Phase Equilibrium Part 6 Chemistry Engineering Department 23102013

Phase Equilibrium Part 6 Chemistry Engineering Department 23/10/2013

Some Definitions System: A body of engineering material under investigation. e. g. Ag – Cu system, Ni. O-Mg. O system (or even sugar-milk system) Component of a system: Pure metals and or compounds of which an alloy is composed, e. g. Cu and Ag or Fe and Fe 3 C. They are the solutes and solvent Solubility Limit ﺍﻟﺬﻭﺑﺎﻧﻴﺔ : The maximum concentration of solute atoms that may dissolve in the Solvent to form a “solid solution” at some temperature.

Definitions Phases: A homogenous portion of a system that has uniform physical and chemical characteristics, e. g. pure material, solid solution, liquid solution, and gaseous solution, ice and water, syrup and sugar. Single phase system = Homogeneous system Multi phase system = Heterogeneous system or mixtures Microstructure: A system’s microstructure is characterized by the number of phases present, their proportions, and the manner in which they are distributed or arranged. • Factors affecting microstructure are: 1. alloying elements present, 2. their concentrations, 3. and the heat treatment of the alloy.
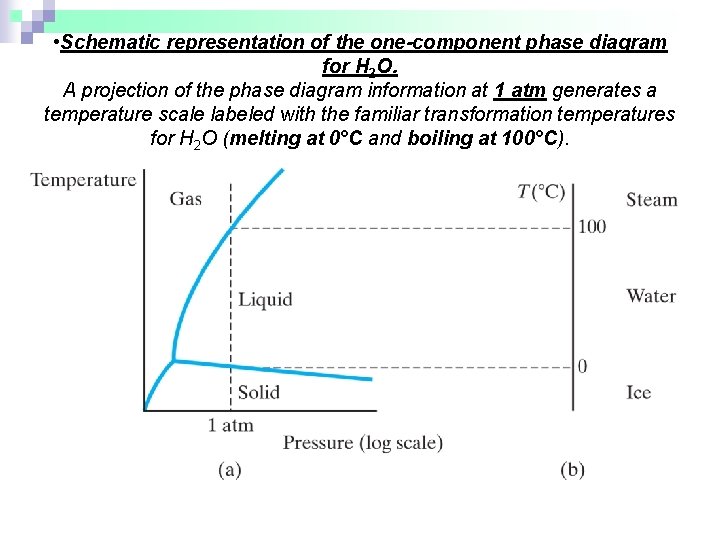
• Schematic representation of the one-component phase diagram for H 2 O. A projection of the phase diagram information at 1 atm generates a temperature scale labeled with the familiar transformation temperatures for H 2 O (melting at 0°C and boiling at 100°C).
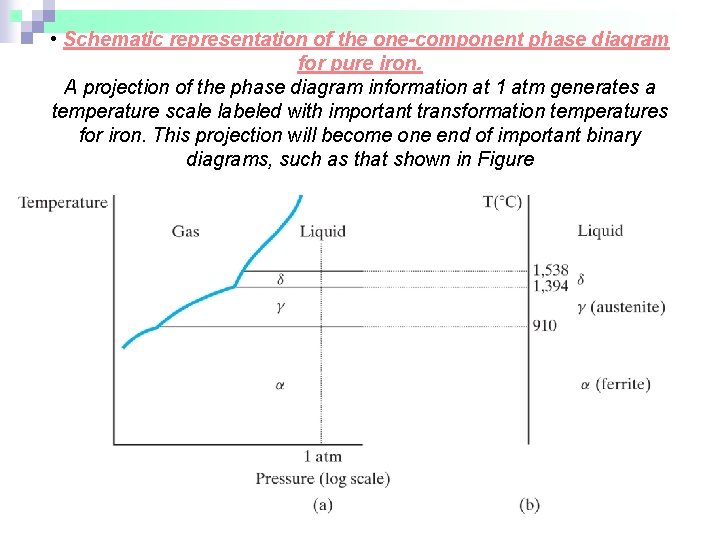
• Schematic representation of the one-component phase diagram for pure iron. A projection of the phase diagram information at 1 atm generates a temperature scale labeled with important transformation temperatures for iron. This projection will become one end of important binary diagrams, such as that shown in Figure

Definitions 1. Phase Equilibrium: • A stable configuration with : • lowest free-energy (internal energy of a system, and also randomness or disorder of the atoms or molecules (entropy). Any change in Temperature, Composition, and Pressure causes an increase in free energy and away from Equilibrium thus forcing a move to another ‘state’ 2. Equilibrium Phase Diagram: It is a “map” of the information about: - the control of microstructure or phase structure of a particular material system. The relationships between temperature and the compositions and the quantities of phases present at equilibrium are represented.

3. Binary Isomorphous Systems: An alloy system that contains two components that attain complete liquid and solid solubility of the components, e. g. , Cu and Ni alloy, It is the simplest binary system. 4. Binary Eutectic Systems (Poly. Morphic): An alloy system that contains two components that has a special composition with a minimum melting temperature.

• When we combine two elements. . . what “equilibrium state” would we expect to get? • In particular, if we specify. . . --a composition (e. g. , wt% Cu - wt% Ni), and --a temperature (T ) and/or a Pressure (P) then. . . How many phases do we get? What is the composition of each phase? How much of each phase do we get? Phase B Phase A Nickel atom Copper atom

Gibb’s Phase Rule: a tool to define the number of phases and/or degrees of phase changes that can be found in a system at equilibrium For any system under study this Gibb’s Phase Rule rule determines: 1. if the system is at equilibrium 2. 3. To predict how many phases can be expected predict how many independent parameters (degrees of freedom) Typically, N = 1 in most condensed systems – pressure is fixed!
