Phase Equilibrium Phase Equilibrium Models Fugacity Equation of

Phase Equilibrium

Phase Equilibrium Models Fugacity (Equation of State) Models a) Soave Redlick Kwong (SRK) Activity Coefficient (g) Models a) Renon (NRTL) b) UNIQUAC C) UNIFAC Idealized Methods a) Depriester Charts

You need data to be accurate For whatever kind of puzzle you’ve got ---You just stick the right formula in ---A solution for every fool --The Indigo Girls

Condition of Equilibrium

Equation of State Approach Soave-Redlich-Kwong (SRK) Equation of State Soave, G. , “Equilibrium Constants from a Modified Redlich-Kwong Equation of State, ” Chemical Engineering Science, 27, 1197 -1203 (1972).

Soave-Redlich-Kwong (SRK) Equation of State

Soave-Redlich-Kwong (SRK) Equation of State

SRK Based Fugacity Coefficient

SRK Example Calculate the liquid and vapor phase fugacity coefficients for the following CO 2 (1) / methane (2) mixture using the SRK equation of state. Is the given composition at equilibrium ? T = -112 F P = 500 Psia y 1 = 0. 072 y 2 = 0. 928 x 1 = 0. 607 x 2 = 0. 393 kij = 0. 0933

Activity Coefficient Approach Binary Margules Equation

Activity Coefficient Approach Binary NRTL (Renon) Equation

Activity Coefficient Models Multi-component NRTL (Renon) Renon, H. , and Prausnitz, J. M. “Local Compositions in Thermodynamic Excess Functions for Liquid Mixtures, ” AICh. E J. , 14 (1), 135 -144 (1968).

Activity Coefficient Models NRTL (Renon) -- aij as a function of molecular classes 0. 30 - hydrocarbons, carbon tetrachloride, non-associated liquids, polar liquids. 0. 20 - hydrocarbons / polar non-associating liquids (hexane – acetone) 0. 40 - hydrocarbons / freons (hexane / perfluro-hexane) 0. 47 - alcohols / hydrocarbons (methanol / butane) 0. 47 - acetonitrile or nitromethane / carbon tetrachloride 0. 30 - water / polar non-associating liquids (water / acetone) 0. 47 - water / self associating liquids (water / pyridine)

Activity Coefficient Models UNIQUAC Combinatorial (size / shape) Residual (intermolecular forces) Abrams, D. S. , Prausnitz, J. M. , “Statistical Thermodynamics of Liquid Mixtures: A New Expression for the Excess Gibbs Energy of Partly or Completely Miscible Systems, ” AICh. E J. , 21 (1), 116 -128 (1975).

Sources of Data Experimental Data Literature Databases Gmehling, J. , and U. Onken Vapor-liquid Equilibrium Data Collection DECHEMA Chem. Data Ser. , 1 -8 (1977 -1984)
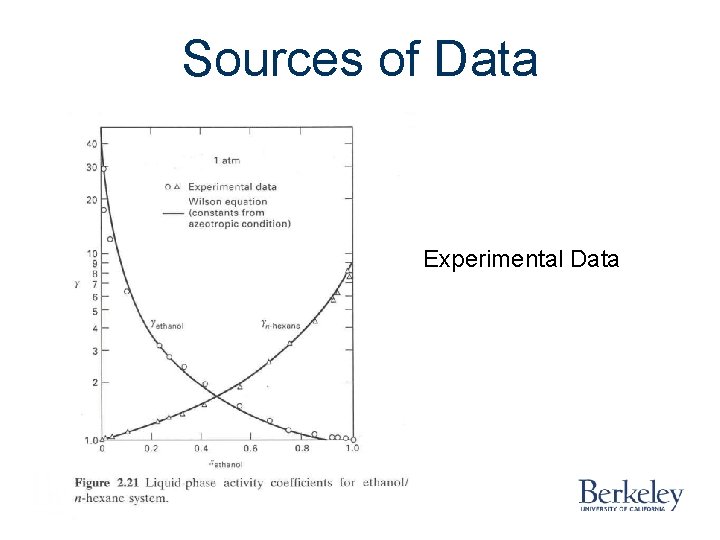
Sources of Data Experimental Data
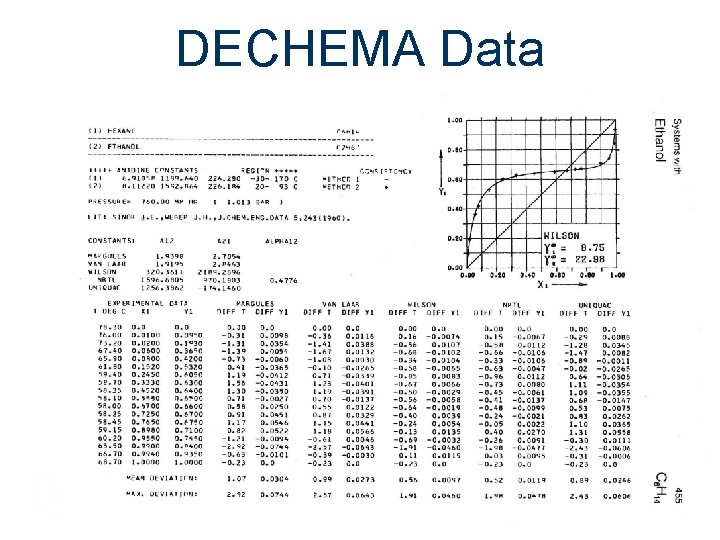
DECHEMA Data

