Phase Equilibria EvaporationCondensation MeltingFreezing Phase transition SublimationCondensation Sg

Phase Equilibria Evaporation-Condensation Melting-Freezing Phase transition Sublimation-Condensation
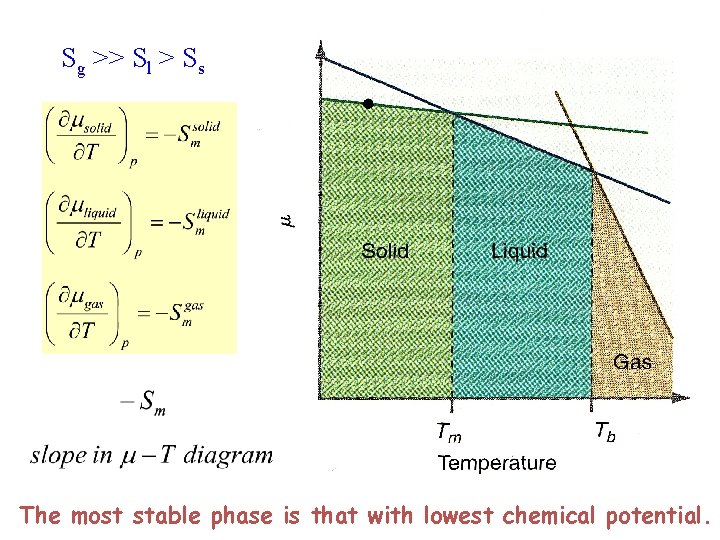
Sg >> Sl > Ss The most stable phase is that with lowest chemical potential.
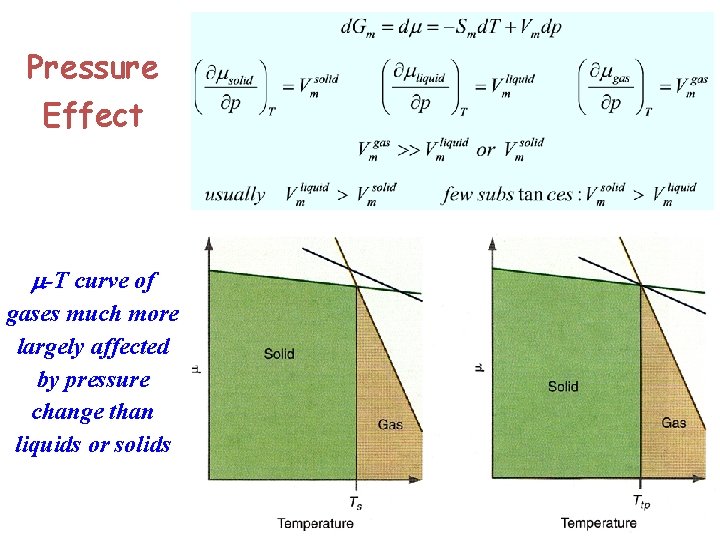
Pressure Effect m-T curve of gases much more largely affected by pressure change than liquids or solids
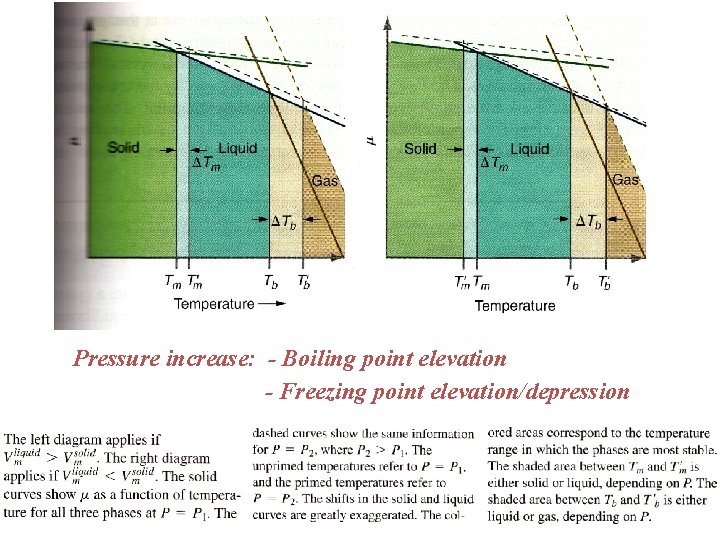
Pressure increase: - Boiling point elevation - Freezing point elevation/depression
