Phase Equilibrium B Sc II year Physical Chemistry

Phase Equilibrium B. Sc. II year Physical Chemistry By Prof. M. K. Bhatnagar Pandit S. N. Shukla University Shahdol (M. P. )

Two Component System

Component system Two Component • • Phase rule : F=C-P+2 Here, C=2 and P=1 => F= 2 -1+2 = 3 We require three dimension diagram with three variables. To draw planar diagram, one variable must be constant. So, we use condensed phase rule. F = C – P + 1

Reason for Condensed Phase Rule • Phase rule : F=C-P+2 • For two component system, C=2 • P=4 (four phases) must be present for zero degree of freedom. So, we reduce one variable and use condensed phase rule. • F=C-P+1
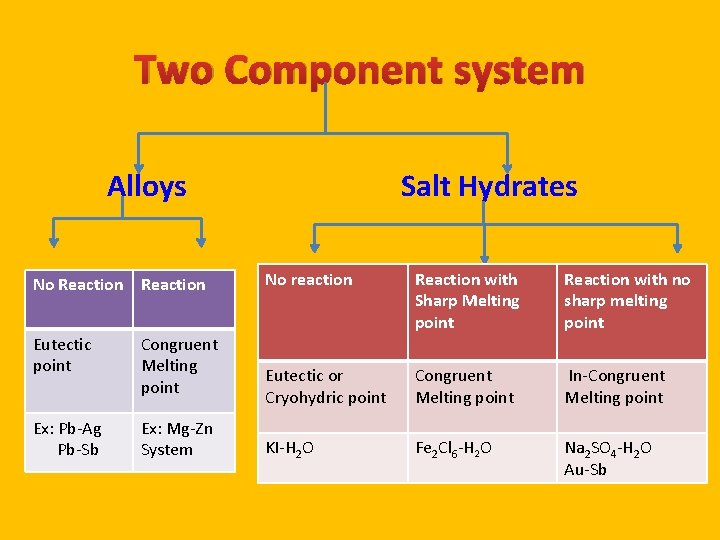
Two Component system Alloys No Reaction Eutectic point Congruent Melting point Ex: Pb-Ag Pb-Sb Ex: Mg-Zn System Salt Hydrates No reaction Reaction with Sharp Melting point Reaction with no sharp melting point Eutectic or Cryohydric point Congruent Melting point In-Congruent Melting point KI-H 2 O Fe 2 Cl 6 -H 2 O Na 2 SO 4 -H 2 O Au-Sb

Definations • Eutectic Point: A eutectic system is a mixture of chemical compounds or elements that has a single chemical composition that solidifies at a lower temperature than any other composition. the temperature at which a particular eutectic mixture freezes or melts. अभ नन गलन क – द ततव एक न शच त सधरत पर एक स थ प घलत ह जबक उनक गलन क अलग ह त ह अलग

Definitions • Congruent Point: When the solid compound AB and the liquid phase have identical composition at the maximum point on the freezing point curve, the corresponding temperature is said to be the congruent melting point of the compound. सरव ग सम गलन क ब द – द ततव स पर पत नय पद रथ ज ठ स और दरव अवसथ म एक सम न सधरत दरश त ह एव एक ह त प पर प घलत ह.

Definitions • Incongruent melting: In-Congruent Point occurs when a substance does not melt uniformly and decomposes into another substance. असरव ग सम गलन क ब द – पद रथ ज गलन क क एकरपत नह दरश त एव एक न शच त त प पर व घट त ह ज त ह.

