Phase Equilibrium Makaopuhi Lava Lake Magma samples recovered

Phase Equilibrium

Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS Prof. Paper, 1004.
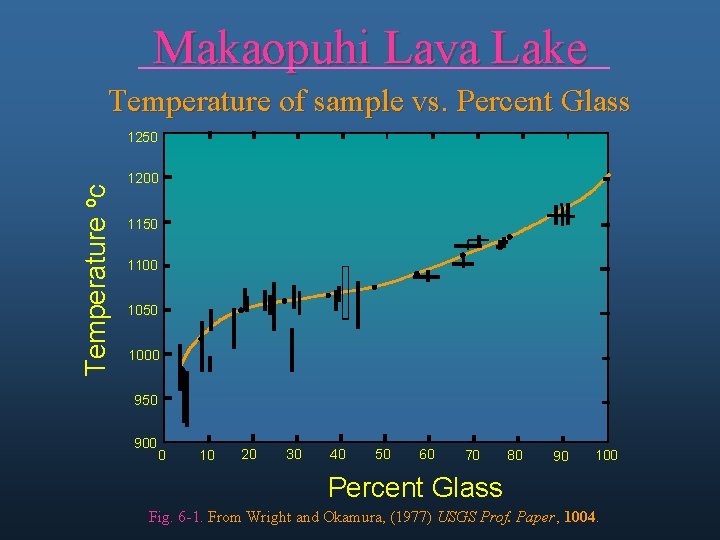
Makaopuhi Lava Lake Temperature of sample vs. Percent Glass Temperature oc 1250 1200 1150 1100 1050 1000 950 900 0 10 20 30 40 50 60 70 80 90 100 Percent Glass Fig. 6 -1. From Wright and Okamura, (1977) USGS Prof. Paper, 1004.
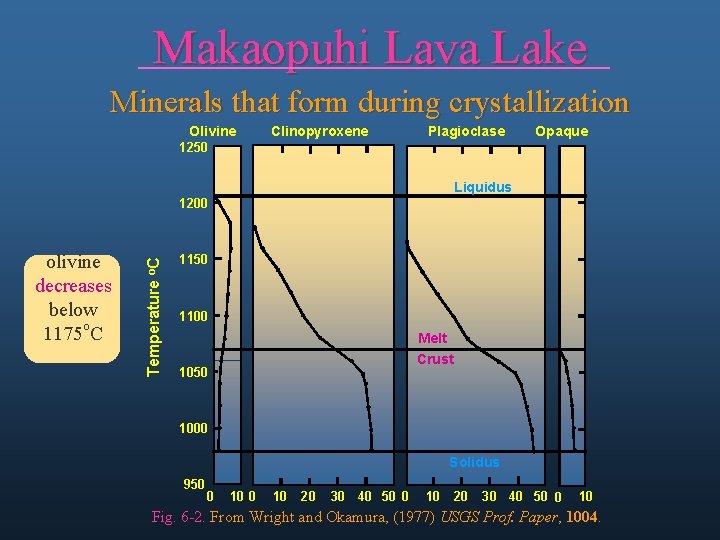
Makaopuhi Lava Lake Minerals that form during crystallization Olivine Clinopyroxene Plagioclase Opaque 1250 Liquidus olivine decreases below 1175 o. C Temperature o. C 1200 1150 1100 Melt Crust 1050 1000 Solidus 950 0 10 20 30 40 50 0 10 Fig. 6 -2. From Wright and Okamura, (1977) USGS Prof. Paper, 1004.