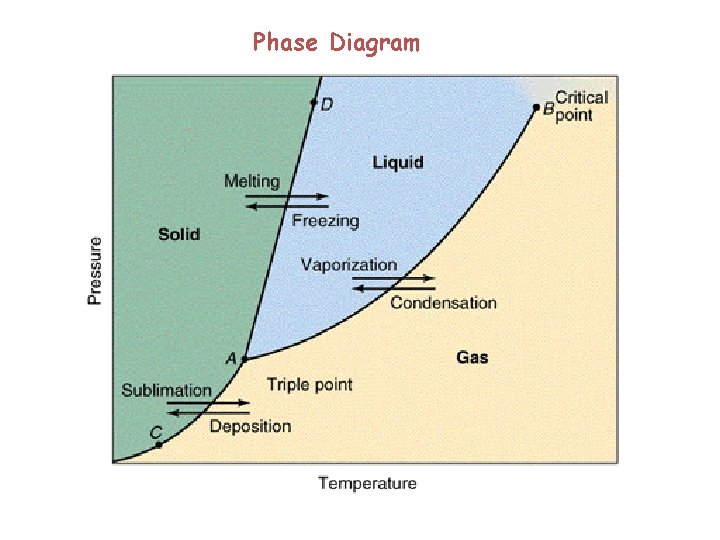
Phase Diagram
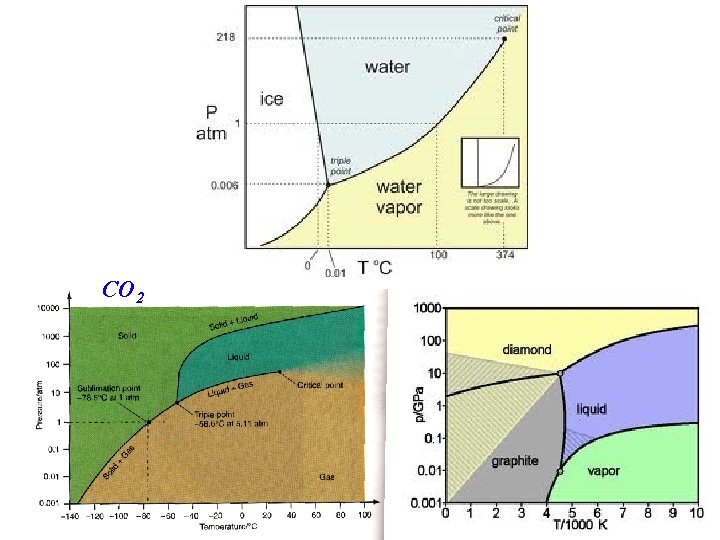
CO 2

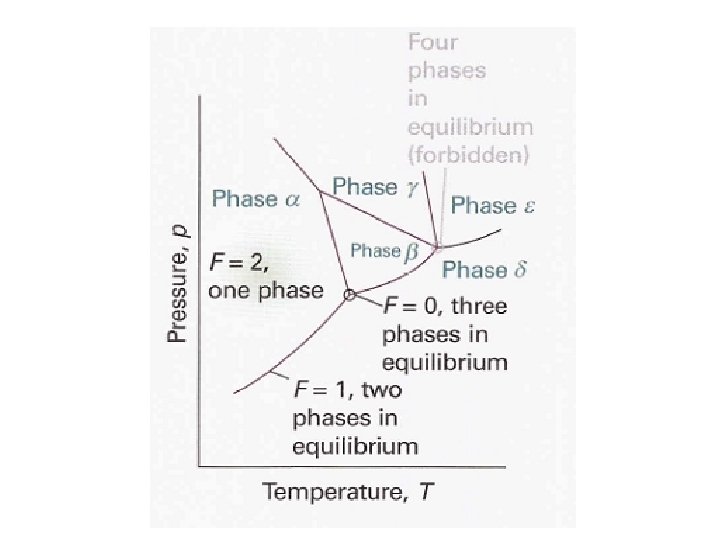
Clapeyron Equation At Equilibrium Clapeyron Equation

Solid-Liquid Equilibrium Slope of p. T-curve § Usually positive § ~ 40 atm/K § 40 atm are needed to change the melting point by 1 K
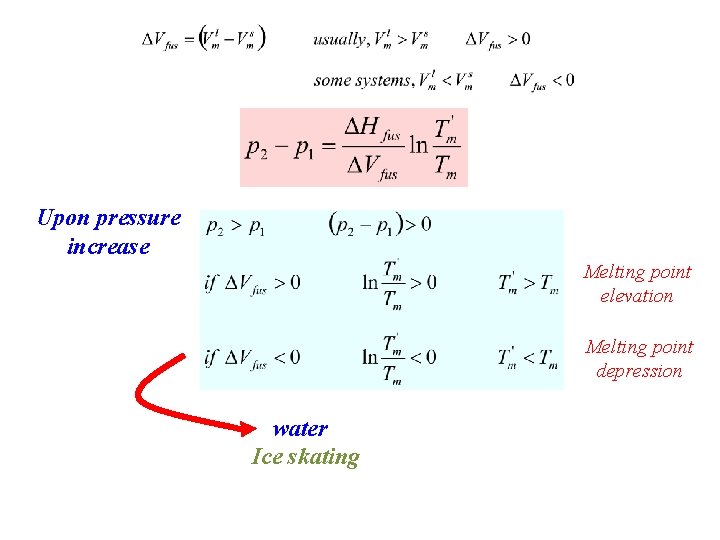
Upon pressure increase Melting point elevation Melting point depression water Ice skating

if pressure is changed by Dp, the melting point will change by DTm

