Phase Diagrams By Anil Kumar Garikapati Assoc Prof

Phase Diagrams By Anil Kumar Garikapati Assoc. Prof. Dept. of Mech. Engg . 1
Phase Diagrams ISSUES TO ADDRESS. . . • When we combine two elements. . . what is the resulting equilibrium state? • In particular, if we specify. . . -- the composition (e. g. , wt% Cu - wt% Ni), and -- the temperature (T ) then. . . How many phases form? What is the composition of each phase? What is the amount of each phase? Phase B Phase A Nickel atom Copper atom 2
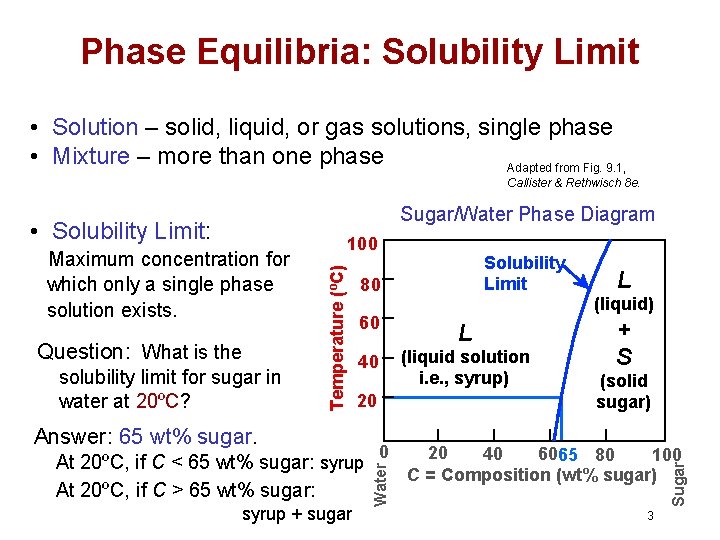
Phase Equilibria: Solubility Limit • Solution – solid, liquid, or gas solutions, single phase • Mixture – more than one phase Adapted from Fig. 9. 1, Callister & Rethwisch 8 e. Sugar/Water Phase Diagram solubility limit for sugar in water at 20ºC? 60 L 40 (liquid solution i. e. , syrup) 20 Answer: 65 wt% sugar. At 20ºC, if C < 65 wt% sugar: syrup At 20ºC, if C > 65 wt% sugar: syrup + sugar Solubility Limit 80 0 Water Question: What is the Temperature (ºC) Maximum concentration for which only a single phase solution exists. 100 L (liquid) + S (solid sugar) 20 40 6065 80 100 C = Composition (wt% sugar) Sugar • Solubility Limit: 3