Silver Lead – Two Component Alloy System • • • Phases Ag Solid Pb Solid Solution Vapours • Effect of prassure is negligable so we ignore vapours and draw T-C Diagram. • And Use Condensed phase Rule F = C – P + 1
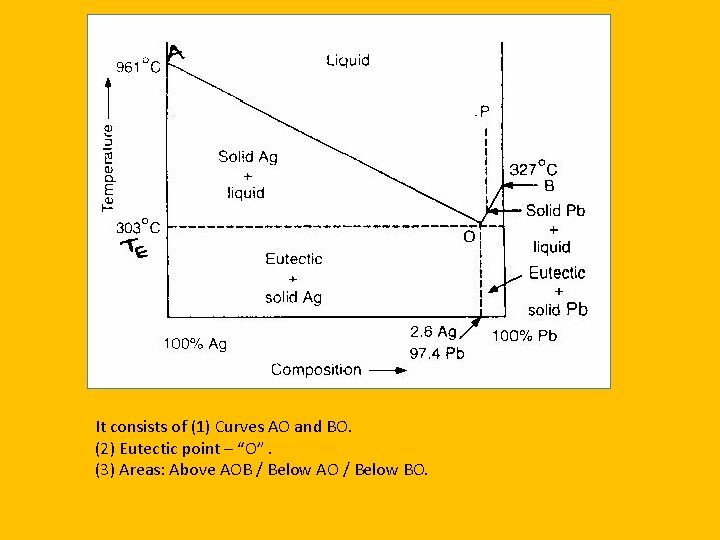
It consists of (1) Curves AO and BO. (2) Eutectic point – “O”. (3) Areas: Above AOB / Below AO / Below BO.
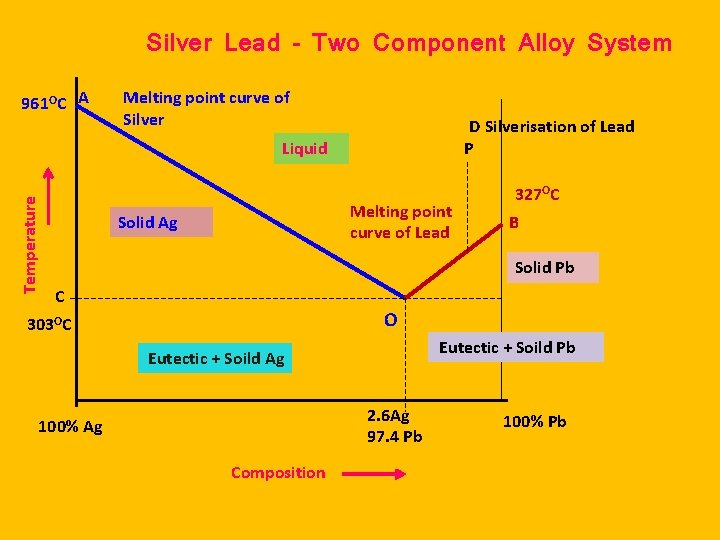
Silver Lead – Two Component Alloy System 961 OC A Melting point curve of Silver D Silverisation of Lead P Temperature Liquid Melting point curve of Lead Solid Ag 327 OC B Solid Pb C O 303 OC Eutectic + Soild Pb Eutectic + Soild Ag 2. 6 Ag 97. 4 Pb 100% Ag Composition 100% Pb

CURVE BO It is the freezing point curve of Pb (327 o. C) It shows decrease in freezing point / melting point of Pb due to the addition of Ag to Pb. Solid Pb is equilibrium with solution of Ag in Pb. F = C – P + 1 F = 2 – 2 + 1 = 1 Uni. Variant

CURVE AO It is the freezing point curve of Ag (961 o. C) It shows decrease in freezing point / melting point of Ag due to the addition of Pb to Ag. Solid Ag is equilibrium with solution of Pb in Ag F = C – P + 1 F = 2 – 2 + 1 = 1 Uni. Variant

Eutectic point ‘O’: The curves AO & BO meet at the point O is called eutectic point. Here, solid Ag, Solid Pb and solution of Ag & Pb are in equilibrium. Thus, C = 2 and P = 3. Hence the reduced phase rule is F = C – P + 1 = 2 – 3 + 1 = 0 and the system is invariant. The point O (303 o. C) represents a fixed composition of 97. 4 % Pb and 2. 6 % Ag, and is called eutectic composition.

Area above AOB: The components Ag and Pb are exist as solution. Thus, C = 2 and P = 1. Hence f = C – P + 1 = 2 – 1 + 1 = 2, the system is bivariant. The system will exist when the temperature T > 303 o. C , Pb < 97. 4 % and Ag > 2. 6 %.
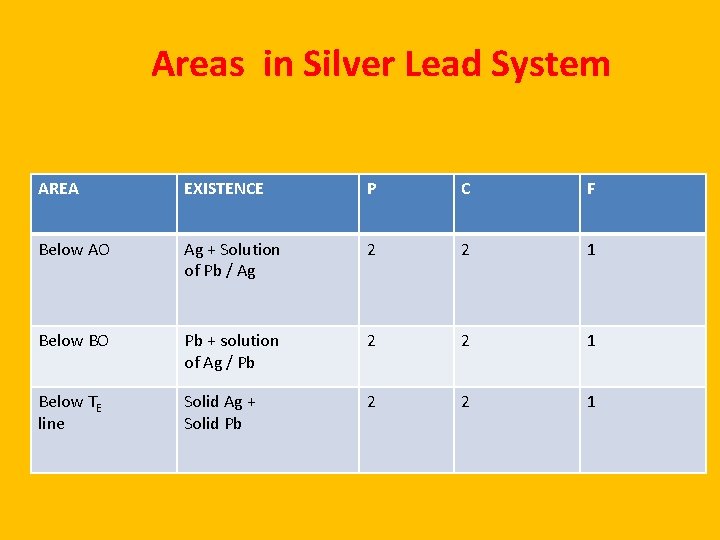
Areas in Silver Lead System AREA EXISTENCE P C F Below AO Ag + Solution of Pb / Ag 2 2 1 Below BO Pb + solution of Ag / Pb 2 2 1 Below TE line Solid Ag + Solid Pb 2 2 1