Liquid-Gas Equilibrium Slope of p. T-curve § always positive § ~ 0. 04 atm/K § Boiling point increases by 25 K upon increasing the pressure by 1 atm. applies also to s-g equilibrium Clausius-Clapeyron Equation
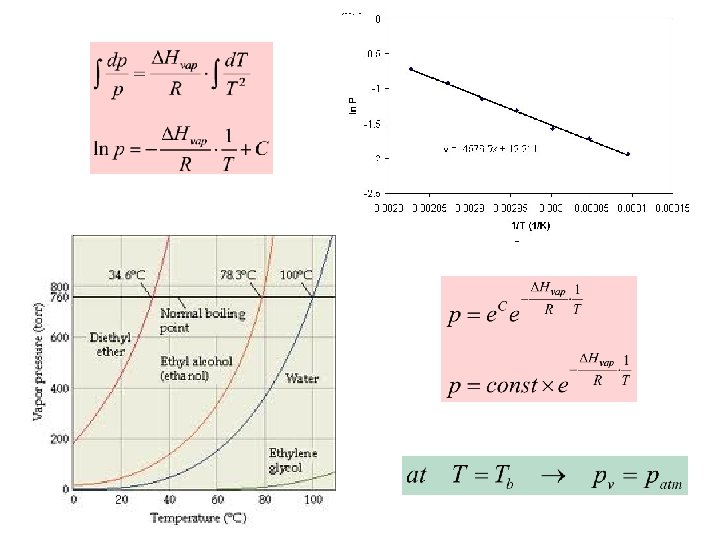

Determine the change in the freezing point of ice upon pressure increase from 1 atm to 2 atm. Vm(water)=18. 02 cm 3/mol and Vm(ice)=19. 63 cm 3/mol at 273. 15 K. DHfus=6. 009 k. J/mol. Benzene has a normal boiling point of 353. 25 K. If benzene is to be boiled at 30 o. C, to what value must the pressure be lowered. DHvap=30. 76 k. J/mol


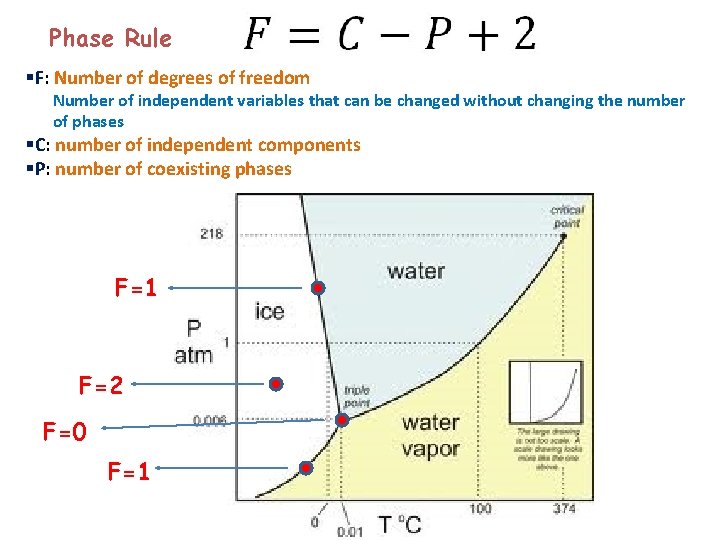
