Phase diagrams of carbon and superconductors Ross Mc

Phase diagrams of carbon and superconductors Ross Mc. Kenzie condensedconcepts. blogspot. com

On liquid-vapour phase boundary d. P/d. T > 0 ! What happens when you don't properly vent a sealed storage tank before emptying it? Hot gas/air in the sealed container is left to cool and …. http: //www. youtube. com/watch? v =2 WJVHt. F 8 Gw. I&NR=1

First-order phase transitions involve discontinuities in the first-order derivatives of the Gibbs free energy. Entropy of gas > liquid > solid Why?
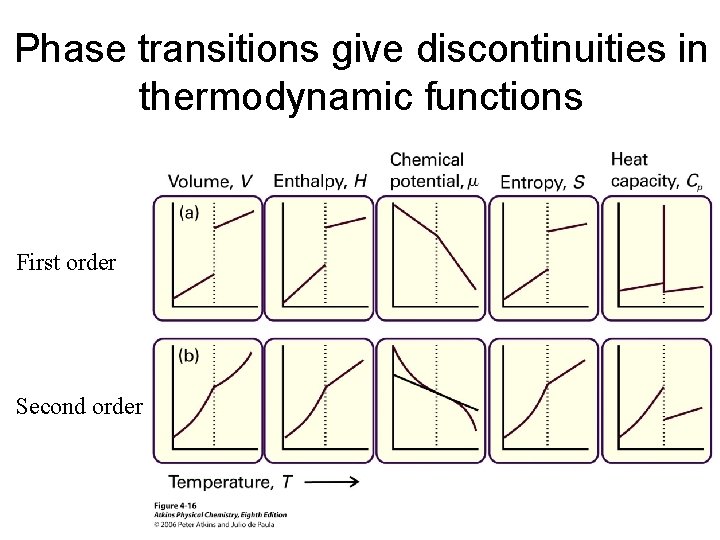
Phase transitions give discontinuities in thermodynamic functions First order Second order

Recommended reading • D. V. Schroeder, Thermal Physics, Addison Wesley, 1999. pp. 166 -186 • Some tutorial questions will be taken from this book. • Textbook for PHYS 3020

Phase diagram of carbon

Never forget • Second law for an open system in equilibrium with an environment at a specified pressure and temperature: The Gibbs free energy (G=U+PV-TS) can never increase • Chemical potential = G per mole • d. G= -S d. T + V d. P

At what pressure will graphite be converted to diamond? • Exercise 4. 23, Atkins & de. Paula • The following data apply to 25 C and 100 k. Pa. Assume the specific volume, Vs is constant with pressure changes. Δf. G (k. J/mol) Vs (cm 3/g) Graphite 0 0. 444 Diamond +2. 8678 0. 284

Gibbs free energy vs. pressure

Superconductors • A new phase of matter. • A macroscopic quantum state! • Conduct electricity with exactly zero electrical resistivity. • Expel magnetic flux (Meissner effect). • Used to levitate magnets http: //h 0. web. u-psud. fr/supraconductivite

Phase diagram of a superconductor: magnetic field vs. temperature • Tc is the transition temperature. • For H > 0, transition is first -order • H=0, T=Tc is a critical point

Exercises • The Clausius-Clapeyron equation can be used to describe the slope of the metalsuperconductor phase boundary. • Schroeder, Problems

Review • A phase diagram (for pressure vs. temperature) tells us which phase is the most stable (i. e. , has the lowest Gibbs free energy).
- Slides: 13