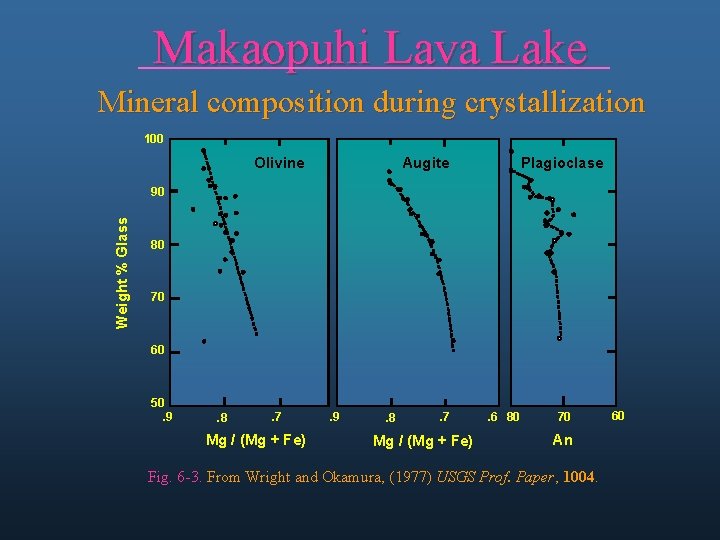
Makaopuhi Lava Lake Mineral composition during crystallization 100 Olivine Augite Plagioclase Weight % Glass 90 80 70 60 50. 9 . 8 . 7 Mg / (Mg + Fe) . 6 80 70 An Fig. 6 -3. From Wright and Okamura, (1977) USGS Prof. Paper, 1004. 60

Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals increases as T decreases 3. The minerals that form do so sequentially, with considerable overlap 4. Minerals that involve solid solution change composition as cooling progresses 5. The melt composition also changes during crystallization 6. The minerals that crystallize (as well as the sequence) depend on T and X of the melt 7. Pressure can affect the types of minerals that form and the sequence 8. The nature and pressure of the volatiles can also affect the minerals and their sequence

The Phase Rule F=C-f+2 F = # degrees of freedom The number of intensive parameters that must be specified in order to completely determine the system f = # of phases Phases are mechanically separable constituents C = minimum # of components Chemical constituents that must be specified in order to define all phases 2 = 2 intensive parameters Usually temperature and pressure for geologists
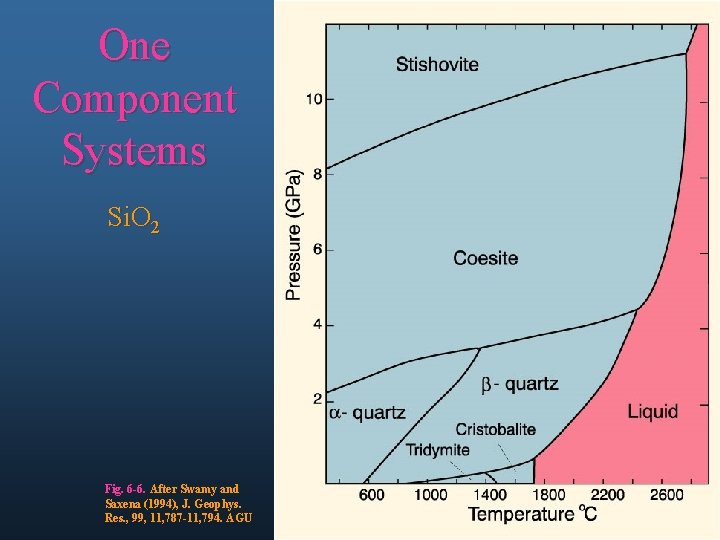
One Component Systems Si. O 2 Fig. 6 -6. After Swamy and Saxena (1994), J. Geophys. Res. , 99, 11, 787 -11, 794. AGU
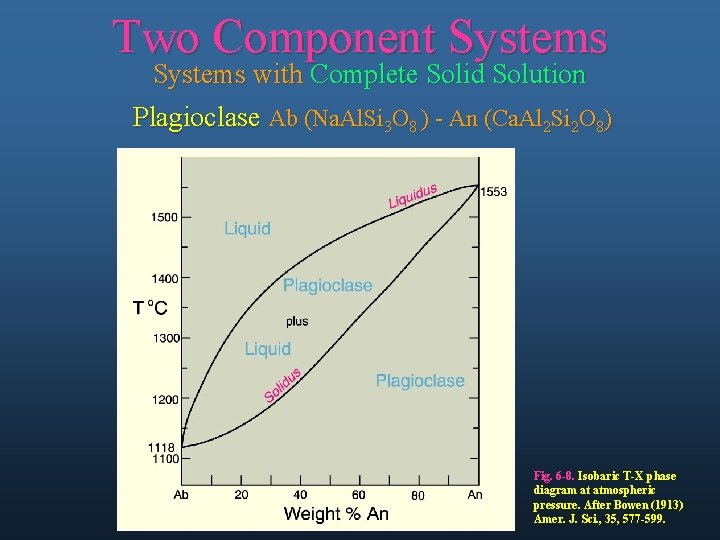
Two Component Systems with Complete Solid Solution Plagioclase Ab (Na. Al. Si 3 O 8 ) - An (Ca. Al 2 Si 2 O 8) Fig. 6 -8. Isobaric T-X phase diagram at atmospheric pressure. After Bowen (1913) Amer. J. Sci. , 35, 577 -599.