Phase Rule §F: Number of degrees of freedom Number of independent variables that can be changed without changing the number of phases §C: number of independent components §P: number of coexisting phases F=1 F=2 F=0 F=1

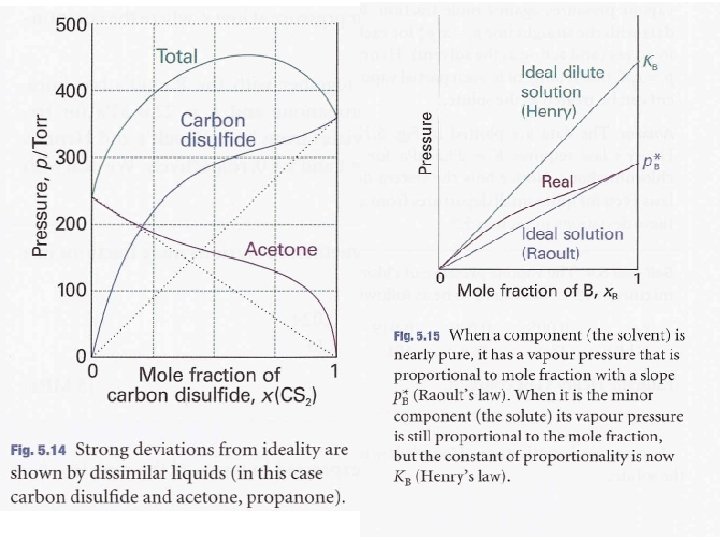
Liquid-Gas Equilibrium of a binary mixture Ideal solution: Energy of interaction AA, BB = A-B Intramolecular forces AA, BB = A-B Ideal solutions obey Raoults Law
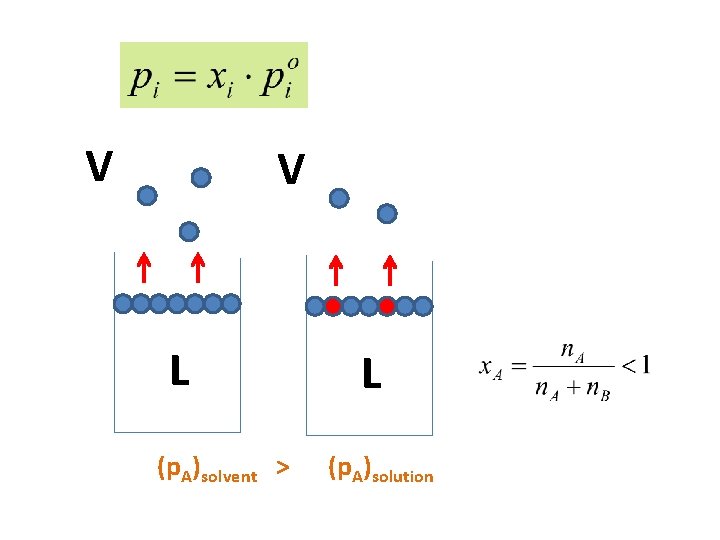
V V L (p. A)solvent > L (p. A)solution
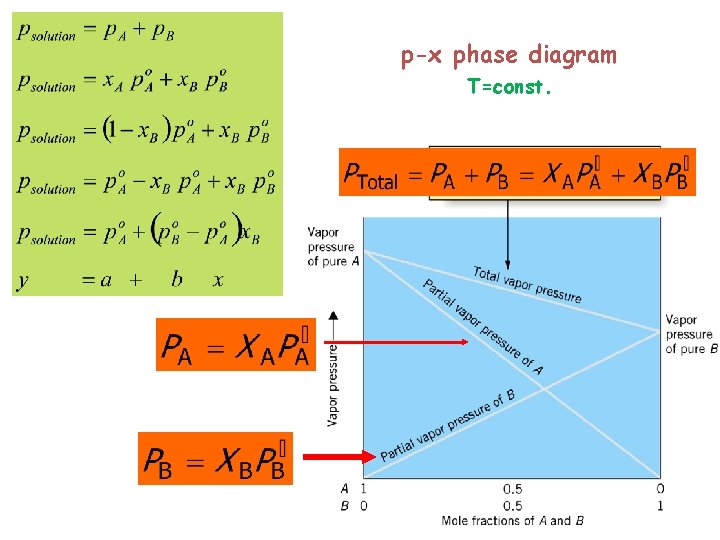
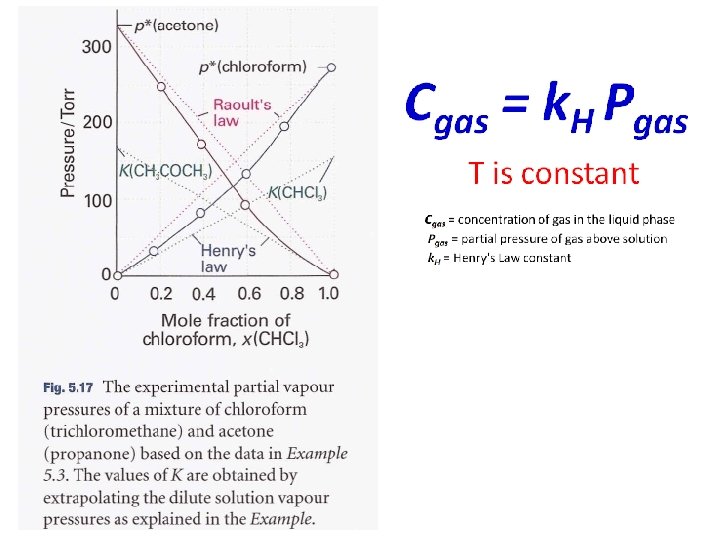
p-x phase diagram T=const.