De-silvarisation of Lead Pattinson Process Amount of silver is very less in Argentoferous lead. By keeping temperature 303 and adding lead Percentage of silver increases up to 2. 4% By Cupal ( Boan ash crucible ) this amount can be adsorbe and materlargy of silver done in Pattinson process. This is important application of silver lead system.

Salt hydrate Potassiom iodide – water system with eutectic point 1. Draw Phase rule diagram a. Melting point of water is Zero degree b. Eutactic point or Chryo (hydridc ) point on -21 degree. c. Melting point of KI is above 200 degree. Q. 1. Name the curves and points and areas. Q. 2. Specify degree of freedom for curve and point. Q. 3. Give Variable which is more effective for the given system
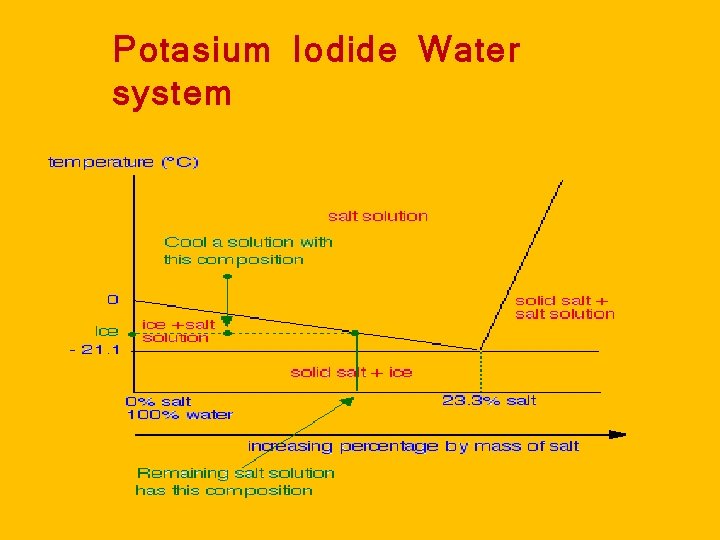
Potasium Iodide Water system

Two Component System Congruent melting Point Zn- Mg System

Phases 1. Solid Zn 2. Solid Mg 3. Mg(Zn)2 4. Solution 5. Vapours It is neglected due to constant pressure