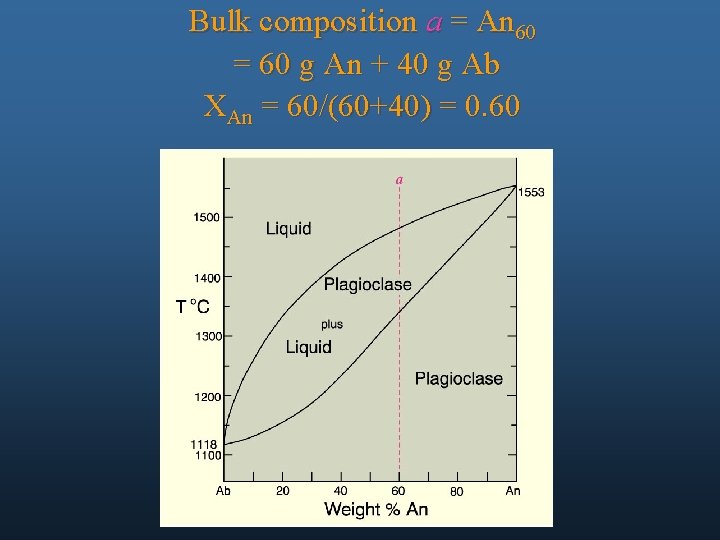
Bulk composition a = An 60 = 60 g An + 40 g Ab XAn = 60/(60+40) = 0. 60
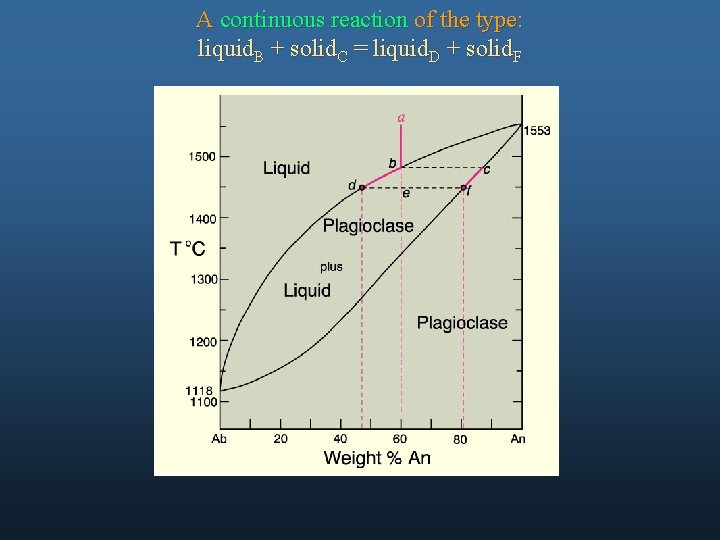
A continuous reaction of the type: liquid. B + solid. C = liquid. D + solid. F
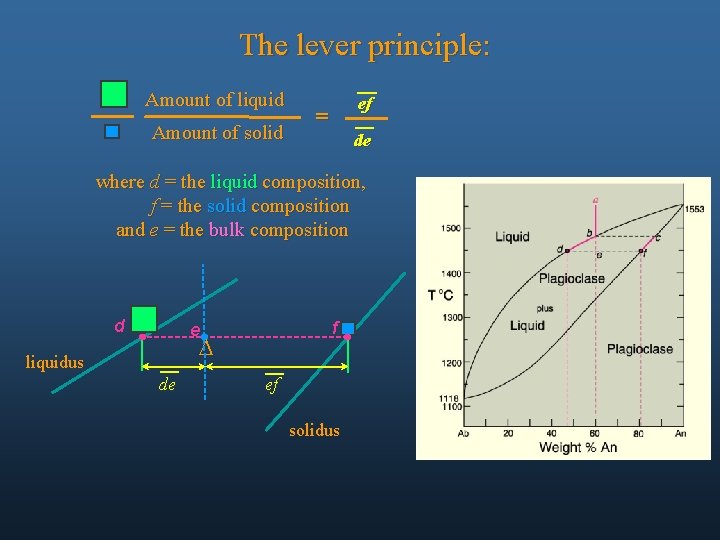
The lever principle: Amount of liquid Amount of solid ef = de where d = the liquid composition, f = the solid composition and e = the bulk composition d f e D liquidus de ef solidus
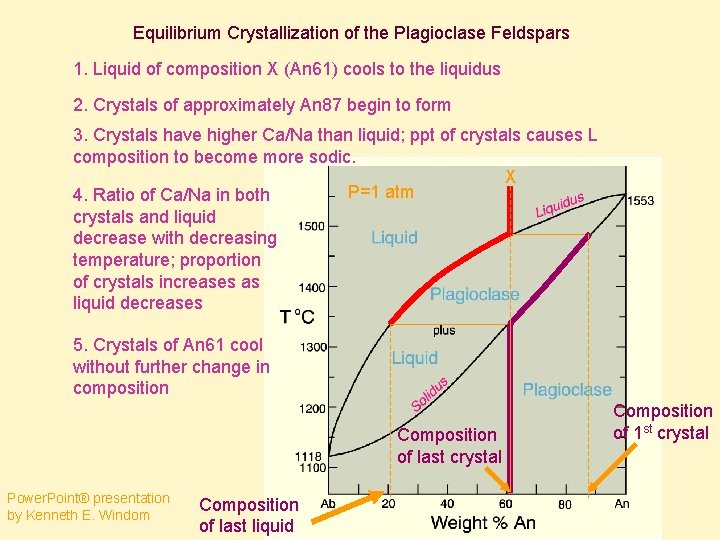
Equilibrium Crystallization of the Plagioclase Feldspars 1. Liquid of composition X (An 61) cools to the liquidus 2. Crystals of approximately An 87 begin