Components and Phases • Components: The elements or compounds which are present in the alloy (e. g. , Al and Cu) • Phases: The physically and chemically distinct material regions that form (e. g. , and ). Aluminum. Copper Alloy Adapted from chapteropening photograph, Chapter 9, Callister, Materials Science & Engineering: An Introduction, 3 e. (lighter phase) (darker phase) 4
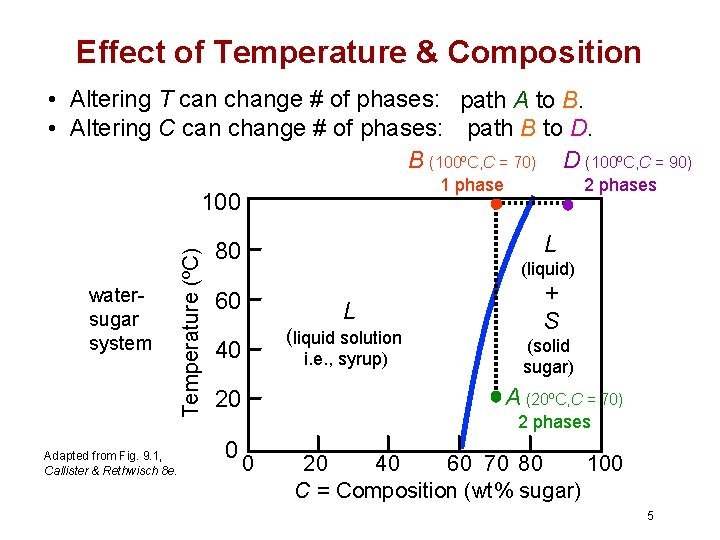
Effect of Temperature & Composition • Altering T can change # of phases: path A to B. • Altering C can change # of phases: path B to D. B (100ºC, C = 70) D (100ºC, C = 90) 1 phase watersugar system Adapted from Fig. 9. 1, Callister & Rethwisch 8 e. Temperature (ºC) 100 L 80 (liquid) 60 L (liquid solution 40 i. e. , syrup) + S (solid sugar) A (20ºC, C = 70) 20 0 2 phases 0 20 40 60 70 80 100 C = Composition (wt% sugar) 5

Criteria for Solid Solubility Simple system (e. g. , Ni-Cu solution) Crystal Structure electroneg r (nm) Ni FCC 1. 9 0. 1246 Cu FCC 1. 8 0. 1278 • Both have the same crystal structure (FCC) and have similar electronegativities and atomic radii (W. Hume – Rothery rules) suggesting high mutual solubility. • Ni and Cu are totally soluble in one another for all proportions. 6
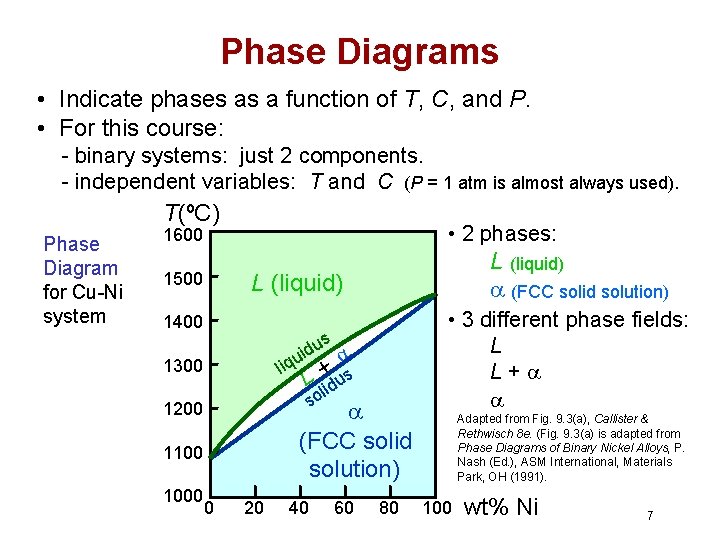
