PHASE EQUILIBRIA AND THE PHASE RULE 662021 The

PHASE EQUILIBRIA AND THE PHASE RULE 6/6/2021

The phase rule The three primary phases of matter are often defined individually under different conditions. In practice we encounter states of matter in coexistence. The position of the equilibrium between these states is affected by the environment (temperature, pressure) and composition. 6/6/2021

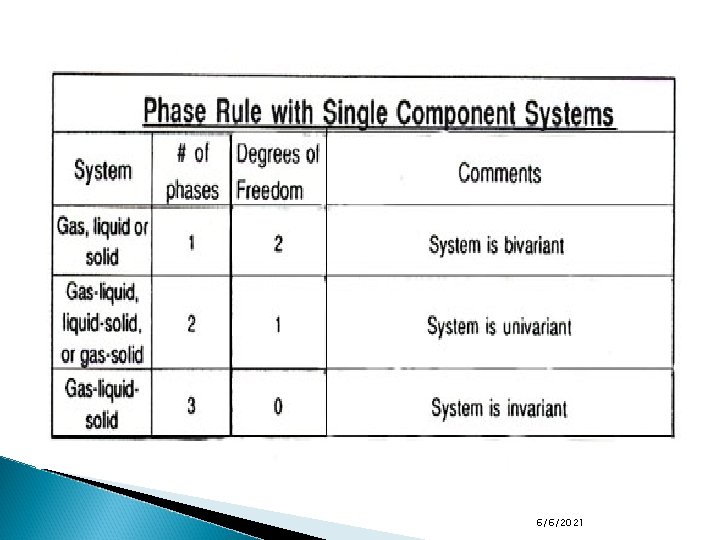
The phase rule Phase Rule ◦ A devise for determining the least number of intensive variables (independent variables that do not depend on the phase volume or mass such as temperature, pressure, density or concentration) that can be changed without changing the equilibrium state of the system, or alternately the least number required to define the state of the system. ◦ This critical number is called F, the number of degrees of freedom of the system and the rule is expressed as follows: Where C is the number of components and P is the number of phases present. 6/6/2021

The phase rule Number of Degrees of Freedom (F) Phase (P) Components or Chemical Entities (C) ◦ The LEAST number of intensive variables that must be known to describe the system completely. Ex. temperature, concentration, pressure, density, etc. ◦ A consistent or homogenous, physically distinct segment of a system that is separated from other segments of the system by bounding surfaces. ◦ The smallest number of constituents by which the composition of each phase in the system at equilibrium can be expressed, usually in the form of a chemical formula or chemical equation. 6/6/2021

The phase rule is used to study and understand the way that temperature, pressure, concentration, etc. affect the phase of a substance. Application: ◦ A gas, e. g. water vapour confined to a particular volume. ◦ Apply phase rule: F=1 -1+2=2. ◦ This means that two intensive variables (temperature and pressure, temperature and concentration) must be known to duplicate this system exactly. ◦ Such a system is usually described as bivariant. 6/6/2021

The phase rule Application: ◦ A liquid such as water in equilibrium with its vapor ( we have 2 phase system) ◦ F=1 -2+2=1. ◦ By stating temperature, the system is completely defined because the pressure under which liquid and vapor can coexist is also fixed. ◦ If we decide to work under a particular pressure, then the temperature of the system is automatically defined: ◦ The system is described as univariant. ◦ When we have a liquid water, vapor and ice ◦ Phase rule states that the degrees of freedom = 1 -3+2=0 ◦ There are no degrees of freedom, if we attempt to vary the conditions of temperature or pressure necessary to maintain the system, we will lose a phase. ◦ The combination is fixed and unique. ◦ The system is invariant. 6/6/2021
6/6/2021