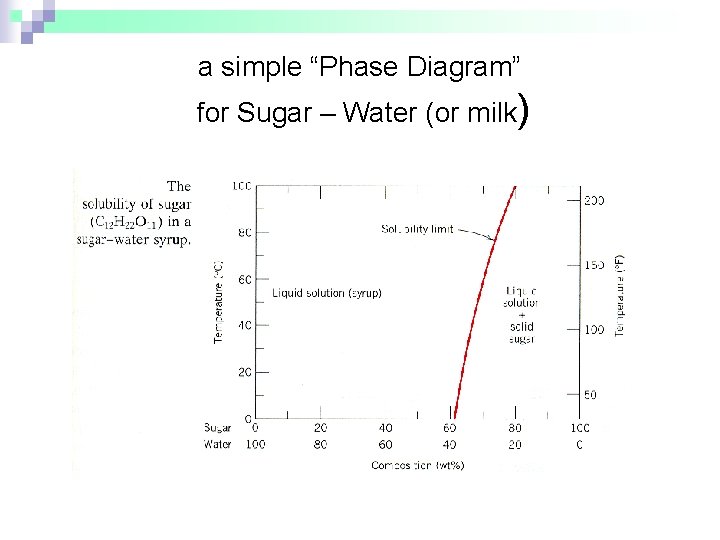
a simple “Phase Diagram” for Sugar – Water (or milk)
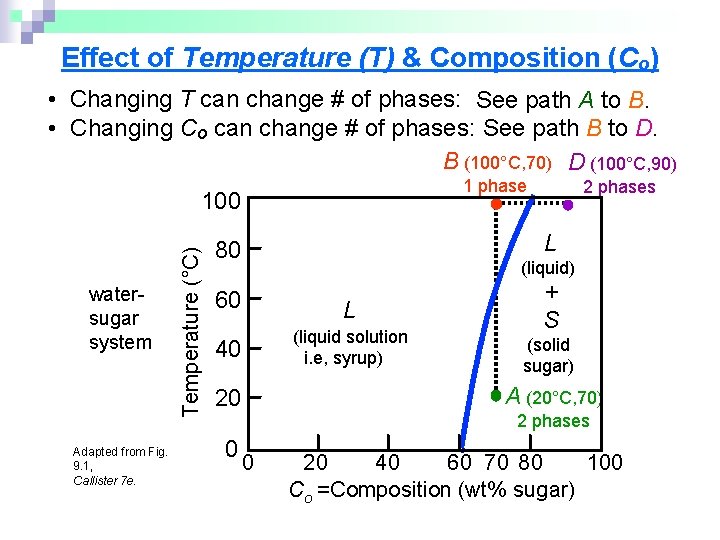
Effect of Temperature (T) & Composition (Co) • Changing T can change # of phases: See path A to B. • Changing Co can change # of phases: See path B to D. B (100°C, 70) D (100°C, 90) 1 phase watersugar system Adapted from Fig. 9. 1, Callister 7 e. Temperature (°C) 100 L 80 (liquid) 60 L (liquid solution i. e, syrup) 40 + S (solid sugar) A (20°C, 70) 20 0 2 phases 0 20 40 60 70 80 100 Co =Composition (wt% sugar)
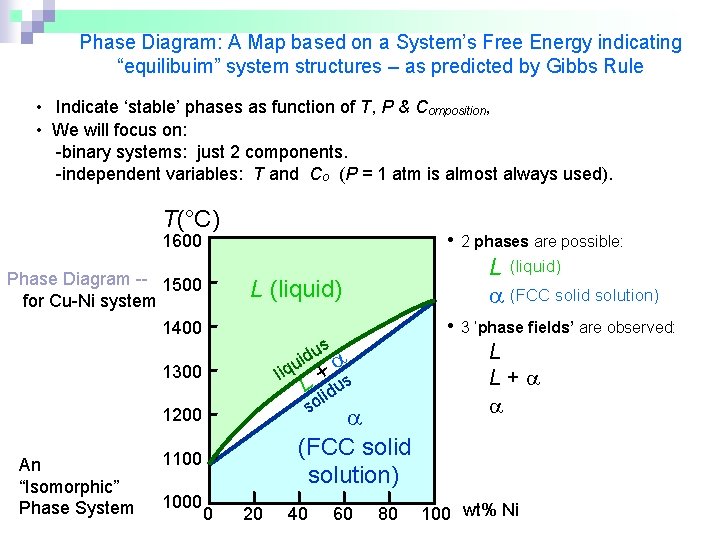
Phase Diagram: A Map based on a System’s Free Energy indicating “equilibuim” system structures – as predicted by Gibbs Rule • Indicate ‘stable’ phases as function of T, P & Composition, • We will focus on: -binary systems: just 2 components. -independent variables: T and Co (P = 1 atm is almost always used). T(°C) • 2 phases are possible: 1600 Phase Diagram -- 1500 for Cu-Ni system 1400 L (liquid) us d i u liq + s L lidu so 1300 (FCC solid solution) 1200 An “Isomorphic” Phase System 1100 1000 L (liquid) (FCC solid solution) 0 20 40 60 80 • 3 ‘phase fields’ are observed: L L+ 100 wt% Ni
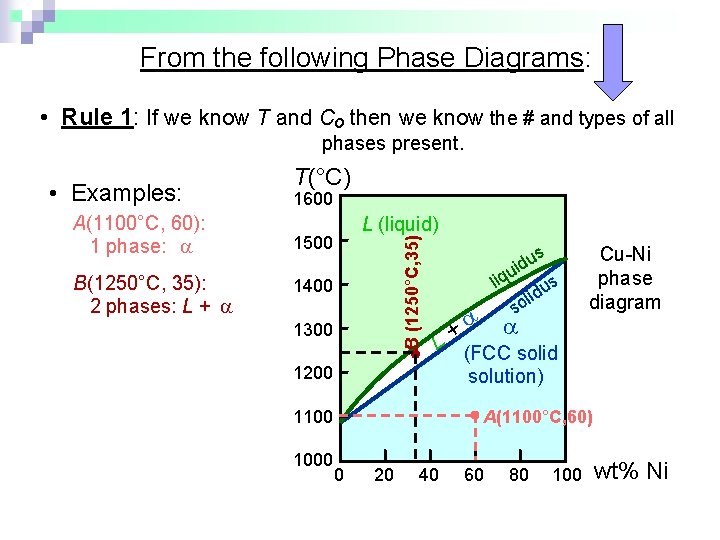
From the following Phase Diagrams: • Rule 1: If we know T and Co then we know the # and types of all phases present. A(1100°C, 60): 1 phase: B(1250°C, 35): 2 phases: L + 1600 L (liquid) 1500 B (1250°C, 35) • Examples: T(°C) 1400 1300 + L 1200 1100 1000 us d i u liq us d i l so Cu-Ni phase diagram (FCC solid solution) A(1100°C, 60) 0 20 40 60 80 100 wt% Ni
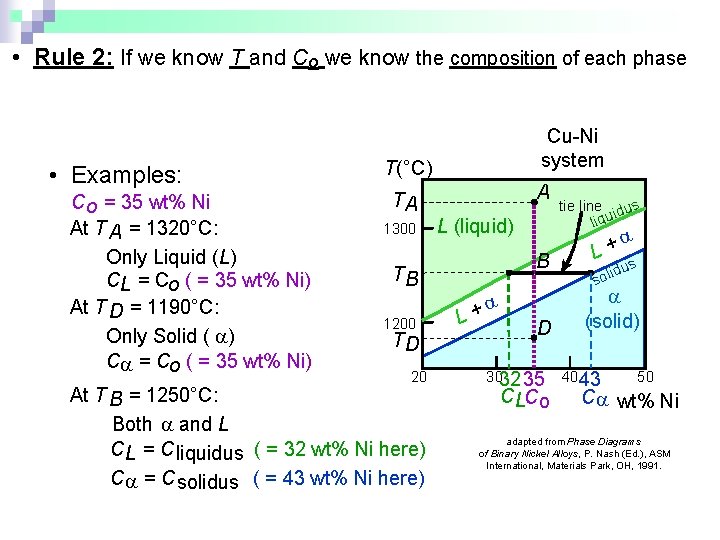
• Rule 2: If we know T and Co we know the composition of each phase • Examples: T(°C) Cu-Ni system A TA Co = 35 wt% Ni tie line dus ui q i l 1300 L (liquid) At T A = 1320°C: + L Only Liquid (L) B s idu l T o B s CL = Co ( = 35 wt% Ni) At T D = 1190°C: L+ (solid) 1200 D Only Solid ( ) TD C = Co ( = 35 wt% Ni) 20 3032 35 4043 50 At T B = 1250°C: CLCo C wt% Ni Both and L adapted from Phase Diagrams CL = C liquidus ( = 32 wt% Ni here) of Binary Nickel Alloys, P. Nash (Ed. ), ASM International, Materials Park, OH, 1991. C = C solidus ( = 43 wt% Ni here)
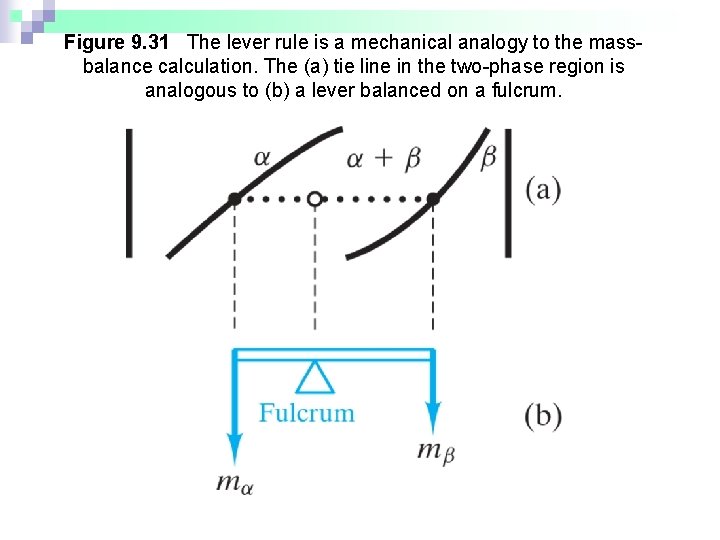
Figure 9. 31 The lever rule is a mechanical analogy to the massbalance calculation. The (a) tie line in the two-phase region is analogous to (b) a lever balanced on a fulcrum.