Phase Diagrams • Indicate phases as a function of T, C, and P. • For this course: - binary systems: just 2 components. - independent variables: T and C (P = 1 atm is almost always used). T(ºC) Phase Diagram for Cu-Ni system • 2 phases: 1600 1500 L (liquid) 1400 us d i u liq L +lidus so 1300 (FCC solid solution) 1200 1100 1000 L (liquid) (FCC solid solution) 0 20 40 60 80 • 3 different phase fields: L L+ Adapted from Fig. 9. 3(a), Callister & Rethwisch 8 e. (Fig. 9. 3(a) is adapted from Phase Diagrams of Binary Nickel Alloys, P. Nash (Ed. ), ASM International, Materials Park, OH (1991). 100 wt% Ni 7
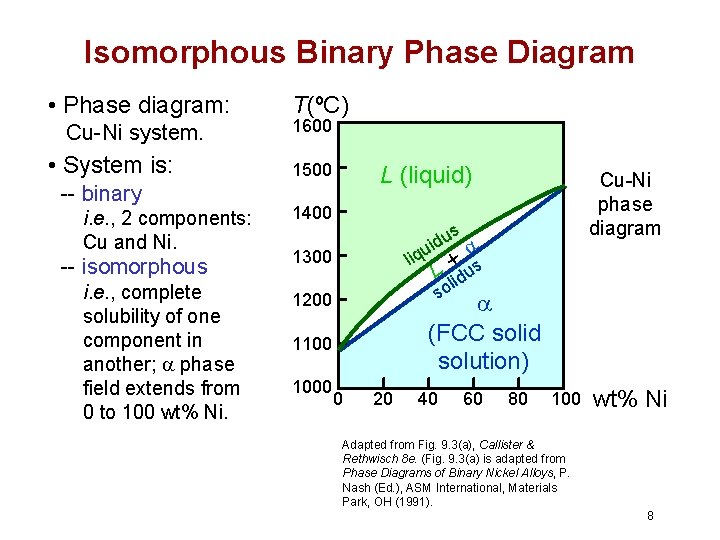
Isomorphous Binary Phase Diagram • Phase diagram: Cu-Ni system. • System is: -- binary i. e. , 2 components: Cu and Ni. -- isomorphous i. e. , complete solubility of one component in another; phase field extends from 0 to 100 wt% Ni. T(ºC) 1600 1500 L (liquid) 1400 us d i u liq L +lidus so 1300 (FCC solid solution) 1200 1100 1000 Cu-Ni phase diagram 0 20 40 60 80 100 Adapted from Fig. 9. 3(a), Callister & Rethwisch 8 e. (Fig. 9. 3(a) is adapted from Phase Diagrams of Binary Nickel Alloys, P. Nash (Ed. ), ASM International, Materials Park, OH (1991). wt% Ni 8
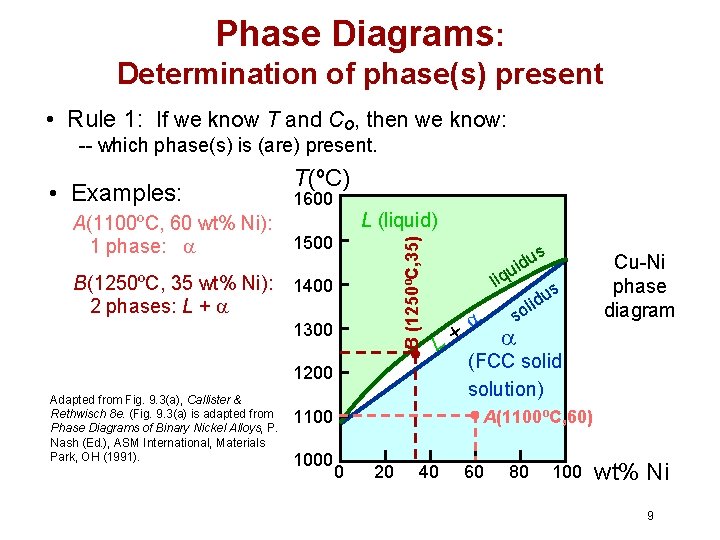