The phase rule ü ü As the number of components increases, so do the required degrees of freedom needed to define the system. Consequently, as the system becomes more complex, it becomes necessary to fix more variables to define the system. As the number of phases in equilibrium increases, the number of the required degrees of freedom becomes less. Liquid water+vapor F=1 -2+2=1 Ethyl alcohol+vapor F=1 -2+2=1 liquid water+liquid ethanol+vapor F=2 -2+2=2 liquid water+liquid benzyl alcohol+vapor mixture F=2 -3+2=1 benzyl alcohol and water form two separate liquid phases and one vapor phase. 6/6/2021
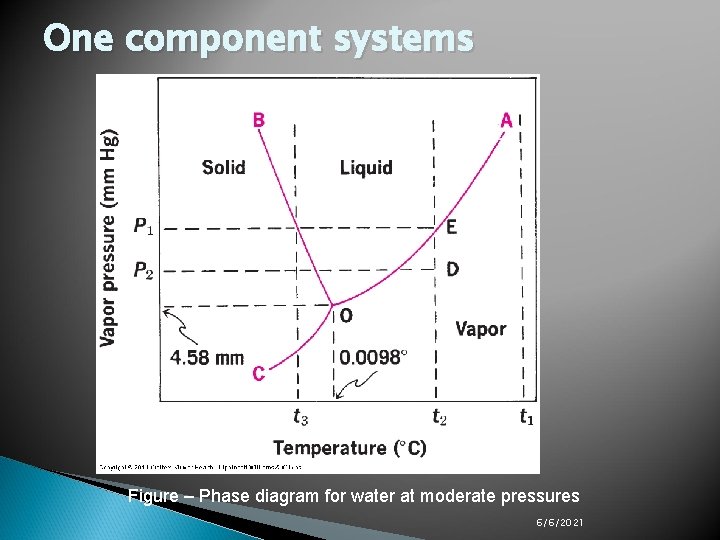
One component systems Figure – Phase diagram for water at moderate pressures 6/6/2021

One component systems The previous figure (phase diagram of water at moderate pressures): Curve OA is known as the vapor pressure curve; its upper limit is at the critical temperature, 374ºC for water, and its lower end terminates at 0. 0098ºC, called the triple point. Along the vapor pressure curve, vapor and liquid coexist in equilibrium. Curve OC is the sublimation curve, and here vapor and solid exist together in equilibrium. Curve OB is the melting point curve at which liquid and solid are in equilibrium. The negative slope of OB shows that the freezing point of water decreases with increasing external pressure. 6/6/2021

The results of changes in pressure (at fixed temperature) or changes in temperature (at fixed pressure) becomes evident by referring to the phase diagram: If t 1 (above the critical temperature) is held constant, no matter how much the pressure is raised, the system remains a gas. At t 2 below the critical temperature and above the triple point, water vapor is converted into liquid water by an increase in pressure. At temperature below the triple point e. g. t 3, an increase in pressure on water in the vapor state converts the vapor first to ice and then at higher pressure into liquid water. At the triple point, all three phases are in equilibrium, that is, the only equilibrium is at this pressure and at this temperature of 0. 0098 ºC. The triple point for air-free water is at 0. 0098ºC and 4. 58 mm Hg. Whereas the freezing point (the point at which liquid water is saturated with air is in equilibrium with ice at a total pressure of 1 atm) is 0 ºC. 6/6/2021

One component systems ü ü ü As mentioned in the table (phase rule with single component systems) and the figure (phase diagram for water at moderate pressures): In any one of the three regions in which pure solid, liquid, or vapor exists, F=1 -1+2=2, so two variables (e. g. temperature and pressure) are needed to describe the system (Bivariant system). Along any of the three lines, where two phases exist, F=12+2=1, hence, only one condition (either temperature or pressure) is needed to define the system (Univariant system). Finally, at the triple point where three phases exist, F=0 (Invariant system). 6/6/2021

Condensed Systems ◦ These are systems in which the vapor phase is ignored and only solid and/or liquid phases are considered. ◦ The vapor phase is disregarded and the pressure is considered to be 1 atm. ◦ The variables considered are: temperature and concentration. 6/6/2021

Two component systems containing liquid phases Miscible systems such as water and ethanol. Immiscible systems such as water and mercury. Partial miscibility (or immiscibility) such as phenol and water. 6/6/2021

Two component systems containing liquid phases Phenol and water. ◦ These liquids are partially miscible in each other. ◦ At certain ratios the liquids are completely miscible and at others they are immiscible. ◦ The 2 degrees of freedom of this mixture are temperature and concentration (condensed system). 6/6/2021

Two component systems containing liquid phases The curve gbhci shows the limits of temperature and concentration within which two liquid phases exist in equilibrium. The region outside this curve contain systems with only one liquid phase. The maximum temperature that these two liquids can exist in a 2 phase system is 66. 8°C. This is called the critical solution temperature or the upper consolute temperature (i. e. The maximum temperature at which the two – phase region exists). In this system above 66. 8°C all combinations of phenol and water will be completely miscible and will be one phase liquid systems. 6/6/2021