to form 3. Crystals have higher Ca/Na than liquid; ppt of crystals causes L composition to become more sodic. X P=1 atm 4. Ratio of Ca/Na in both crystals and liquid decrease with decreasing temperature; proportion of crystals increases as liquid decreases 5. Crystals of An 61 cool without further change in composition Composition of last crystal Power. Point® presentation by Kenneth E. Windom Composition of last liquid Composition of 1 st crystal
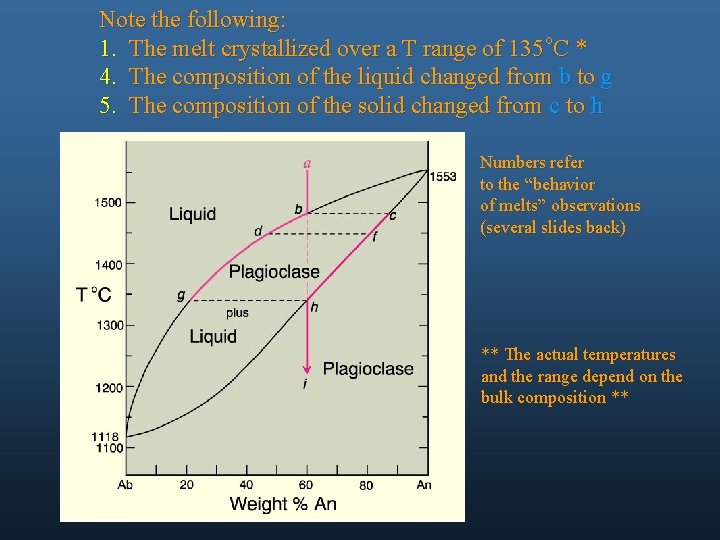
Note the following: 1. The melt crystallized over a T range of 135 o. C * 4. The composition of the liquid changed from b to g 5. The composition of the solid