Phase Diagrams: Determination of phase(s) present • Rule 1: If we know T and Co, then we know: -- which phase(s) is (are) present. A(1100ºC, 60 wt% Ni): 1 phase: B(1250ºC, 35 wt% Ni): 2 phases: L + 1600 L (liquid) 1500 B (1250ºC, 35) • Examples: T(ºC) 1400 1300 s u uid liq L+ 1200 Adapted from Fig. 9. 3(a), Callister & Rethwisch 8 e. (Fig. 9. 3(a) is adapted from Phase Diagrams of Binary Nickel Alloys, P. Nash (Ed. ), ASM International, Materials Park, OH (1991). (FCC solid solution) A(1100ºC, 60) 1100 1000 s du i l so Cu-Ni phase diagram 0 20 40 60 80 100 wt% Ni 9
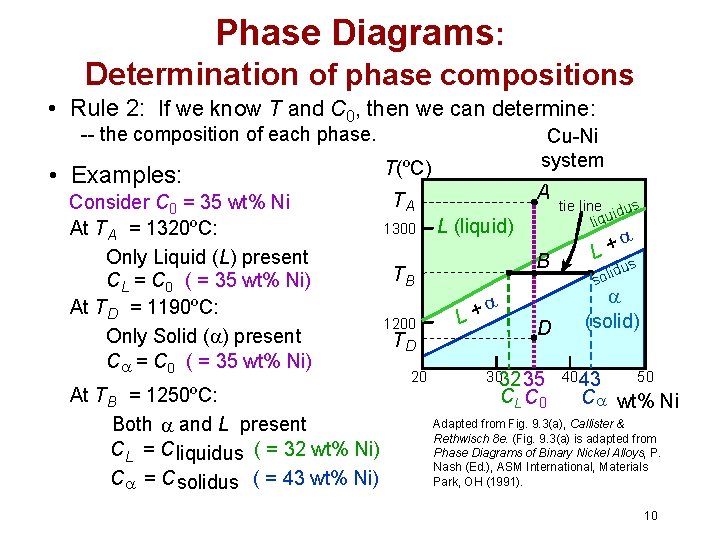
Phase Diagrams: Determination of phase compositions • Rule 2: If we know T and C 0, then we can determine: -- the composition of each phase. • Examples: Consider C 0 = 35 wt% Ni At TA = 1320ºC: Only Liquid (L) present CL = C 0 ( = 35 wt% Ni) At TD = 1190ºC: Only Solid ( ) present C = C 0 ( = 35 wt% Ni) At TB = 1250ºC: Both and L present CL = C liquidus ( = 32 wt% Ni) C = C solidus ( = 43 wt% Ni) Cu-Ni system T(ºC) A TA 1300 L (liquid) TB 1200 TD 20 L + tie line dus i liqu B L+ D (solid) 3032 35 CL C 0 s idu l o s 4043 50 C wt% Ni Adapted from Fig. 9. 3(a), Callister & Rethwisch 8 e. (Fig. 9. 3(a) is adapted from Phase Diagrams of Binary Nickel Alloys, P. Nash (Ed. ), ASM International, Materials Park, OH (1991). 10
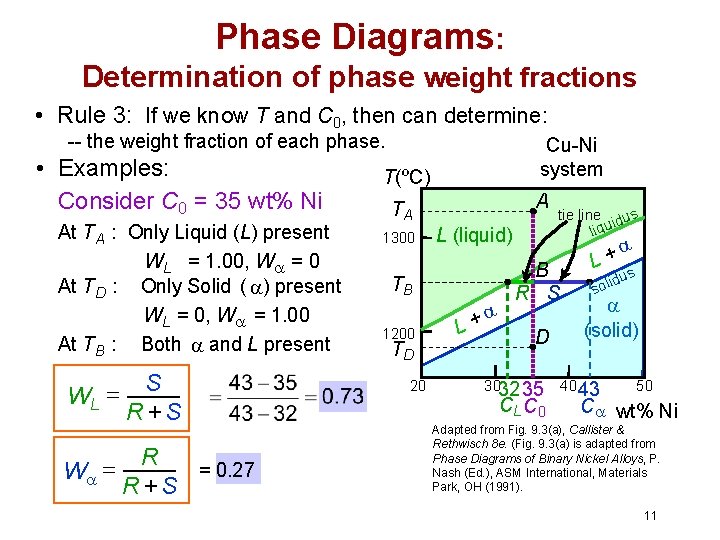