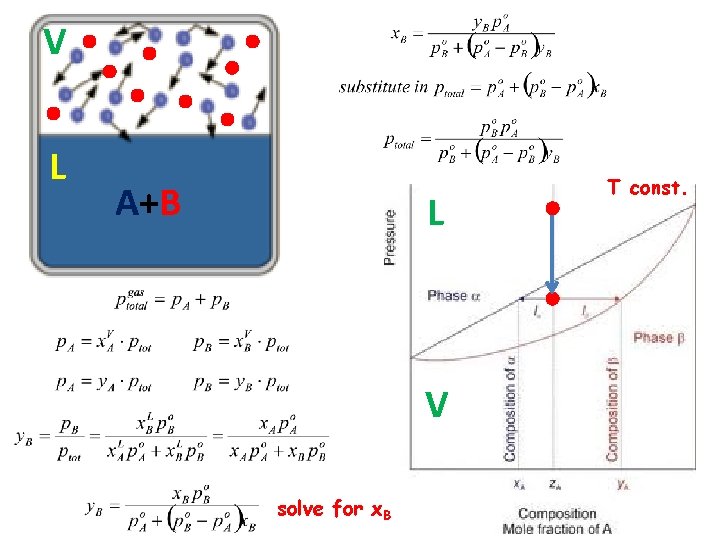
V L A+B L V solve for x. B T const.

Ex. Benzene and Toluene • Consider a mixture of benzene, C 6 H 6, and toluene, C 7 H 8, containing 1. 0 mol benzene and 2. 0 mol toluene. At 20 °C, the vapor pressures of the pure substances are: P°benzene = 75 torr P°toluene = 22 torr • Assuming the mixture obeys Raoult’s law, what is the total pressure above this solution? 23
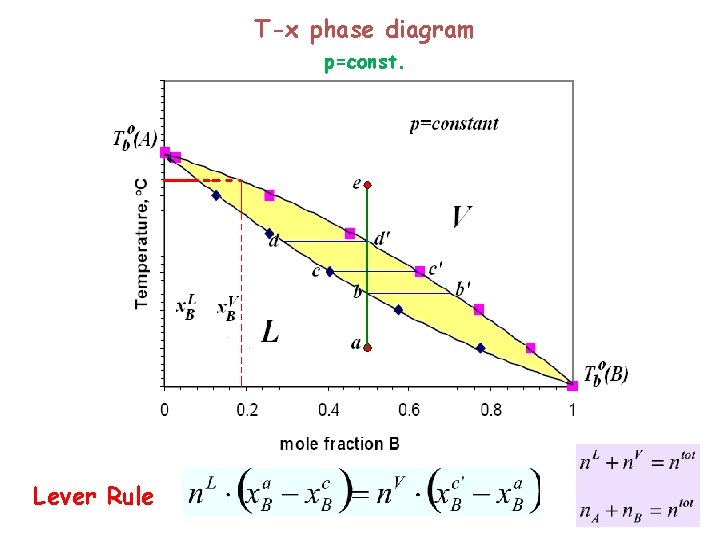
T-x phase diagram p=const. Lever Rule
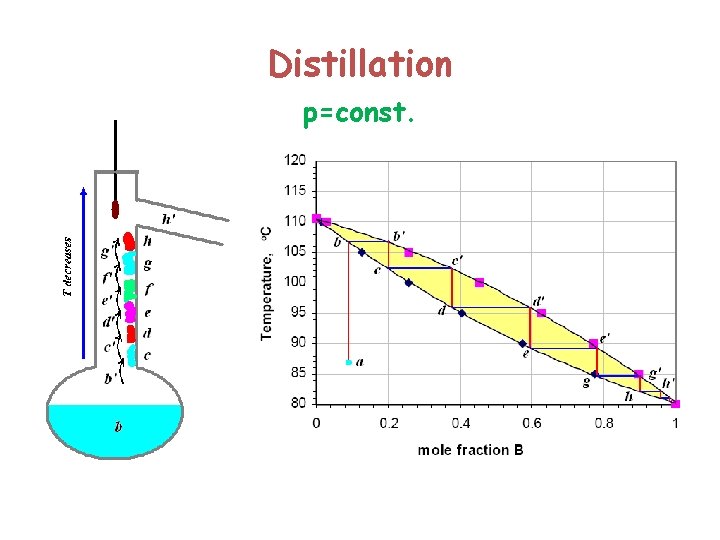
Distillation p=const.