Infinite Dilution Activity Coefficient (NRTL)

NRTL Example For the 2 -propanol (1) / water (2) system at 1 atm the infinite dilution activity coefficients are: g 1 = 11. 54 and g 2 = 3. 050. Determine t 12 and t 21 for use in the NRTL equation Determine the activity coefficient at the azeotrope composition (x 1 = 0. 6854)

10 Minute Problem The Van Laar activity coefficient model (Table 2. 9, page 57) is being used to predict the distribution of a binary mixture. Determine the activity coefficient value for a 50/50 molar mixture given the flowing infinite dilution activity coefficient data.

UNIFAC UNIQUAC Functional-group Activity Coefficient A Group Contribution Approach Fredenslund, A. , Jones, R. L. , Prausnitz, J. M. , “Group Contribution Estimation of Activity Coefficients in Non-ideal Liquid Mixtures, ” AICh. E J. , 21 (6), 1086 -1099 (1975).
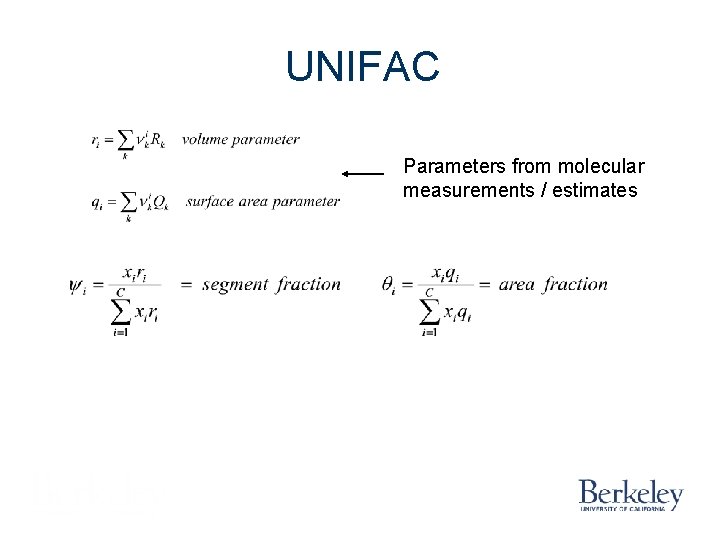
UNIFAC Parameters from molecular measurements / estimates

UNIFAC Combinatorial Component Groups in mixture Residual Component Groups in components

UNIFAC Example Determine the activity coefficients of acetone (1) and n-pentane (2) at 307 K and x 1=0. 047