Phase Diagrams: Determination of phase weight fractions • Rule 3: If we know T and C 0, then can determine: -- the weight fraction of each phase. • Examples: Consider C 0 = 35 wt% Ni At TA : Only Liquid (L) present WL = 1. 00, W = 0 At TD : Only Solid ( ) present WL = 0, W = 1. 00 At TB : Both and L present WL = W = S R +S R = 0. 27 R +S Cu-Ni system T(ºC) A TA 1300 L (liquid) TB 1200 TD 20 + L tie line dus i liqu B R S D 3032 35 CL C 0 + L s idu l o s (solid) 40 43 50 C wt% Ni Adapted from Fig. 9. 3(a), Callister & Rethwisch 8 e. (Fig. 9. 3(a) is adapted from Phase Diagrams of Binary Nickel Alloys, P. Nash (Ed. ), ASM International, Materials Park, OH (1991). 11
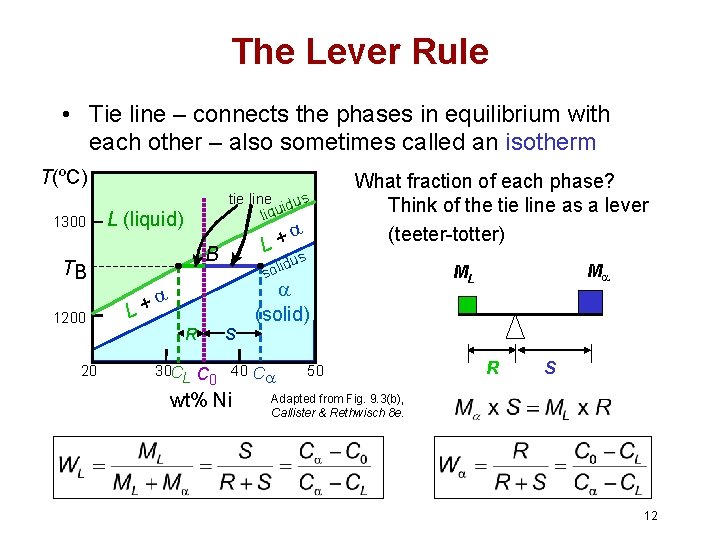
The Lever Rule • Tie line – connects the phases in equilibrium with each other – also sometimes called an isotherm T(ºC) 1300 tie line dus i liqu L (liquid) B TB 1200 20 + L us id sol L+ R 30 CL What fraction of each phase? Think of the tie line as a lever (teeter-totter) S (solid) C 0 40 C wt% Ni 50 M ML R S Adapted from Fig. 9. 3(b), Callister & Rethwisch 8 e. 12
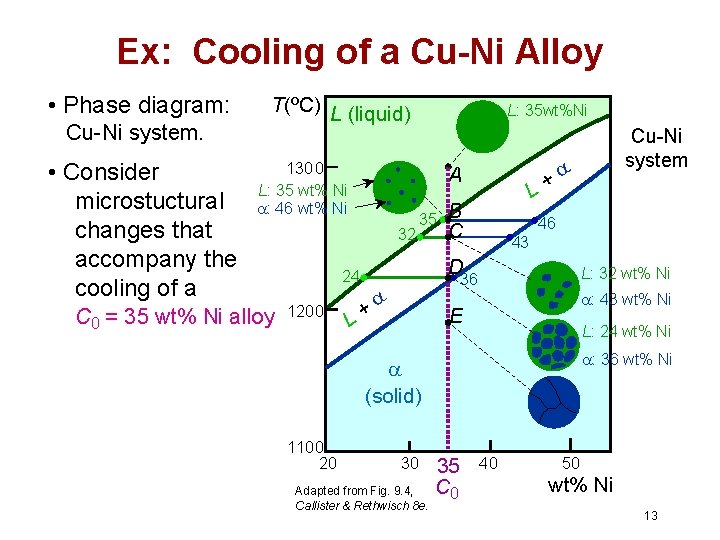