Congruent Melting Point Congruent Point: When the solid compound AB and the liquid phase have identical composition at the maximum point on the freezing point curve, the corresponding temperature is said to be the congruent melting point of the compound. Zn and Mg react and form a new compound Mg(Zn)2 Which is stable and melt at temperature at 590. It is stable in solid as well as liquid form सरव गसम गलन क ब द - Zn एव आपसम कर य कर नय पद रथ Mg(Zn)2 Mg बन त ह ज ठ स एवम दरव अवसथ म सथ ई ह त ह इसल य यह ततर सरव ग सम गलन क क ततर कहल त ह
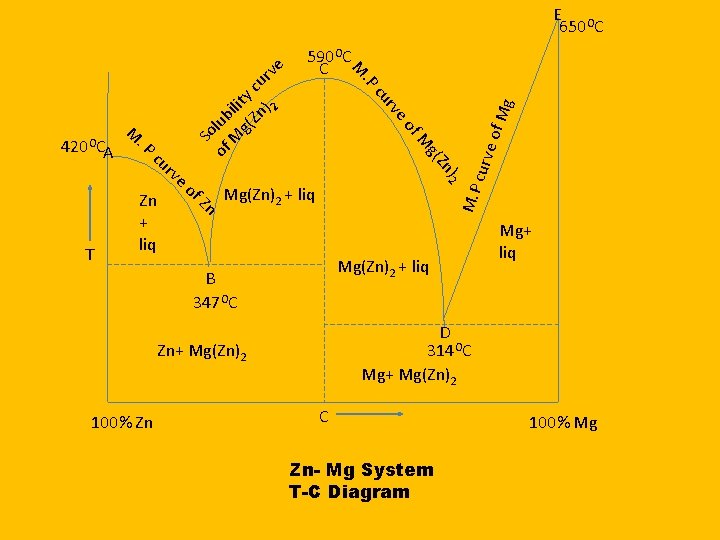
E O 650 C Mg Mg(Zn)2 + liq B 347 OC Mg+ liq D 314 OC Mg+ Mg(Zn)2 Zn+ Mg(Zn)2 100% Zn e of M. P Zn Mg(Zn)2 + liq curv )2 Zn of g( M Zn + liq rv e of cu ve T . P ur A M c. P 420 OC ur c y lit n) 2 i ub g(Z l So f M o 590 OC C M ve C Zn- Mg System T-C Diagram 100% Mg
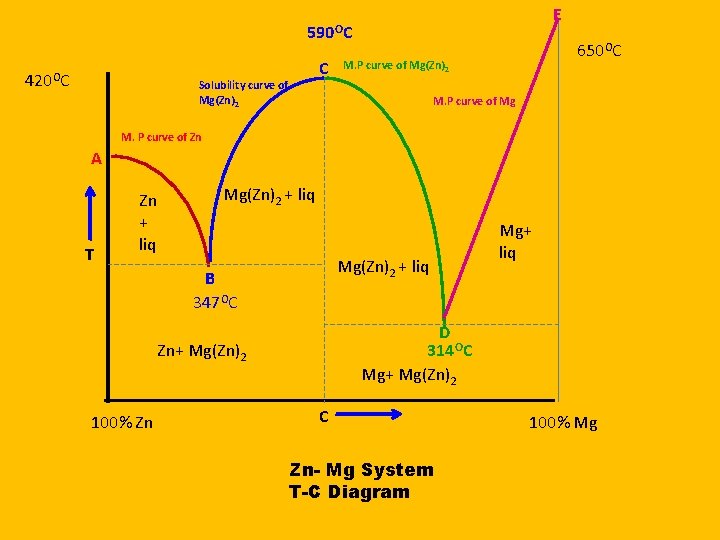
E 590 OC 420 OC Solubility curve of Mg(Zn)2 C 650 OC M. P curve of Mg(Zn)2 M. P curve of Mg M. P curve of Zn A T Zn + liq Mg(Zn)2 + liq B 347 OC D 314 OC Mg+ Mg(Zn)2 Zn+ Mg(Zn)2 100% Zn Mg+ liq C Zn- Mg System T-C Diagram 100% Mg
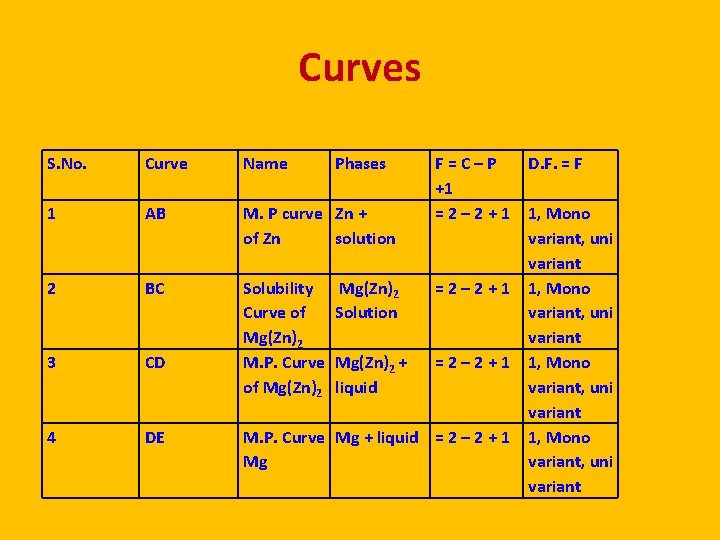
Curves S. No. Curve Name Phases 1 AB M. P curve Zn + of Zn solution 2 BC 3 CD Solubility Curve of Mg(Zn)2 M. P. Curve of Mg(Zn)2 4 DE F = C – P +1 = 2 – 2 + 1 Mg(Zn)2 Solution = 2 – 2 + 1 Mg(Zn)2 + liquid = 2 – 2 + 1 M. P. Curve Mg + liquid = 2 – 2 + 1 Mg D. F. = F 1, Mono variant, uni variant
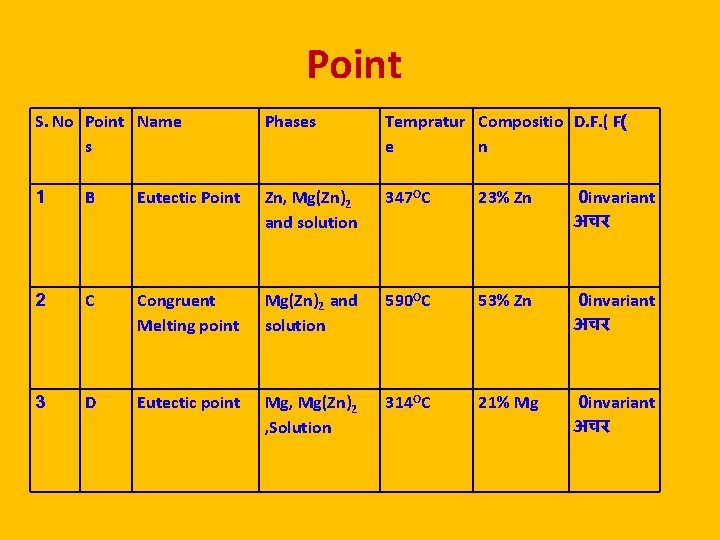
Point S. No Point Name s Phases Tempratur Compositio D. F. ( F( e n 1 B Eutectic Point Zn, Mg(Zn)2 and solution 347 OC 23% Zn 0 invariant अचर 2 C Congruent Melting point Mg(Zn)2 and solution 590 OC 53% Zn 0 invariant अचर 3 D Eutectic point Mg, Mg(Zn)2 , Solution 314 OC 21% Mg 0 invariant अचर
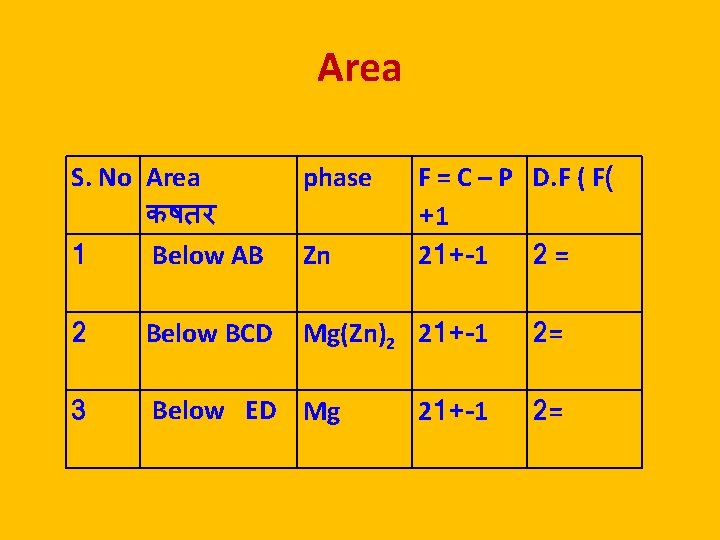
Area S. No Area कषतर 1 Below AB phase 2 Below BCD Mg(Zn)2 21+-1 3 Below ED Mg Zn F = C – P D. F ( F( +1 21+-1 2 = 21+-1 2= 2=