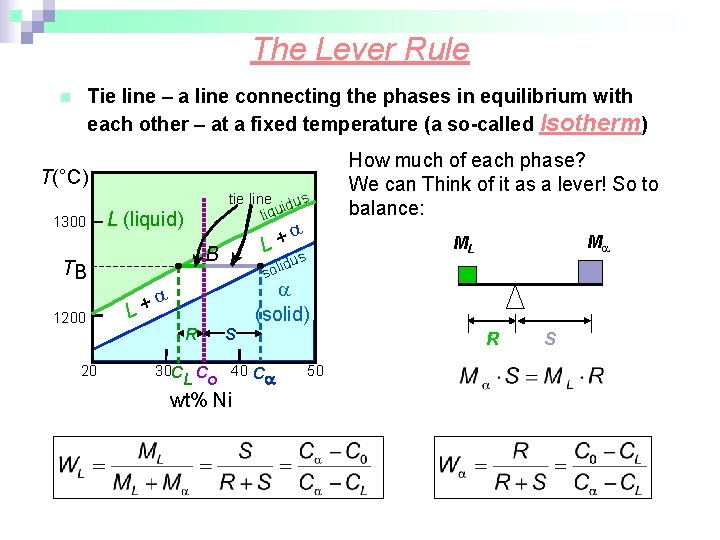
The Lever Rule Tie line – a line connecting the phases in equilibrium with each other – at a fixed temperature (a so-called Isotherm) n T(°C) 1300 tie line dus i liqu L (liquid) B TB 1200 20 + L M ML us id sol L+ R How much of each phase? We can Think of it as a lever! So to balance: S (solid) 30 C C 40 C L o wt% Ni R 50 S
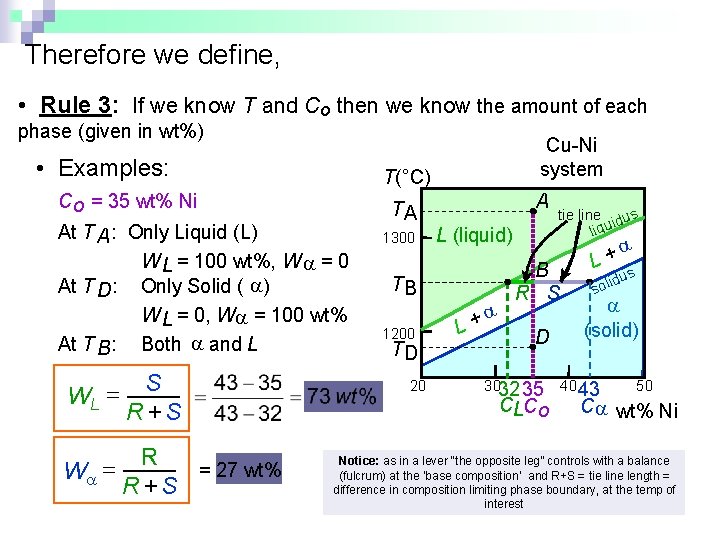
Therefore we define, • Rule 3: If we know T and Co then we know the amount of each phase (given in wt%) • Examples: T(°C) Co = 35 wt% Ni At T A : Only Liquid (L) W L = 100 wt%, W = 0 At T D: Only Solid ( ) W L = 0, W = 100 wt% At T B : Both and L WL = S R +S W = R = 27 wt% R +S Cu-Ni system TA 1300 A L (liquid) TB 1200 TD 20 + L tie line dus i liqu B R S D 3032 35 CLCo + L s idu l o s (solid) 40 43 50 C wt% Ni Notice: as in a lever “the opposite leg” controls with a balance (fulcrum) at the ‘base composition’ and R+S = tie line length = difference in composition limiting phase boundary, at the temp of interest
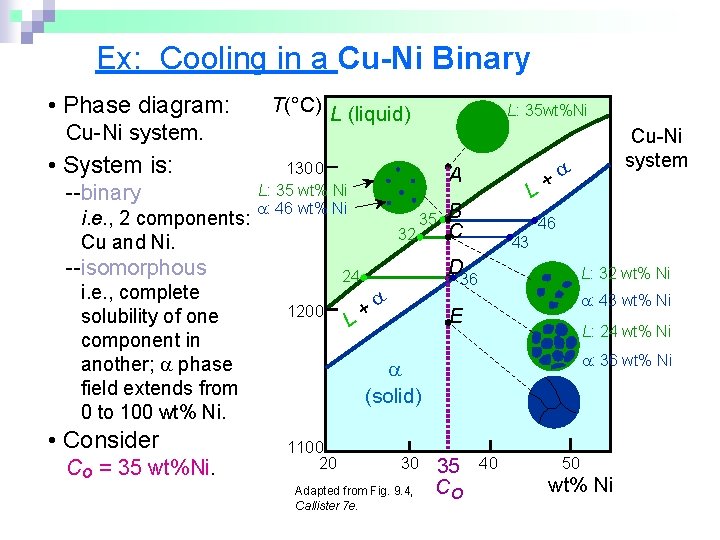
Ex: Cooling in a Cu-Ni Binary • Phase diagram: Cu-Ni system. • System is: --binary i. e. , 2 components: Cu and Ni. T(°C) L (liquid) 1300 L: 35 wt% Ni : 46 wt% Ni i. e. , complete solubility of one component in another; phase field extends from 0 to 100 wt% Ni. • Consider Co = 35 wt%Ni. A 32 --isomorphous L: 35 wt%Ni 35 B C 46 43 D 24 1200 L+ L: 32 wt% Ni 36 + L : 43 wt% Ni E L: 24 wt% Ni : 36 wt% Ni (solid) 1100 20 30 Adapted from Fig. 9. 4, Callister 7 e. Cu-Ni system 35 Co 40 50 wt% Ni