Ex: Cooling of a Cu-Ni Alloy • Phase diagram: Cu-Ni system. • Consider microstuctural changes that accompany the cooling of a T(ºC) L (liquid) 1300 L: 35 wt% Ni : 46 wt% Ni C 0 = 35 wt% Ni alloy L: 35 wt%Ni A 32 35 B C 46 43 D 24 1200 L+ L: 32 wt% Ni 36 + L : 43 wt% Ni E L: 24 wt% Ni : 36 wt% Ni (solid) 1100 20 30 Adapted from Fig. 9. 4, Callister & Rethwisch 8 e. Cu-Ni system 35 C 0 40 50 wt% Ni 13
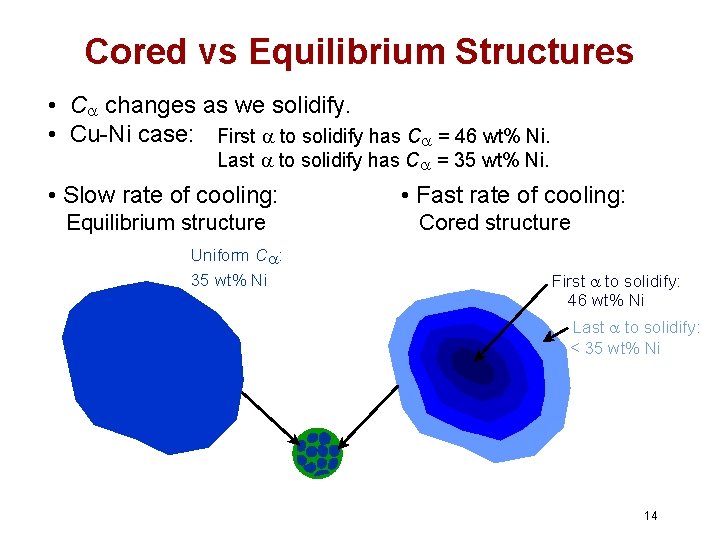
Cored vs Equilibrium Structures • C changes as we solidify. • Cu-Ni case: First to solidify has C = 46 wt% Ni. Last to solidify has C = 35 wt% Ni. • Slow rate of cooling: Equilibrium structure Uniform C : 35 wt% Ni • Fast rate of cooling: Cored structure First to solidify: 46 wt% Ni Last to solidify: < 35 wt% Ni 14
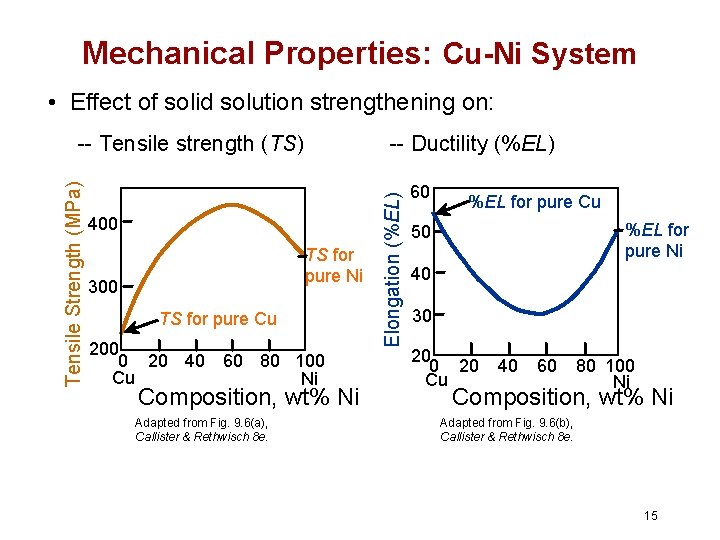
Mechanical Properties: Cu-Ni System • Effect of solid solution strengthening on: -- Ductility (%EL) 400 TS for pure Ni 300 TS for pure Cu 200 0 20 40 60 80 100 Cu Ni Composition, wt% Ni Adapted from Fig. 9. 6(a), Callister & Rethwisch 8 e. Elongation (%EL) Tensile Strength (MPa) -- Tensile strength (TS) 60 %EL for pure Cu %EL for pure Ni 50 40 30 20 Cu 40 60 80 100 Ni Composition, wt% Ni Adapted from Fig. 9. 6(b), Callister & Rethwisch 8 e. 15
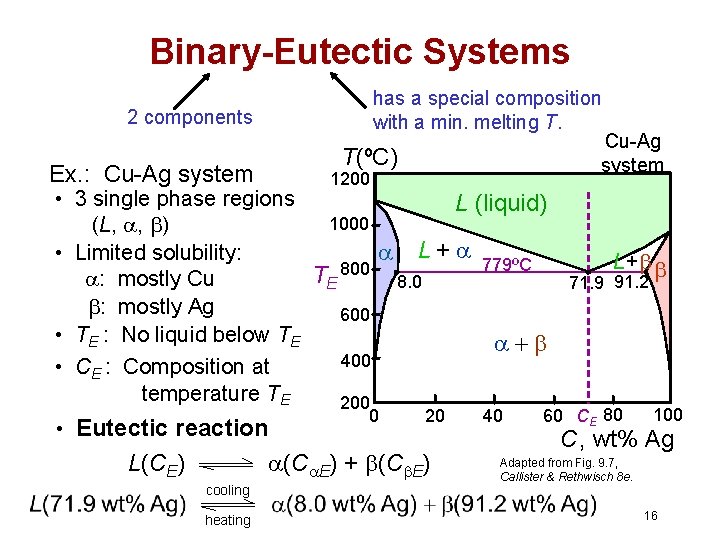