changed from c to h Numbers refer to the “behavior of melts” observations (several slides back) ** The actual temperatures and the range depend on the bulk composition **
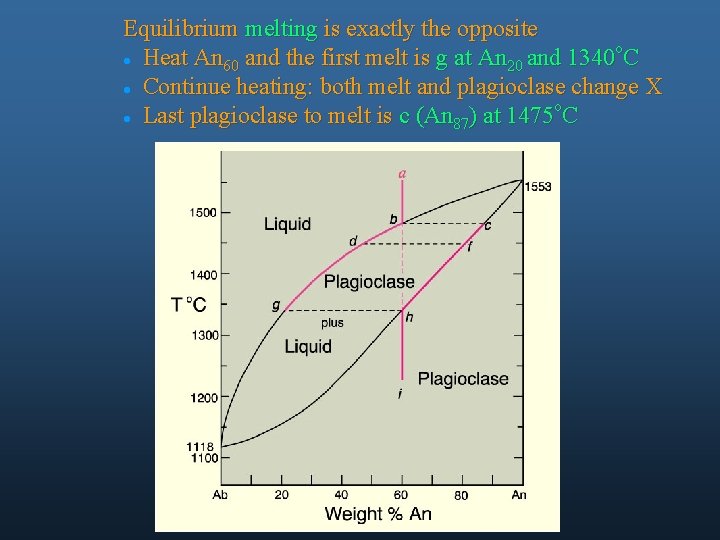
Equilibrium melting is exactly the opposite o l Heat An and the first melt is g at An and 1340 C 60 20 l Continue heating: both melt and plagioclase change X o l Last plagioclase to melt is c (An ) at 1475 C 87
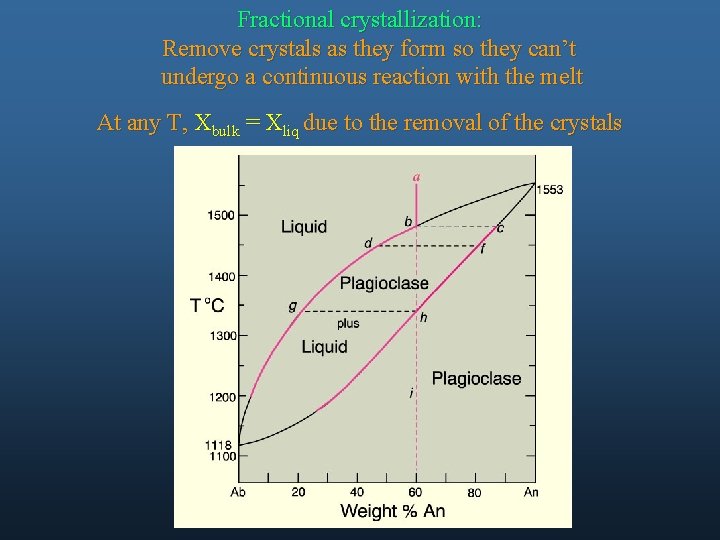
Fractional crystallization: Remove crystals as they form so they can’t undergo a continuous reaction with the melt At any T, Xbulk = Xliq due to the removal of the crystals