1. Cored vs Equilibrium Phases • C changes as we solidify. • Cu-Ni case: First to solidify has C = 46 wt% Ni. Last to solidify has C = 35 wt% Ni. • Fast rate of cooling: Cored structure • Slow rate of cooling: Equilibrium structure First to solidify: 46 wt% Ni Last to solidify: < 35 wt% Ni Uniform C : 35 wt% Ni
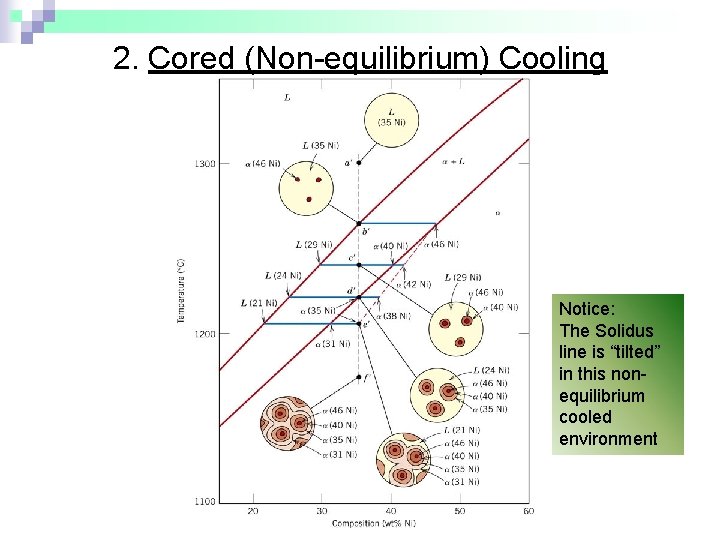
2. Cored (Non-equilibrium) Cooling Notice: The Solidus line is “tilted” in this nonequilibrium cooled environment
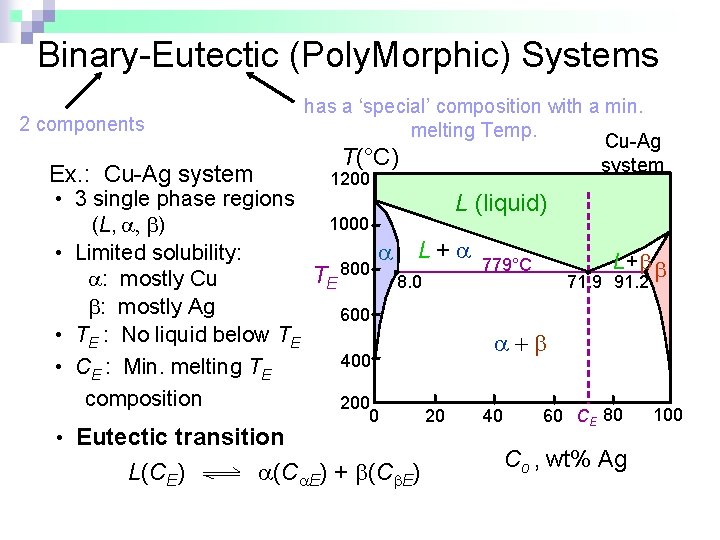
Binary-Eutectic (Poly. Morphic) Systems has a ‘special’ composition with a min. melting Temp. Cu-Ag T(°C) system 2 components Ex. : Cu-Ag system 1200 • 3 single phase regions L (liquid) 1000 (L, , ) L + 779°C • Limited solubility: L+ 800 T : mostly Cu 8. 0 71. 9 91. 2 E : mostly Ag 600 • TE : No liquid below TE 400 • CE : Min. melting TE composition 200 • Eutectic transition L(CE) 0 (C E) + (C E) 20 40 60 CE 80 Co , wt% Ag 100
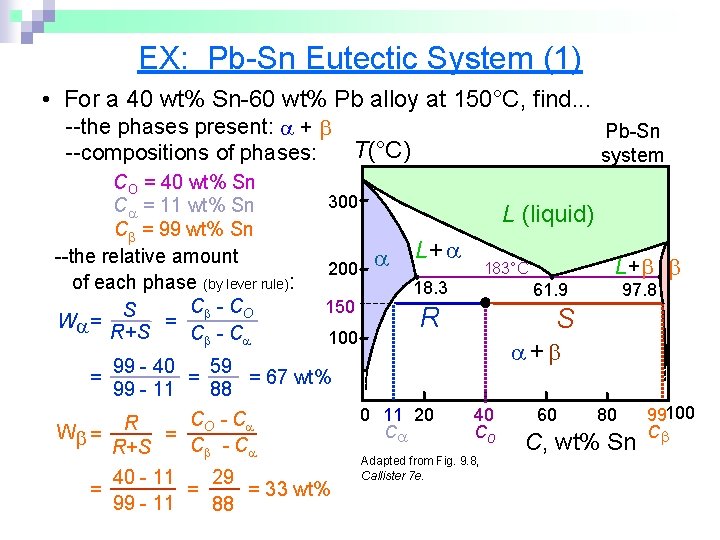
EX: Pb-Sn Eutectic System (1) • For a 40 wt% Sn-60 wt% Pb alloy at 150°C, find. . . --the phases present: + T(°C) --compositions of phases: CO = 40 wt% Sn C = 11 wt% Sn C = 99 wt% Sn --the relative amount of each phase (by lever rule): C - CO S = W = R+S C - C Pb-Sn system 300 200 150 100 99 - 40 59 = = 67 wt% 99 - 11 88 C - C W = R = O C - C R+S L (liquid) L+ 183°C 18. 3 61. 9 R L+ 97. 8 S + = = 40 - 11 29 = = 33 wt% 99 - 11 88 0 11 20 C 40 Co Adapted from Fig. 9. 8, Callister 7 e. 60 80 C, wt% Sn 99100 C
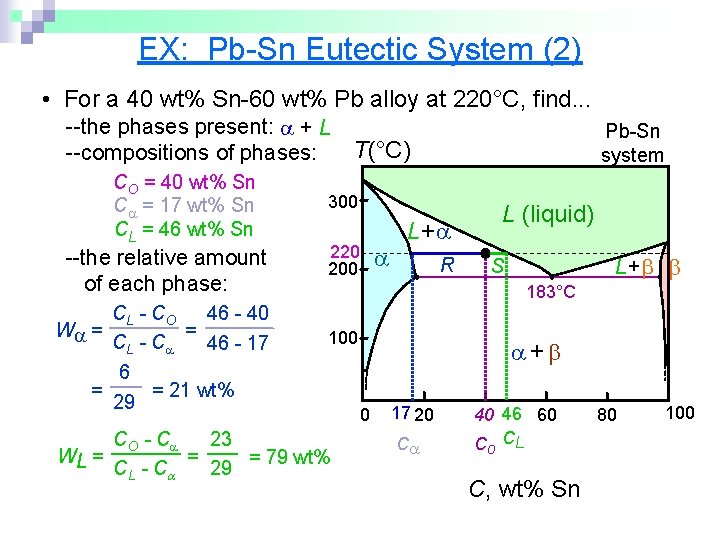
