similar in total patient cohort and those with TP 53 mutation § Combination was well tolerated, with no significant immune-related AEs and some improvement in cytopenias while on therapy § Randomized phase III trial planned vs venetoclax + azacitidine in patients with untreated TP 53 -mutant AML 1. Sallman. ASH 2020. Abstr 330. 2. Fenaux. JCO. 2010; 28: 562. 3. Dombret. Blood. 2015; 126: 291. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASH 2020! Short slideset summaries and additional CME-certified analyses with expert commentary on key studies in: § § § Acute and chronic leukemias Lymphomas and chronic lymphocytic leukemia Myelodysplastic syndromes and myeloproliferative neoplasms Myeloma Nonmalignant hematology clinicaloptions. com/oncology
- Slides: 12