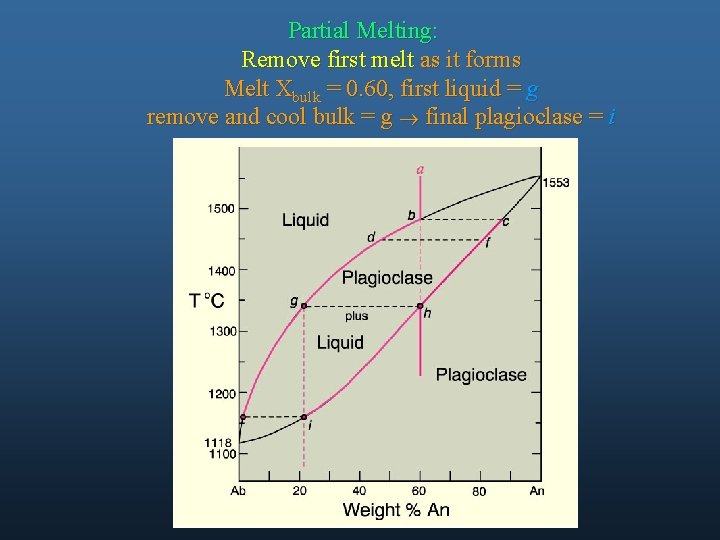
Partial Melting: Remove first melt as it forms Melt Xbulk = 0. 60, first liquid = g remove and cool bulk = g ® final plagioclase = i
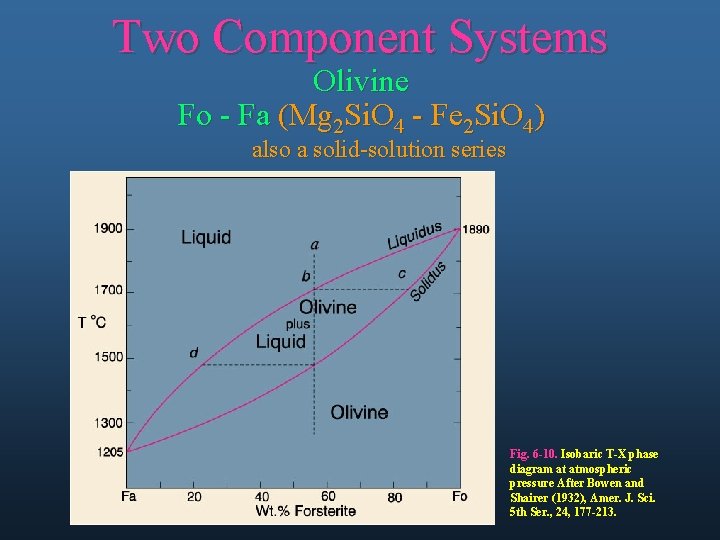
Two Component Systems Olivine Fo - Fa (Mg 2 Si. O 4 - Fe 2 Si. O 4) also a solid-solution series Fig. 6 -10. Isobaric T-X phase diagram at atmospheric pressure After Bowen and Shairer (1932), Amer. J. Sci. 5 th Ser. , 24, 177 -213.
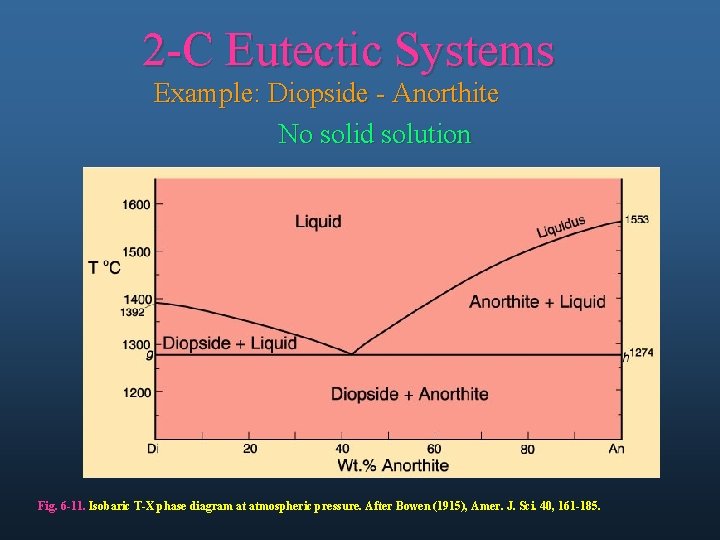
2 -C Eutectic Systems Example: Diopside - Anorthite No solid solution Fig. 6 -11. Isobaric T-X phase diagram at atmospheric pressure. After Bowen (1915), Amer. J. Sci. 40, 161 -185.