EX: Pb-Sn Eutectic System (2) • For a 40 wt% Sn-60 wt% Pb alloy at 220°C, find. . . --the phases present: + L T(°C) --compositions of phases: CO = 40 wt% Sn C = 17 wt% Sn CL = 46 wt% Sn --the relative amount of each phase: CL - C O 46 - 40 = W = CL - C 46 - 17 6 = = 21 wt% 29 Pb-Sn system 300 220 200 L (liquid) L+ R L+ S 183°C 100 CO - C 23 = WL = = 79 wt% CL - C 29 + 0 17 20 C 40 46 60 C CL o C, wt% Sn 80 100

Microstructures In Eutectic Systems: I • Co < 2 wt% Sn • Result: T(°C) L: Co wt% Sn 400 L --at extreme ends 300 --polycrystal of grains i. e. , only one solid phase. L 200 L+ (Pb-Sn System) : Co wt% Sn TE 100 + 0 Co 10 20 30 Co, wt% Sn 2% (room Temp. solubility limit)
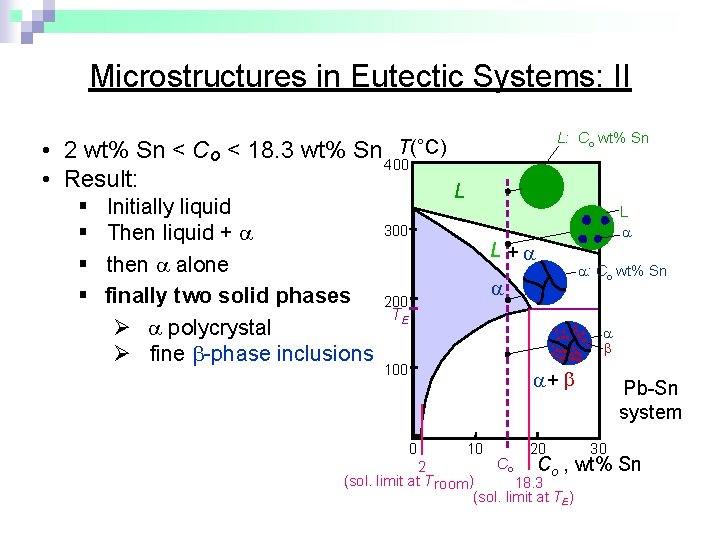
Microstructures in Eutectic Systems: II L: Co wt% Sn T(°C) • 2 wt% Sn < Co < 18. 3 wt% Sn 400 • Result: § Initially liquid § Then liquid + § then alone § finally two solid phases Ø polycrystal Ø fine -phase inclusions L L 300 L + 200 TE : Co wt% Sn 100 + 0 10 20 Pb-Sn system 30 Co Co , wt% 2 (sol. limit at T room ) 18. 3 (sol. limit at TE) Sn
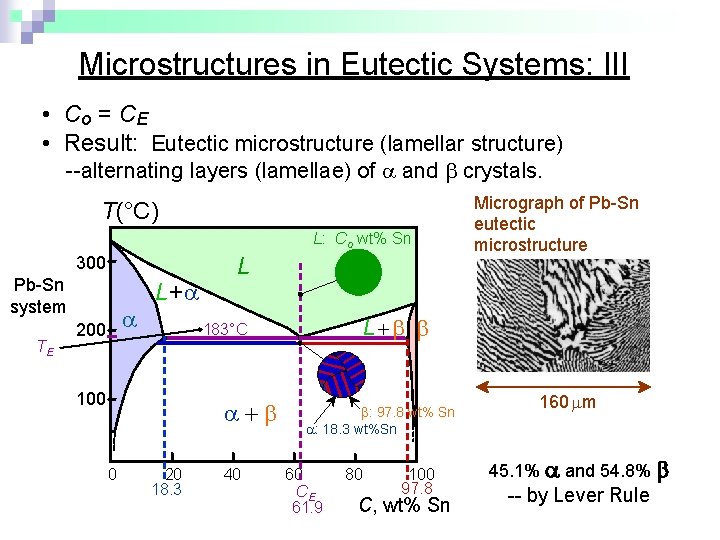
Microstructures in Eutectic Systems: III • Co = CE • Result: Eutectic microstructure (lamellar structure) --alternating layers (lamellae) of and crystals. T(°C) L: Co wt% Sn 300 Pb-Sn system TE 200 L+ L 0 L 183°C 100 20 18. 3 40 Micrograph of Pb-Sn eutectic microstructure : 97. 8 wt% Sn : 18. 3 wt%Sn 60 CE 61. 9 80 100 97. 8 C, wt% Sn 160 m 45. 1% and 54. 8% -- by Lever Rule
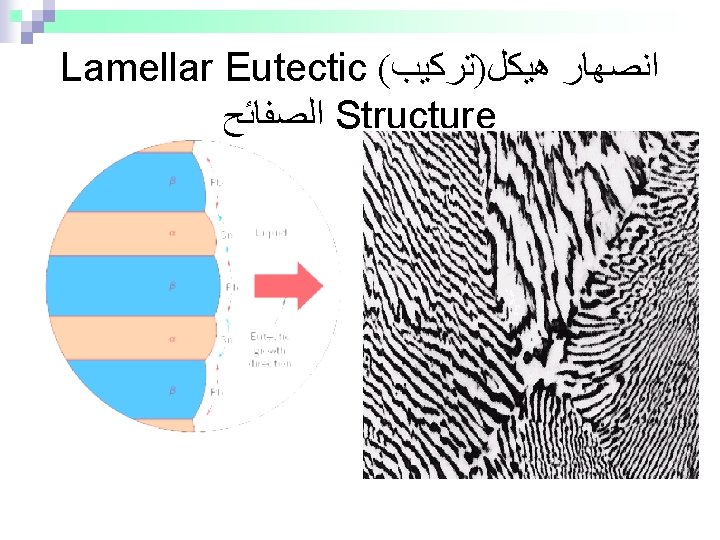
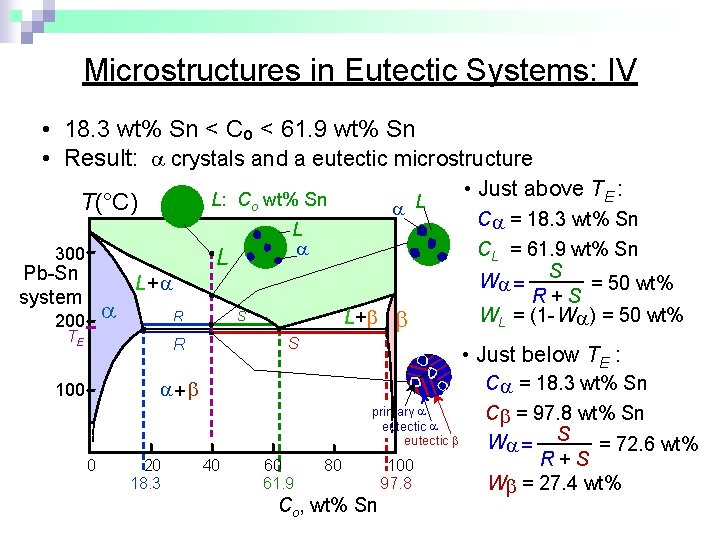