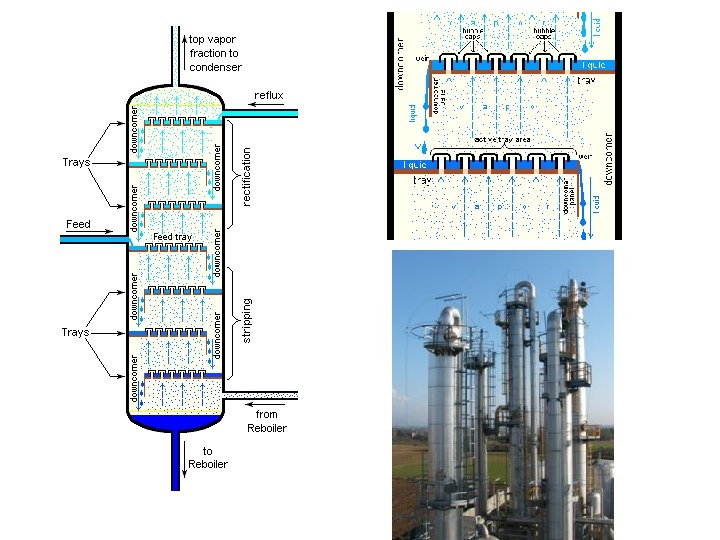
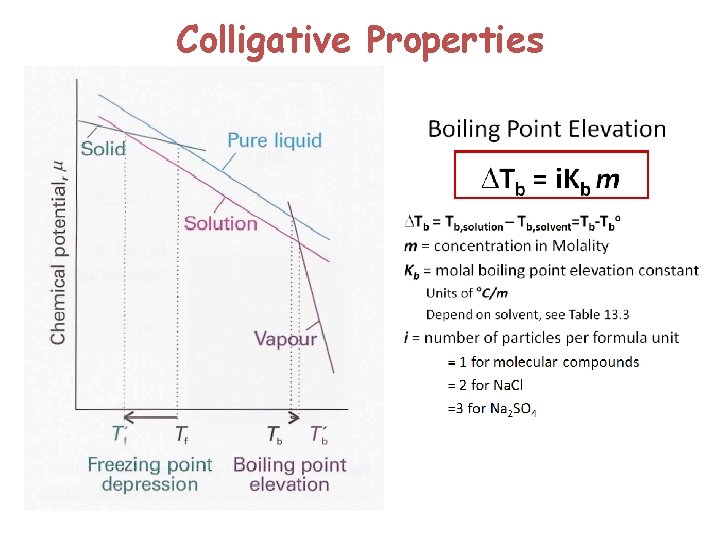
Colligative Properties
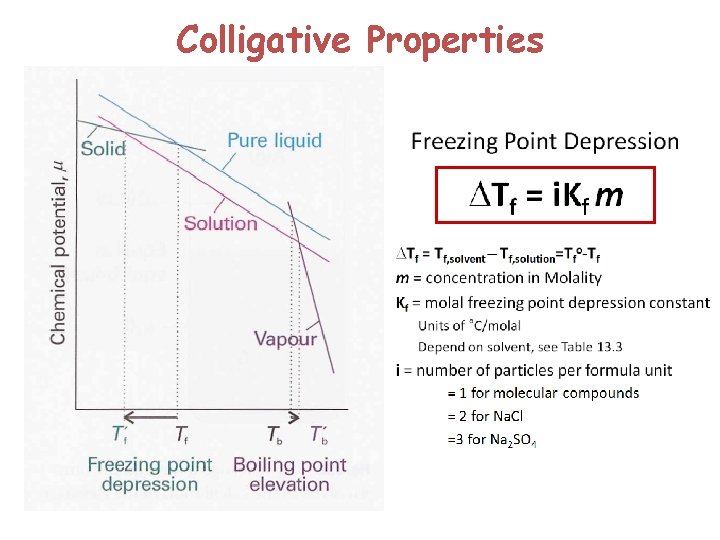
Colligative Properties
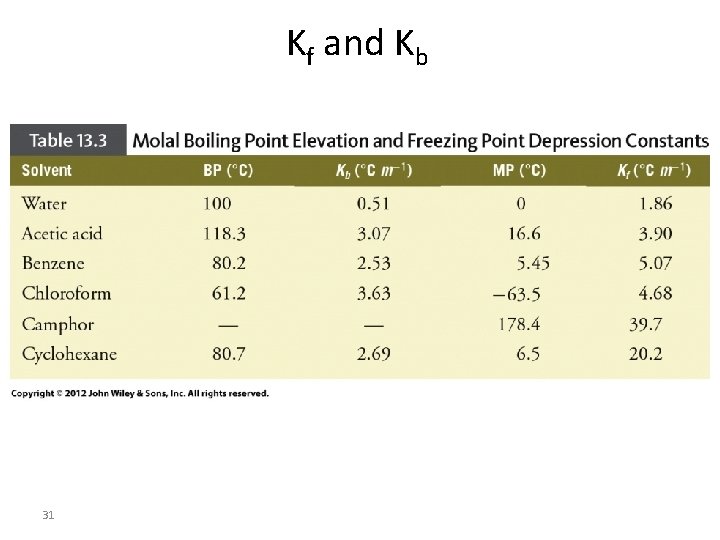
Kf and Kb 31
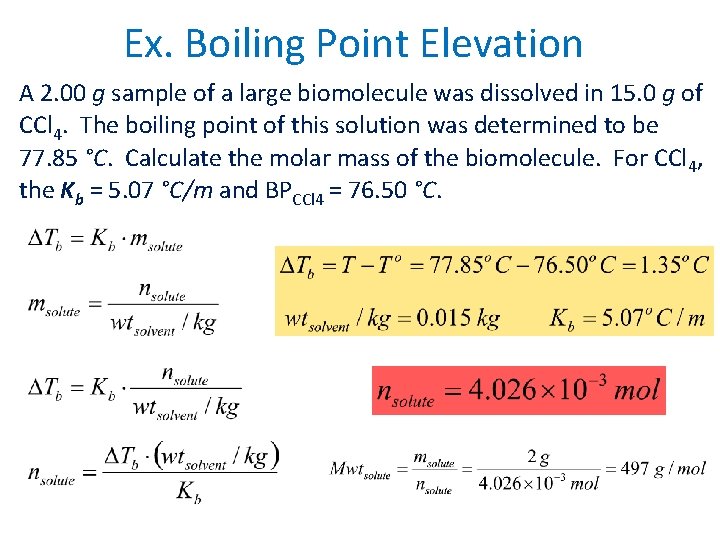
Ex. Boiling Point Elevation A 2. 00 g sample of a large biomolecule was dissolved in 15. 0 g of CCl 4. The boiling point of this solution was determined to be 77. 85 °C. Calculate the molar mass of the biomolecule. For CCl 4, the Kb = 5. 07 °C/m and BPCCl 4 = 76. 50 °C.

Ex. Freezing Point Depression Estimate the freezing point of a permanent type of antifreeze solution made up of 100. 0 g ethylene glycol, C 2 H 6 O 2, (MM = 62. 07) and 100. 0 g H 2 O (MM = 18. 02). 33
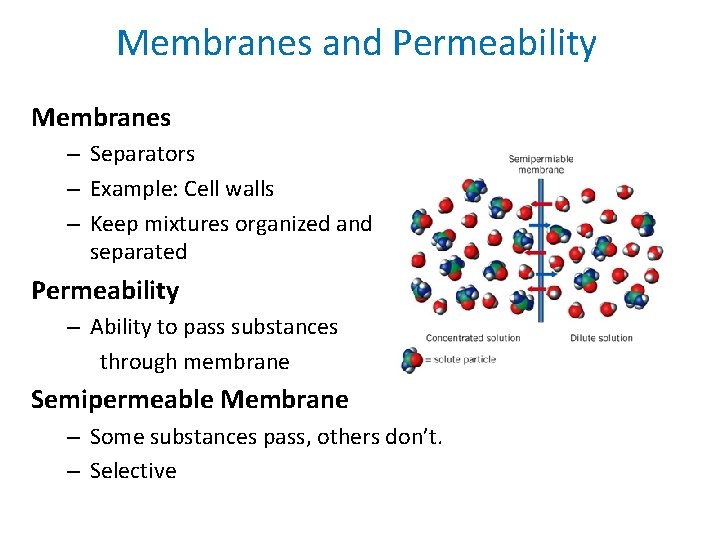