Salt Hydrate System with Congruent melting Point Ferric Chloride – water System
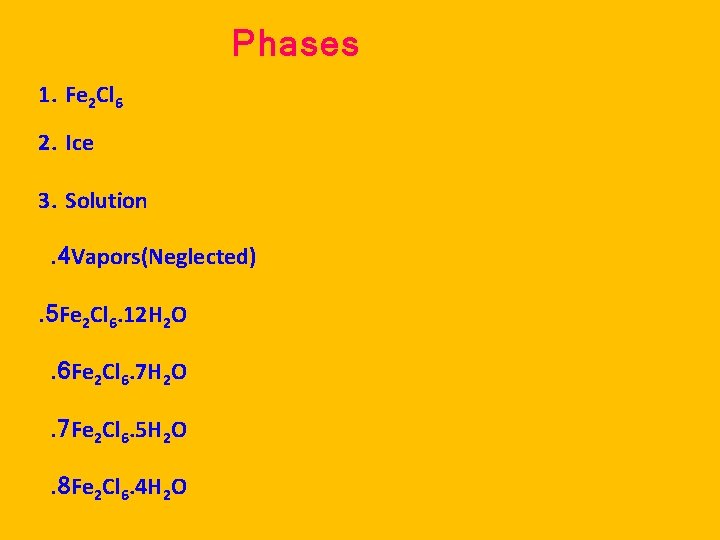
Phases 1. Fe 2 Cl 6 2. Ice 3. Solution. 4 Vapors(Neglected). 5 Fe 2 Cl 6. 12 H 2 O. 6 Fe 2 Cl 6. 7 H 2 O. 7 Fe 2 Cl 6. 5 H 2 O. 8 Fe 2 Cl 6. 4 H 2 O
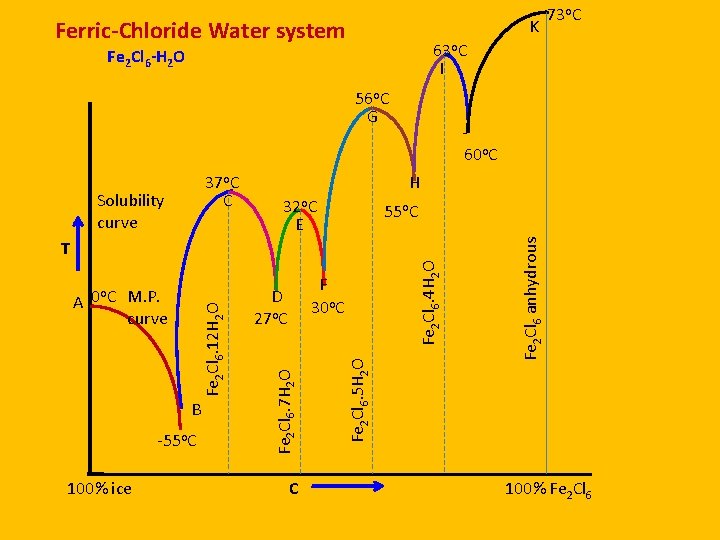
Ferric-Chloride Water system K 63 o. C I Fe 2 Cl 6 -H 2 O 56 o. C G Solubility curve J 60 o. C H 32 o. C E 55 o. C -55 o. C 100% ice C Fe 2 Cl 6. 5 H 2 O B F 30 o. C D 27 o. C Fe 2 Cl 6. 7 H 2 O Fe 2 Cl 6. 12 H 2 O o A 0 C M. P. curve Fe 2 Cl 6. 4 H 2 O T Fe 2 Cl 6 anhydrous 37 o. C C 73 o. C 100% Fe 2 Cl 6