Microstructures in Eutectic Systems: IV • 18. 3 wt% Sn < Co < 61. 9 wt% Sn • Result: crystals and a eutectic microstructure T(°C) L: Co wt% Sn 300 L L Pb-Sn system 200 L L+ R TE L+ S S R C = 18. 3 wt% Sn primary eutectic 0 20 18. 3 C = 18. 3 wt% Sn CL = 61. 9 wt% Sn W = S = 50 wt% R+S WL = (1 - W ) = 50 wt% • Just below TE : + 100 • Just above TE : 40 60 61. 9 80 Co, wt% Sn 100 97. 8 C = 97. 8 wt% Sn W = S = 72. 6 wt% R+S W = 27. 4 wt%
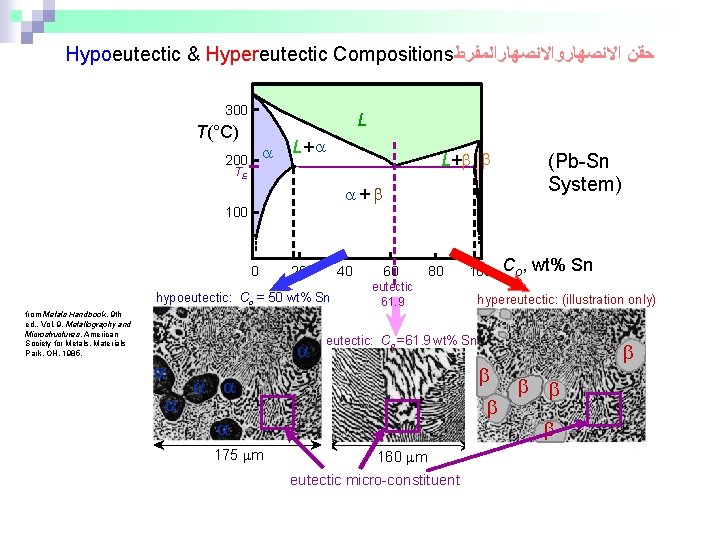
Hypoeutectic & Hypereutectic Compositions ﺣﻘﻦ ﺍﻻﻧﺼﻬﺎﺭﻭﺍﻻﻧﺼﻬﺎﺭﺍﻟﻤﻔﺮﻁ 300 L T(°C) 200 L+ TE + 100 0 20 40 hypoeutectic: Co = 50 wt% Sn from Metals Handbook, 9 th ed. , Vol. 9, Metallography and Microstructures, American Society for Metals, Materials Park, OH, 1985. 60 80 eutectic 61. 9 Co, wt% Sn hypereutectic: (illustration only) eutectic: Co = 61. 9 wt% Sn 175 m 100 (Pb-Sn System) 160 m eutectic micro-constituent
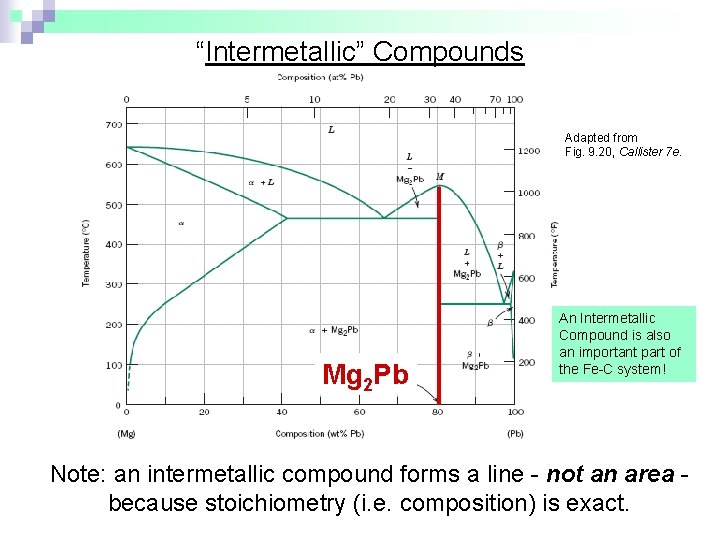
“Intermetallic” Compounds Adapted from Fig. 9. 20, Callister 7 e. Mg 2 Pb An Intermetallic Compound is also an important part of the Fe-C system! Note: an intermetallic compound forms a line - not an area because stoichiometry (i. e. composition) is exact.

Eutectoid & Peritectic – some definitions n n Eutectic: a liquid in equilibrium with two solids cool L heat + Eutectoid: solid phase in equilibrium with two solid phases S 2 S 1+S 3 intermetallic compound - cementite n cool heat + Fe 3 C (727ºC) Peritectic: liquid + solid 1 in equilibrium with a single solid 2 (Fig 9. 21) S 1 + L S 2 cool +L heat (1493ºC)
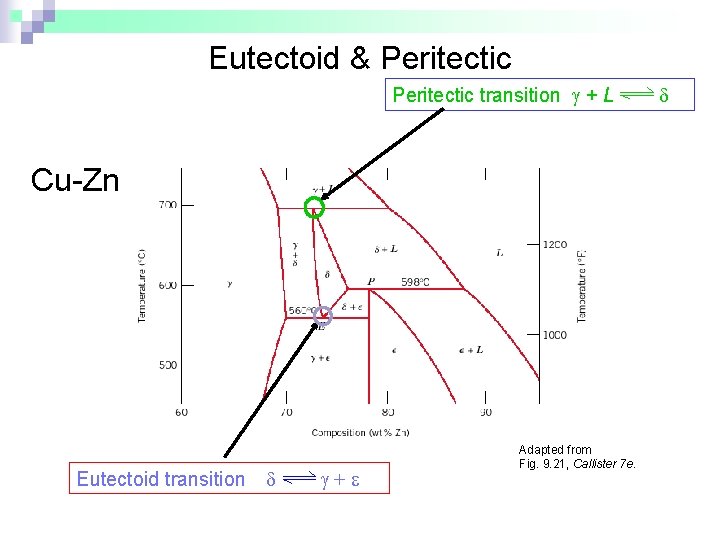
Eutectoid & Peritectic transition + L Cu-Zn Phase diagram Eutectoid transition + Adapted from Fig. 9. 21, Callister 7 e.