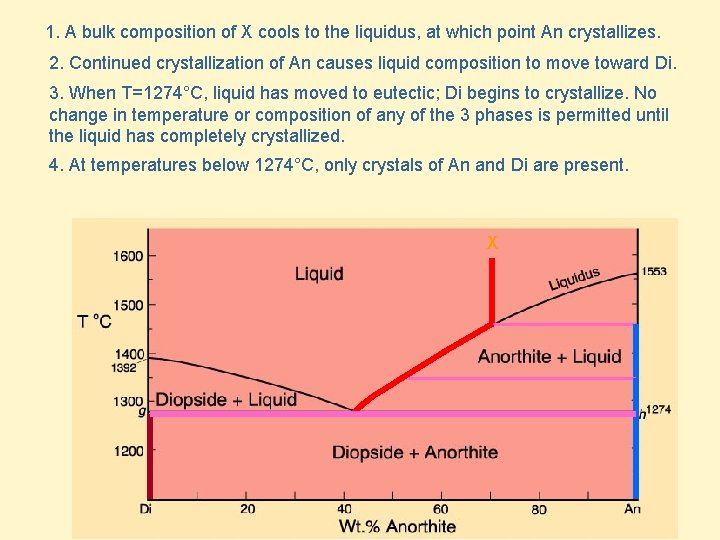
1. A bulk composition of X cools to the liquidus, at which point An crystallizes. 2. Continued crystallization of An causes liquid composition to move toward Di. 3. When T=1274°C, liquid has moved to eutectic; Di begins to crystallize. No change in temperature or composition of any of the 3 phases is permitted until the liquid has completely crystallized. 4. At temperatures below 1274°C, only crystals of An and Di are present. X
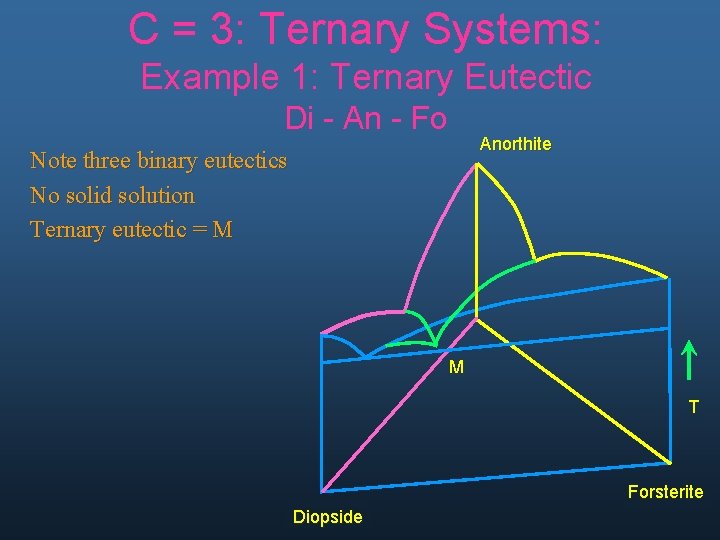
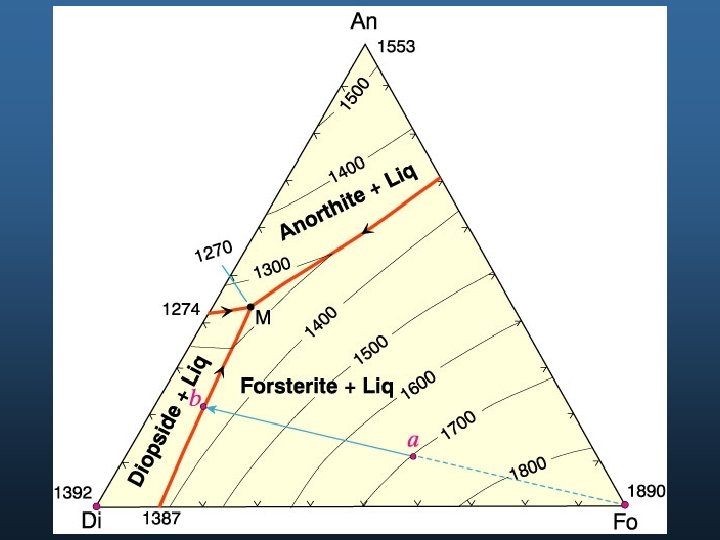
C = 3: Ternary Systems: Example 1: Ternary Eutectic Di - An - Fo Anorthite Note three binary eutectics No solid solution Ternary eutectic = M M T Forsterite Diopside

Crystallization Behavior of Melts 1. Cooling melts crystallize from a liquid to a solid over a range of temperatures (and pressures) 2. Several minerals crystallize over this T range, and the number of minerals increases as T decreases 3. The minerals that form do so sequentially, with considerable overlap 4. Minerals that involve solid solution change composition as cooling progresses 5. The melt composition also changes during crystallization 6. The minerals that crystallize (as well as the sequence) depend on T and X of the melt 7. Pressure can affect the types of minerals that form and the sequence 8. The nature and pressure of the volatiles can also affect the minerals and their sequence
- Slides: 23