Curves A. Melting point Curves 1. AB- Ice 2. CD-Fe 2 Cl 6. 12 H 2 O 3. EF-Fe 2 Cl 6. 7 H 2 O 4. GH-Fe 2 Cl 6. 5 H 2 O 5. IJ-Fe 2 Cl 6. 4 H 2 O F=1 univariant B. Solubility Curves 1. BC-Fe 2 Cl 6. 12 H 2 O 2. DE-Fe 2 Cl 6. 7 H 2 O 3. EG-Fe 2 Cl 6. 5 H 2 O 4. HI-Fe 2 Cl 6. 4 H 2 O 5. JK-Fe 2 Cl 6 anhydrous

Points A. Eutectic or cryohydric points 1. B-ice-Soln. Fe 2 Cl 6. 12 H 2 O 2. D-Fe 2 Cl 6. 12 H 2 O-Soln. Fe 2 Cl 6. 7 H 2 O 3. F-Fe 2 Cl 6. 7 H 2 O-Soln. Fe 2 Cl 6. 5 H 2 O 4. H-Fe 2 Cl 6. 5 H 2 O-Soln. Fe 2 Cl 6. 4 H 2 O 5. J-Fe 2 Cl 6. 4 H 2 O-Soln. Fe 2 Cl 6 B. Congruent M. P. 1. C-Fe 2 Cl 6. 12 H 2 O 2. E-Fe 2 Cl 6. 7 H 2 O 3. G-Fe 2 Cl 6. 5 H 2 O 4. I-Fe 2 Cl 6. 4 H 2 O 5. K-Fe 2 Cl 6 anhydrous


Expected Questions Q. 1. Define a. Eutectic Point. b. Congrueny melting point c. Incongruent melting point Q. 2. Draw a Phase diagram of two component system which form Salt Hydrate. Q. 3. Give application of Phase rule in Extraction of mettles. Q. 4. Explain Dsilversiation of Lead.

Thanks Prof. M. K. Bhatnagar Pandit S. N. Shukla University Shahdol (M. P. ) Email- drmahendrab 15@gmail. com
- Slides: 35