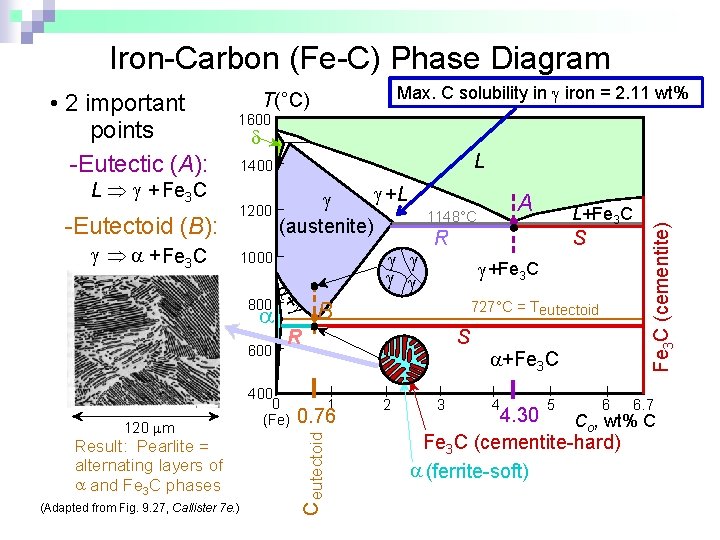
Iron-Carbon (Fe-C) Phase Diagram L Þ + Fe 3 C -Eutectoid (B): Max. C solubility in iron = 2. 11 wt% T(°C) 1600 L 1400 1200 Þ + Fe 3 C +L (austenite) + 800 600 Result: Pearlite = alternating layers of and Fe 3 C phases (Adapted from Fig. 9. 27, Callister 7 e. ) S +Fe 3 C 727°C = Teutectoid R 400 0 (Fe) S 1 0. 76 L+Fe 3 C R B C eutectoid 120 m 1000 A 1148°C 2 3 +Fe 3 C 4 5 6 Fe 3 C (cementite) • 2 important points -Eutectic (A): 6. 7 4. 30 Co, wt% C Fe 3 C (cementite-hard) (ferrite-soft)
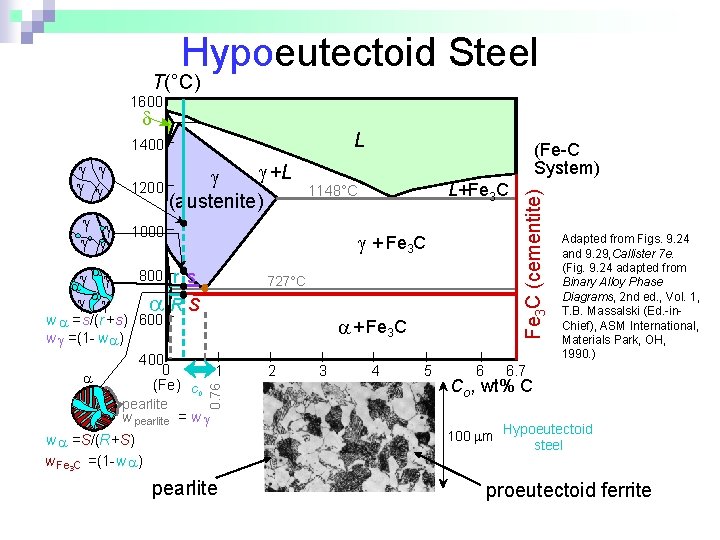
Hypoeutectoid Steel T(°C) 1600 L +L 1200 (austenite) 800 + Fe 3 C r s 727°C RS w =s/(r +s) 600 w =(1 - w ) 400 0 (Fe) pearlite L+Fe 3 C 1148°C 1000 + Fe 3 C 1 C 0 0. 76 (Fe-C System) w pearlite = w w =S/(R+S) w Fe 3 C =(1 -w ) pearlite 2 3 4 5 6 Fe 3 C (cementite) 1400 Adapted from Figs. 9. 24 and 9. 29, Callister 7 e. (Fig. 9. 24 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in. Chief), ASM International, Materials Park, OH, 1990. ) 6. 7 Co , wt% C 100 m Hypoeutectoid steel proeutectoid ferrite
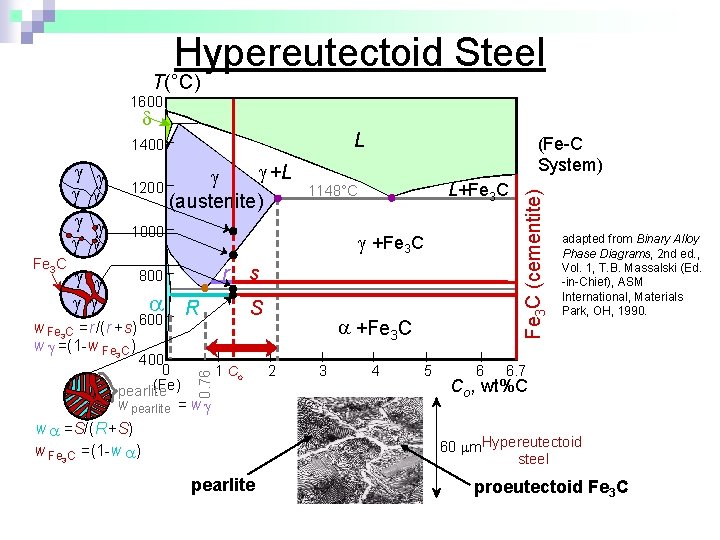
Hypereutectoid Steel T(°C) 1600 L Fe 3 C +L 1200 (austenite) 1000 r 800 w Fe 3 C =r/(r +s) w =(1 -w Fe 3 C ) R 600 400 0 (Fe) pearlite L+Fe 3 C 1148°C +Fe 3 C 0. 76 (Fe-C System) s S 1 Co w pearlite = w w =S/(R+S) w Fe 3 C =(1 -w ) +Fe 3 C 2 3 4 5 6 Fe 3 C (cementite) 1400 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in-Chief), ASM International, Materials Park, OH, 1990. 6. 7 Co , wt%C 60 m. Hypereutectoid steel pearlite proeutectoid Fe 3 C

Example: Phase Equilibria For a 99. 6 wt% Fe-0. 40 wt% C at a temperature just below the eutectoid, determine the following: a) b) c) composition of Fe 3 C and ferrite ( ) the amount of carbide (cementite) in grams that forms per 100 g of steel the amount of pearlite and proeutectoid ferrite ( )
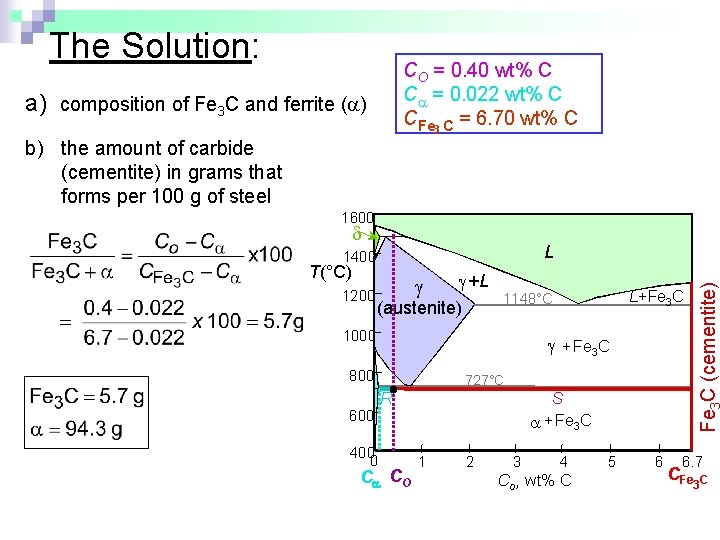
The Solution: CO = 0. 40 wt% C C = 0. 022 wt% C CFe 3 C = 6. 70 wt% C a) composition of Fe 3 C and ferrite ( ) b) the amount of carbide (cementite) in grams that forms per 100 g of steel 1600 T(°C) 1200 +L (austenite) + Fe 3 C 800 727°C R S + Fe 3 C 600 400 0 L+Fe 3 C 1148°C 1000 Fe C (cementite) L 1400 C CO 1 2 3 4 Co , wt% C 5 6 6. 7 CFe 3 C