Two component systems containing liquid phases: Figure – Temperature-composition diagram for the system consisting of water and phenol 6/6/2021
6/6/2021

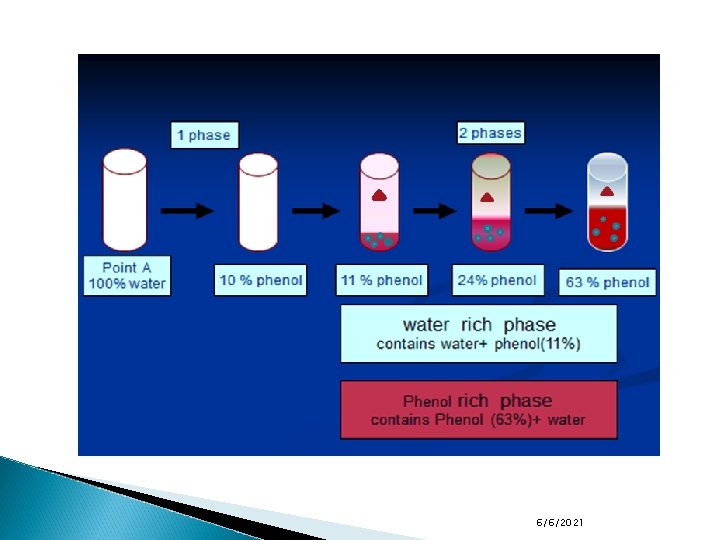
Two component systems containing liquid phases Starting at the point a, a system with 100% water and adding known increments of phenol to a fixed weight of water while maintaining the system at 50 o. C will result in the formation of a single liquid phase (water with dissolved phenol). This continues until point b. At point b, a minute amount of a second phase appears. The concentration of phenol and water at which this occurs is 11% by weight of phenol in water. 6/6/2021

Two component systems containing liquid phases Analysis of the second phase, which separates out in the bottom, shows it to contain 63% by weight of phenol in water. This composition corresponds to point c in the diagram. As we prepare mixtures with increasing quantities of phenol (we proceed from point b to point c), the amount of the phenol-rich phase (B) increases and the amount of the water-rich phase (A) decreases. Once the total concentration of phenol exceeds 63% at 50 o. C, a single phenol-rich liquid phase is formed. 6/6/2021

Two component systems containing liquid phases The line bc drawn across the region containing two phases is termed the tie line. All systems prepared on a tie line, at equilibrium will separate into phases of constant composition, these phases are termed conjugate phases. For example, any system represented by a point on the line bc at 50 o. C separate to give a pair of conjugate phases whose compositions are b and c. However, the relative amounts of the two layers or phases vary. 6/6/2021

Two component systems containing liquid phases If we prepared a system containing 24% by weight of phenol and 76% by weight of water (point d), at equilibrium we will have two liquid phases. The upper one (A) has a composition of 11% phenol in water and the lower one (B) has a composition of 63% phenol in water. Phase B will lie below phase A because it is rich in phenol which has a higher density than water. 6/6/2021

Two component systems containing liquid phases The relative weights of the two phases can be calculated using the following formula: Working on a tie line in a phase diagram enables us to calculate the weight of the phases, in addition to the composition of each phase. Thus, it becomes a simple matter to calculate the distribution of phenol (or water) throughout the system as a whole See example discussed page 40 in the fourth edition (page 51 in fifth edition, page 45 in sixth edition). See problem 2 -21 page 52 in the fourth edition (2 -21 page 687 in fifth edition, 2 -14 in sixth edition). 6/6/2021

The use of Tie line in calculations: As an example, let us suppose that we mixed 24 g of phenol with 76 g of water, warmed the mixture to 50 o. C, and allowed it to reach equilibrium at this temperature. Weight phase A/weight of phase B = dc/bd =6324/24 -11=39/13 =3/1 Weight of A= ¾ x 100= 75, wt. of B = ¼ x 100= 25 Phase A=75 gm , phase B =25 gm. Amount of phenol in A=75 x 11/100= 8. 25 gm Amount of phenol in B= 25 x 63/100= 15. 75 gm 24 gm

The use of Tie line in calculations:

Application of Tie line: To formulate systems containing more than one component where it may be advantageous to achieve a single-phase product. Handling of solid phenol, a necrotic agent (caustic agent), is facilitated in the pharmacy if a solution of phenol and water is used. The most convenient formulation of a single liquid phase solution was 80% w/v, equivalent to about 76% w/w. This mixture has a freezing point of about 3. 5 o. C, compared to liquefied phenol USP which contains approximately 90% w/w/ phenol and freezes at 17 o. C.
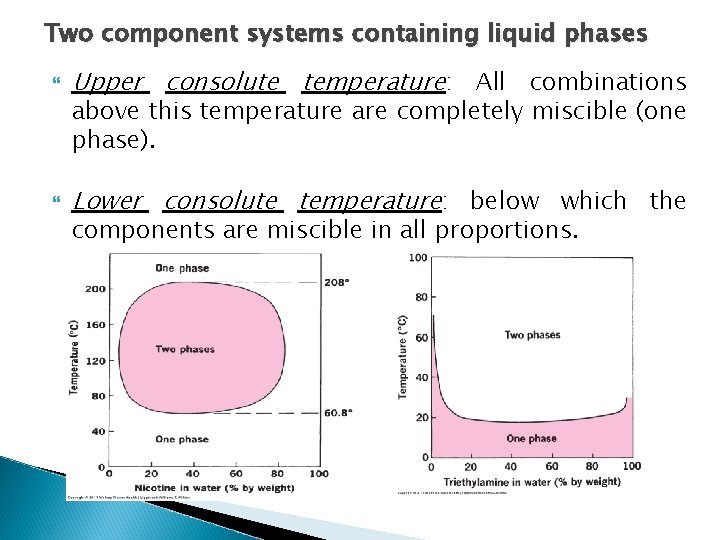
Two component systems containing liquid phases Upper consolute temperature: All combinations Lower consolute temperature: below which the above this temperature are completely miscible (one phase). components are miscible in all proportions.

Two component systems containing solid and liquid phases: Solid – liquid mixtures in which both components are miscible in the liquid state and completely immiscible as solids. This is because the solid phases consist of pure crystalline components. These are said to be eutectic mixtures. The name (eutectic) comes from the greek 'eutektos', meaning 'easily melted‘. 6/6/2021
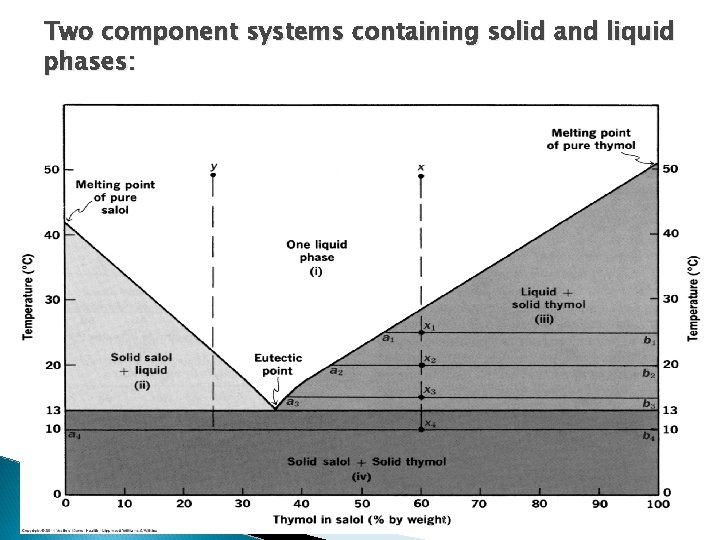
Two component systems containing solid and liquid phases: 6/6/2021

Two component systems containing solid and liquid phases: The eutectic temperature is the lowest temperature at which the liquid phase can occur. Above the temperature the components are liquid and below the components are solids. At this temperature, the components in the liquid state pass all at once to the solid phase. A eutectic composition is the composition of two (or more) compounds that exhibits a melting temperature lower than that of any other mixture of the compounds. In the previous example, it was 13 o. C and this occurs for a mix contains 34% thymol in salol. This point on the phase diagram is known as the eutectic point. This is attributed to the fact that the solid phase in the eutectic composition is an intimate mixture of fine crystals of two compounds. This leads to the phenomenon of “contact melting”, resulting in the lowest melting point over a composition range. The primary criterion for eutectic formation is the mutual solubility of the components in the liquid or melt phase. Systems of two or more components show different interactions (how solutes interact in solution) at different concentrations, with the eutectic point provides the favoured composition for the solutes in solution. 6/6/2021