Membranes and Permeability Membranes – Separators – Example: Cell walls – Keep mixtures organized and separated Permeability – Ability to pass substances through membrane Semipermeable Membrane – Some substances pass, others don’t. – Selective
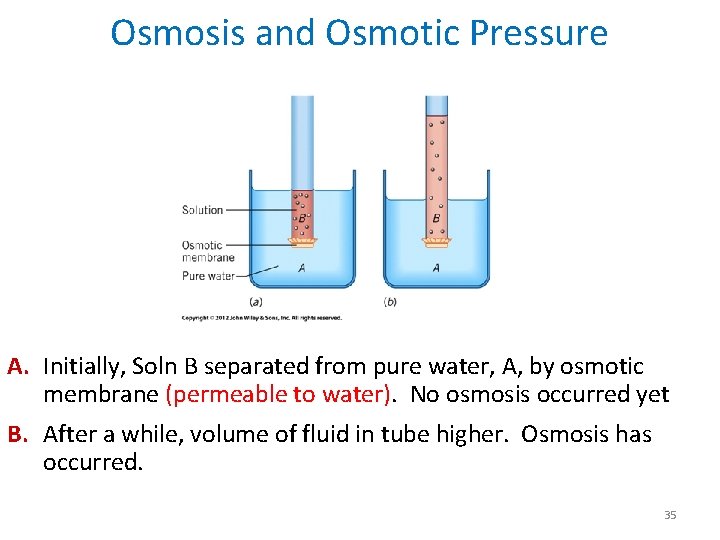
Osmosis and Osmotic Pressure A. Initially, Soln B separated from pure water, A, by osmotic membrane (permeable to water). No osmosis occurred yet B. After a while, volume of fluid in tube higher. Osmosis has occurred. 35
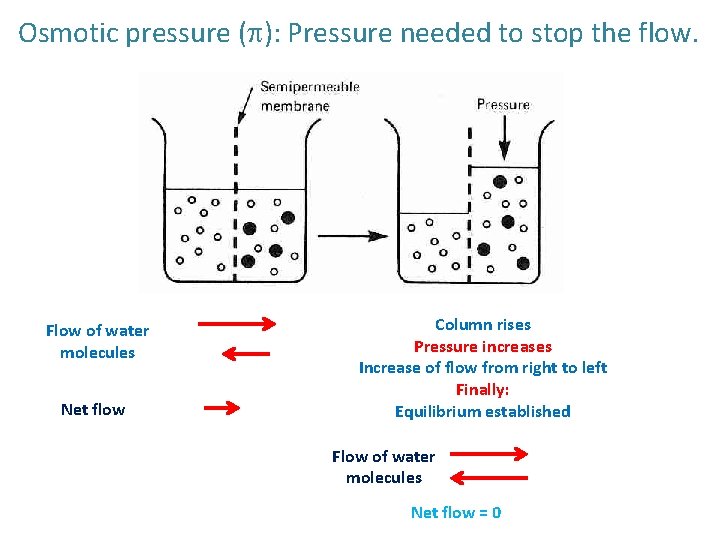
Osmotic pressure (p): Pressure needed to stop the flow. Flow of water molecules Net flow Column rises Pressure increases Increase of flow from right to left Finally: Equilibrium established Flow of water molecules Net flow = 0

Equation for Osmotic Pressure • Assumes dilute solutions p=i. MRT – p = osmotic pressure – i = number of ions per formula unit = 1 for molecules – M = molarity