Binary-Eutectic Systems has a special composition with a min. melting T. 2 components Cu-Ag system T(ºC) Ex. : Cu-Ag system 1200 • 3 single phase regions L (liquid) 1000 (L, , ) L + 779ºC • Limited solubility: TE 800 8. 0 : mostly Cu : mostly Ag 600 • TE : No liquid below TE 400 • CE : Composition at temperature TE 200 • Eutectic reaction 0 20 (C E) + (C E) L(CE) cooling heating 40 L+ 71. 9 91. 2 60 CE 80 100 C, wt% Ag Adapted from Fig. 9. 7, Callister & Rethwisch 8 e. 16
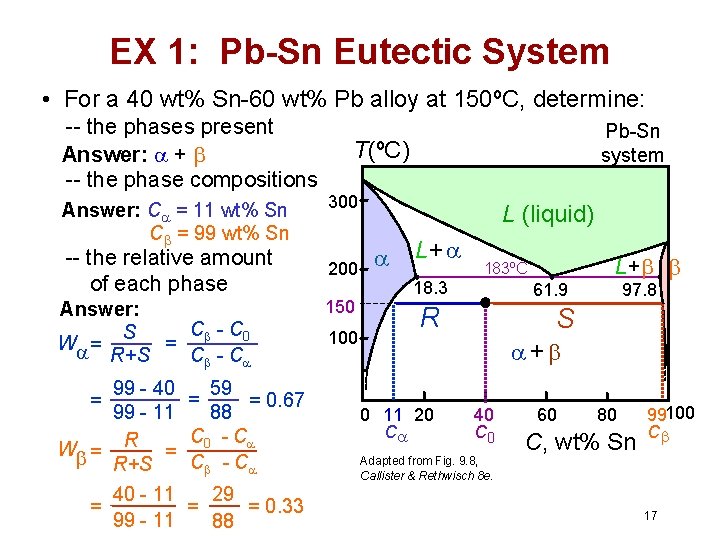
EX 1: Pb-Sn Eutectic System • For a 40 wt% Sn-60 wt% Pb alloy at 150ºC, determine: -- the phases present Answer: + -- the phase compositions Answer: C = 11 wt% Sn C = 99 wt% Sn -- the relative amount of each phase Answer: C - C 0 S = W = R+S C - C 99 - 40 99 - 11 W = R+S 40 - 11 = 99 - 11 = 59 = 0. 67 88 C 0 - C C - C = = 29 = 0. 33 88 Pb-Sn system T(ºC) 300 200 150 100 L (liquid) L+ 18. 3 183ºC 61. 9 R L+ 97. 8 S + 0 11 20 C 40 C 0 Adapted from Fig. 9. 8, Callister & Rethwisch 8 e. 60 80 C, wt% Sn 99100 C 17
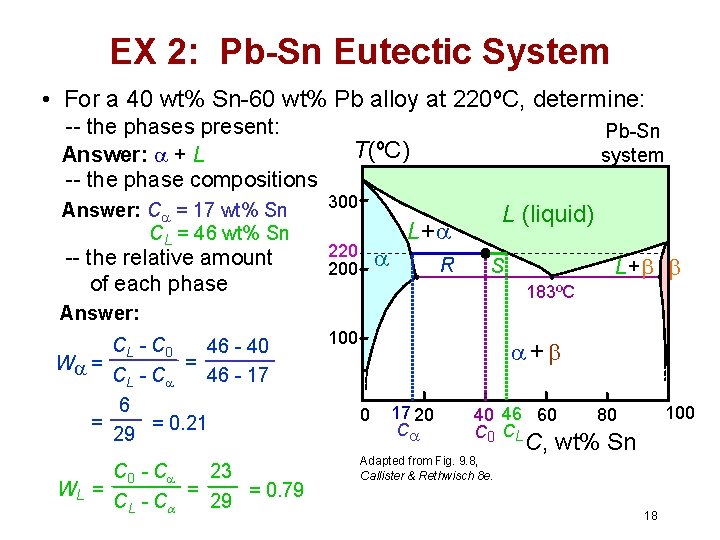
EX 2: Pb-Sn Eutectic System • For a 40 wt% Sn-60 wt% Pb alloy at 220ºC, determine: -- the phases present: Answer: + L -- the phase compositions Answer: C = 17 wt% Sn CL = 46 wt% Sn -- the relative amount of each phase Pb-Sn system T(ºC) 300 220 200 L (liquid) L+ R 183ºC Answer: CL - C 0 46 - 40 = W = CL - C 46 - 17 6 = = 0. 