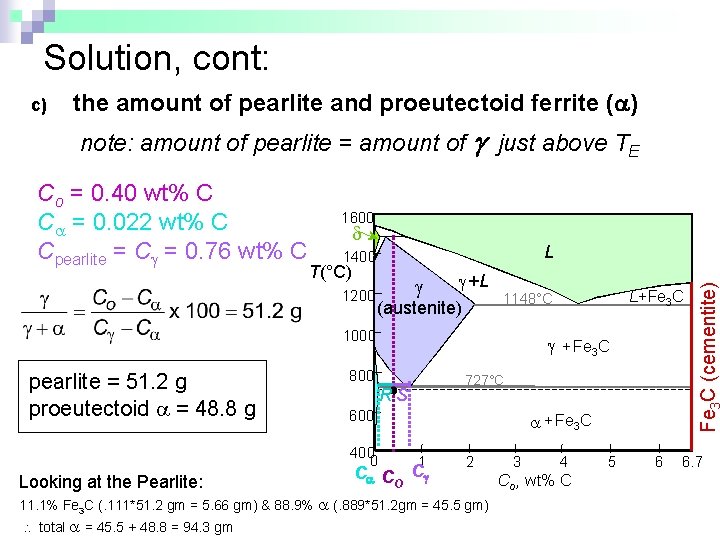
Solution, cont: c) the amount of pearlite and proeutectoid ferrite ( ) note: amount of pearlite = amount of g just above TE 1600 L 1400 T(°C) 1200 +L (austenite) 1000 pearlite = 51. 2 g proeutectoid = 48. 8 g Looking at the Pearlite: + Fe 3 C 800 727°C RS 600 400 0 + Fe 3 C 1 C CO C 2 11. 1% Fe 3 C (. 111*51. 2 gm = 5. 66 gm) & 88. 9% (. 889*51. 2 gm = 45. 5 gm) total = 45. 5 + 48. 8 = 94. 3 gm L+Fe 3 C 1148°C 3 4 Co , wt% C 5 6 Fe C (cementite) Co = 0. 40 wt% C C = 0. 022 wt% C Cpearlite = C = 0. 76 wt% C 6. 7
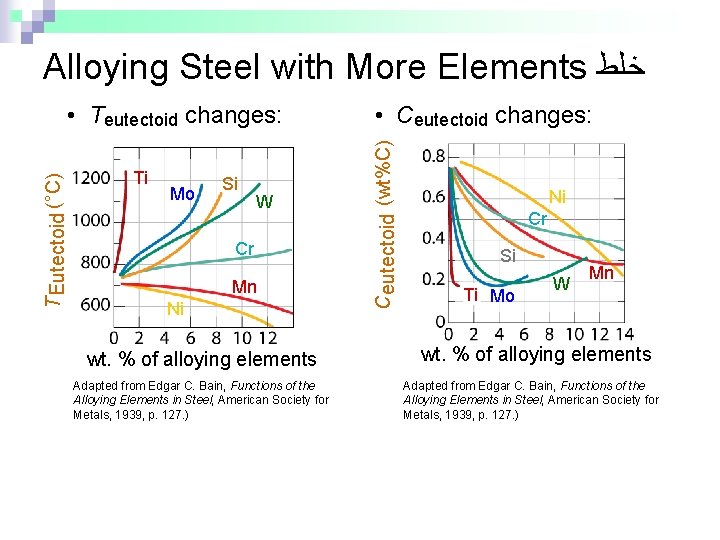
Alloying Steel with More Elements ﺧﻠﻂ Ti Mo Si W Cr Mn Ni wt. % of alloying elements Adapted from Edgar C. Bain, Functions of the Alloying Elements in Steel, American Society for Metals, 1939, p. 127. ) • Ceutectoid changes: Ceutectoid (wt%C) T Eutectoid (°C) • Teutectoid changes: Ni Cr Si Ti Mo W Mn wt. % of alloying elements Adapted from Edgar C. Bain, Functions of the Alloying Elements in Steel, American Society for Metals, 1939, p. 127. )
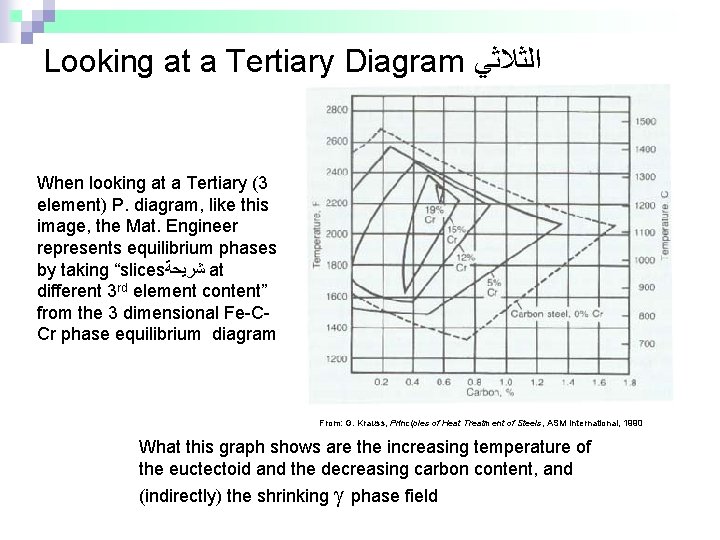
Looking at a Tertiary Diagram ﺍﻟﺜﻼﺛﻲ When looking at a Tertiary (3 element) P. diagram, like this image, the Mat. Engineer represents equilibrium phases by taking “slices ﺷﺮﻳﺤﺔ at different 3 rd element content” from the 3 dimensional Fe-CCr phase equilibrium diagram From: G. Krauss, Principles of Heat Treatment of Steels, ASM International, 1990 What this graph shows are the increasing temperature of the euctectoid and the decreasing carbon content, and (indirectly) the shrinking phase field
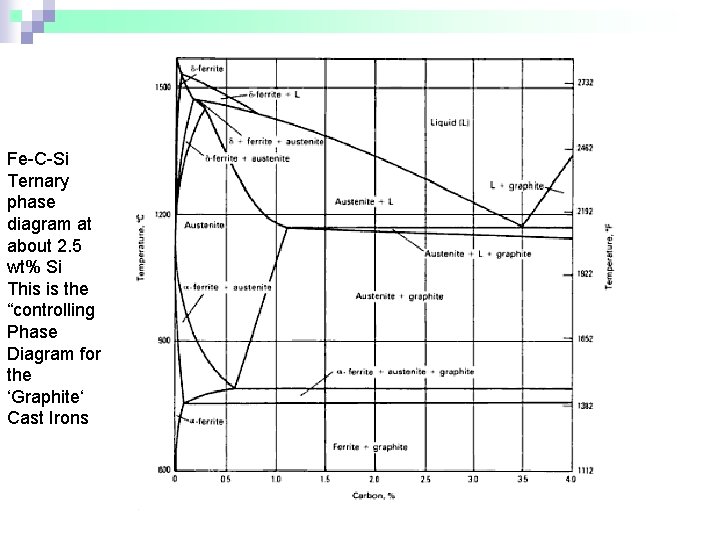
Fe-C-Si Ternary phase diagram at about 2. 5 wt% Si This is the “controlling Phase Diagram for the ‘Graphite‘ Cast Irons

Summary • Phase diagrams are useful tools to determine: 1. -- the number and types of phases, 2. -- the wt% of each phase, 3. -- and the composition of each phase for a given T and composition of the system. • Alloying to produce a solid solution usually 1. --increases the tensile strength ( ﻣﻘﺎﻭﻣﺔ ﺍﻟﺸﺪ TS) 2. --decreases the ductility ﺍﻟﻠﻴﻮﻧﺔ. • Binary ﺍ ﺍﻟﺜﻨﺎﺋﻲ eutectics and binary eutectoids allow for (cause) a range of microstructures.

Thank for all n 23/10/2013 n
- Slides: 43