Two component systems containing solid and liquid phases: At the eutectic point, three phases (liquid, solid salol, solid thymol) coexist. The eutectic point in this condensed system represent an invariant system (2 components and 3 phases) (pressure is fixed). Prilocaine (mp 37 -38 o. C) and lidocaine (mp 68 -69 o. C) are local anesthetics that form an eutectic mixture at 1: 1 ratios with a melting point of 18 o. C, this combination can be used to prepare a liquid dosage form that may be used for topical applications. 6/6/2021

Phase equilibrium in three component systems 6/6/2021

Phase equilibrium in three component systems In a system containing three components, but only one phase there are 4 degrees of freedom. These are temperature, pressure, and the concentrations of 2 of the 3 components. This system is considered non-condensed. If we hold the temperature constant and the system is condensed where pressure is held at 1 atm then the degrees of freedom of the system are only 2. 6/6/2021

Phase equilibrium in three component systems Each of the corners or apexes of the triangle represent 100% by weight of one component (A, B or C). The three lines joining the corner points represent twocomponent mixtures of the three possible combinations of A, B and C. The area within the triangle represents all possible combinations of A, B and C to give three component systems. If a line is drawn through any apex to a point on the opposite side (e. g. line DC in the previous figure), then all systems represented by points on such a line have constant ratio of two components, in this case A and B. Any line drawn parallel to one side of the triangle, for example, line HI in the previous figure, represents ternary systems in which the proportion ( or percent by weight) of one component is constant. 6/6/2021

Phase equilibrium in three component systems 6/6/2021

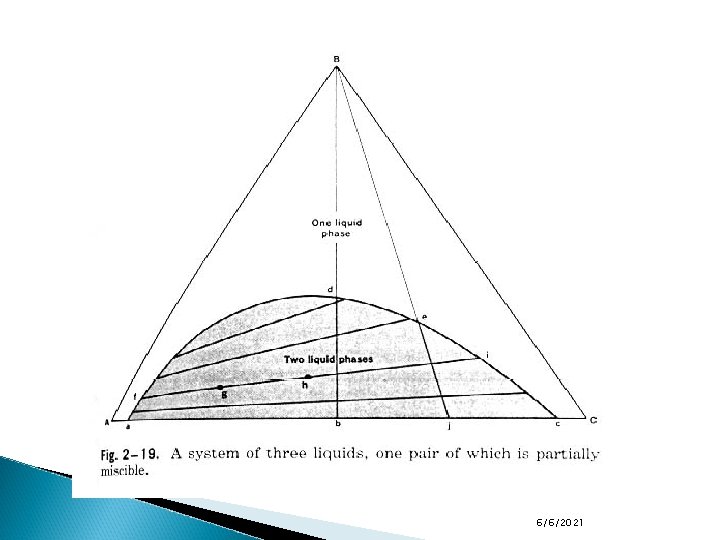
Ternary Systems with one pair of partially miscible liquids In a system in which there are 2 liquids which are partially miscible and then you add a liquid that is miscible with both original liquids there will be a phase in which all liquids are completely miscible or are one phase. An example of this is benzene and water which are partially miscible. Alcohol is completely miscible with both benzene and water. The addition of sufficient alcohol to a two-phase system of benzene and water would produce a single liquid phase in which all three components are miscible. Alcohol in this case is acting in a manner comparable to that of temperature in the binary phenol-water system. See figure on the next slide. The curve afdeic, frequently termed the binodal curve, marks the extent of the two-phase region. 6/6/2021
6/6/2021

Ternary Systems with one pair of partially miscible liquids In the previous figure (a system of three liquids, one pair of which is partially miscible), F=2 in a single-phase region, so two concentrations must be defined to fix the particular region. Within the binodal curve, afdeic, F=1 so we need to know one concentration term, since this will allow the composition of one phase to be fixed on the binodal curve. From the tie line, we can then obtain the composition of the conjugate phase. The tie lines in this case are not parallel to the base of the triangle. It depends on the relative solubility of the third component (alcohol) in the other two components (water, benzene). 6/6/2021
- Slides: 38