of solution • Molality, m, would be better, but M simplifies • Especially for dilute solutions, where m M – T = Kelvin Temperature – R = Ideal Gas constant = 0. 082057 L·atm·mol 1 K 1 37
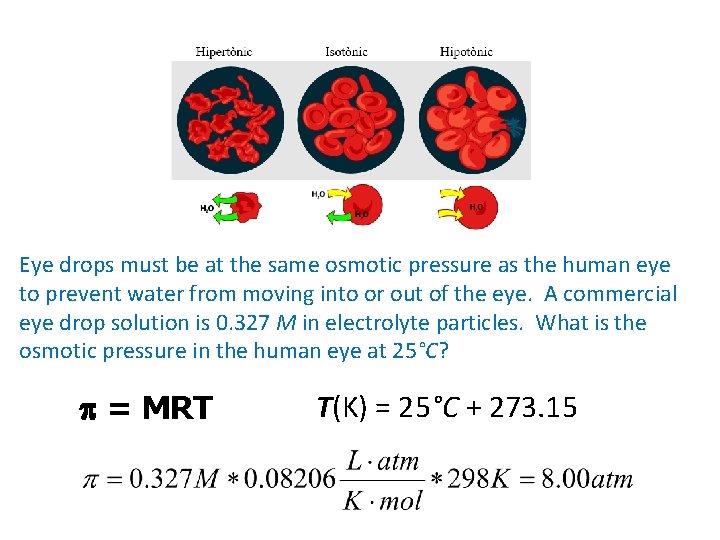
Eye drops must be at the same osmotic pressure as the human eye to prevent water from moving into or out of the eye. A commercial eye drop solution is 0. 327 M in electrolyte particles. What is the osmotic pressure in the human eye at 25°C? p = MRT T(K) = 25°C + 273. 15

Using p to determine MM The osmotic pressure of an aqueous solution of certain protein was measured to determine its molar mass. The solution contained 3. 50 mg of protein in sufficient H 2 O to form 5. 00 m. L of solution. The measured osmotic pressure of this solution was 1. 54 torr at 25 °C. Calculate the molar mass of the protein.
- Slides: 39