UNIFAC Example Combinatorial Term ( CH 3 (1 A) CH 2 (1 B) C=O (8) CH 2 (1 B) CH 3 (1 A) Acetone (1) Pentane (2) )
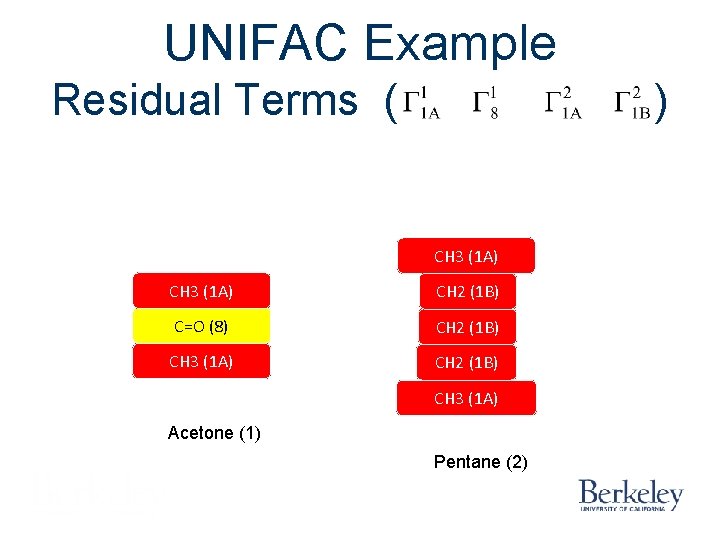
UNIFAC Example Residual Terms ( ) CH 3 (1 A) CH 2 (1 B) C=O (8) CH 2 (1 B) CH 3 (1 A) Acetone (1) Pentane (2)
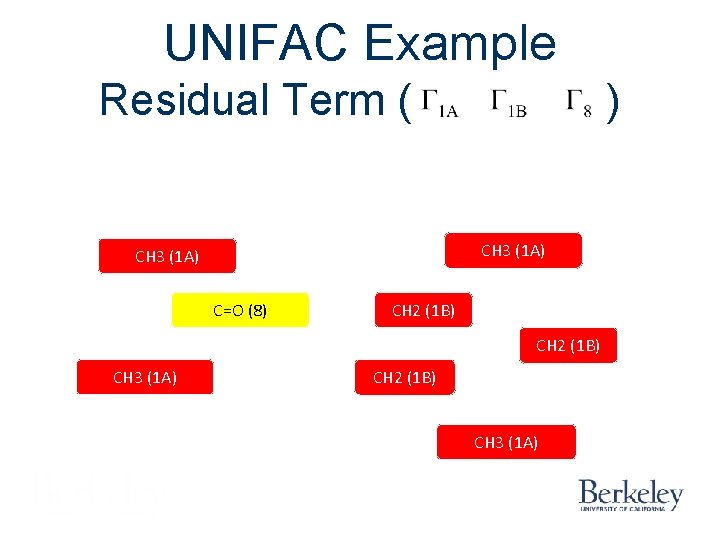
UNIFAC Example Residual Term ( ) CH 3 (1 A) C=O (8) CH 2 (1 B) CH 3 (1 A)

Equilibrium Flash Vapor Feed Liquid

Practical Problem
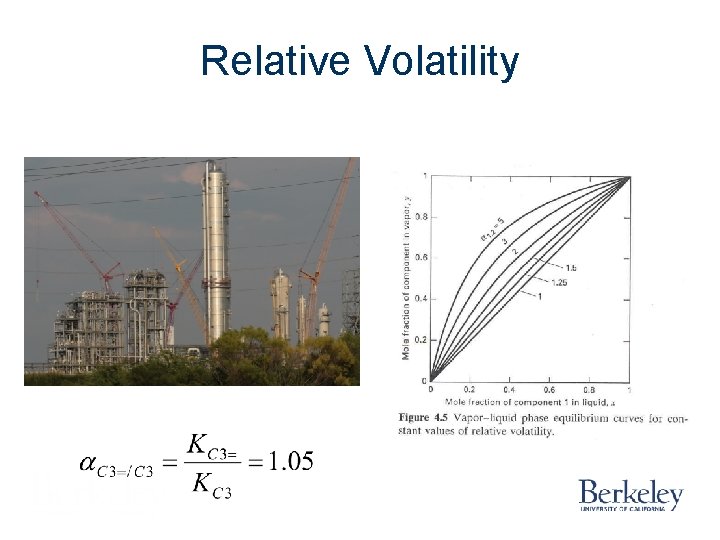
Relative Volatility

K Factor Depriester Chart

Isothermal Flash Calculation V, yi Fixed T, P F, zi L, xi Trial and Error solution

Flash Example Calculate the bubble point, dew point and fractional flash (T= 180 F) for the following mixture at 200 Psia. C 2 C 3 n-C 4 n-C 5 = 8 mole % = 22 mole % = 53 mole % = 17 mole %
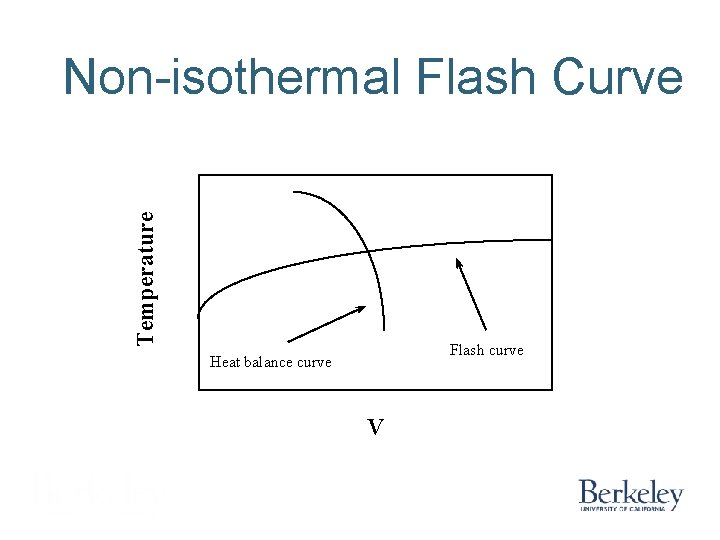
Temperature Non-isothermal Flash Curve Flash curve Heat balance curve V
- Slides: 34