21 29 C 0 - C 23 = WL = = 0. 79 CL - C 29 L+ S 100 + 0 17 20 C 40 46 60 C 0 CL Adapted from Fig. 9. 8, Callister & Rethwisch 8 e. 100 80 C, wt% Sn 18

Microstructural Developments in Eutectic Systems I • For alloys for which C 0 < 2 wt% Sn • Result: at room temperature -- polycrystalline with grains of phase having composition C 0 T(ºC) 400 L L 300 200 TE 100 Adapted from Fig. 9. 11, Callister & Rethwisch 8 e. L: C 0 wt% Sn L+ : C 0 wt% Sn (Pb-Sn System) + 0 10 20 30 C , wt% Sn 2 (room T solubility limit) 19
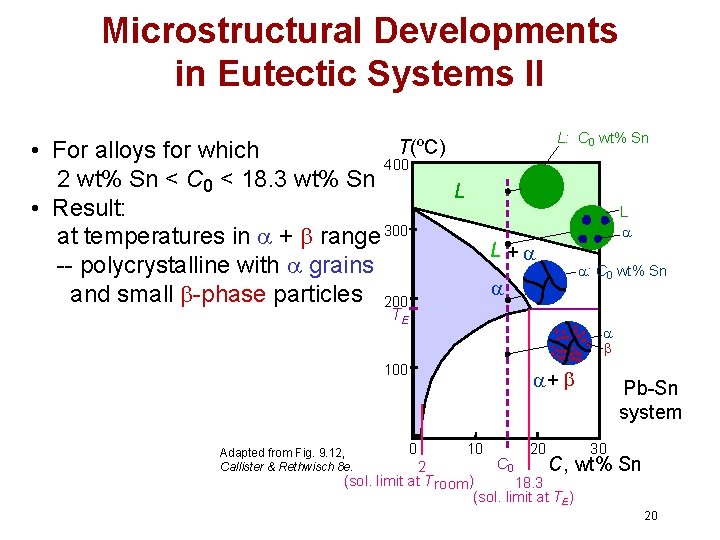
Microstructural Developments in Eutectic Systems II L: C 0 wt% Sn T(ºC) • For alloys for which 400 2 wt% Sn < C 0 < 18. 3 wt% Sn • Result: at temperatures in + range 300 -- polycrystalline with grains and small -phase particles 200 L L + TE : C 0 wt% Sn 100 Adapted from Fig. 9. 12, Callister & Rethwisch 8 e. L + 0 10 20 Pb-Sn system 30 C 0 C, wt% 2 (sol. limit at T room ) 18. 3 (sol. limit at TE) Sn 20
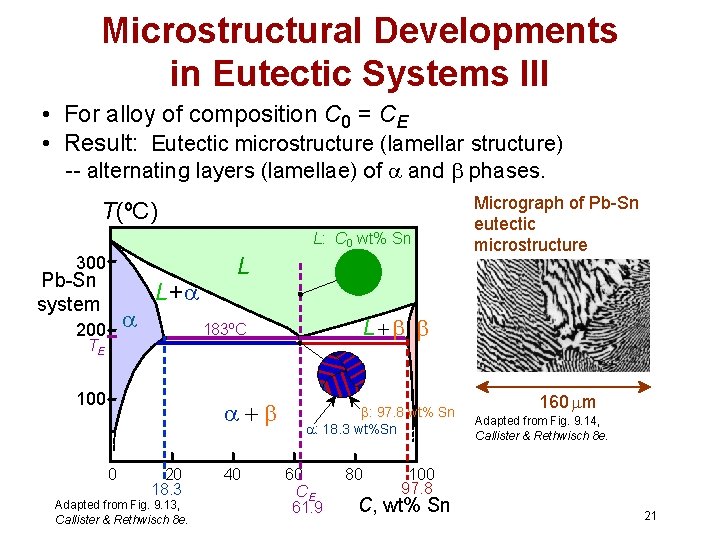
Microstructural Developments in Eutectic Systems III • For alloy of composition C 0 = CE • Result: Eutectic microstructure (lamellar structure) -- alternating layers (lamellae) of and phases. T(ºC) L: C 0 wt% Sn 300 Pb-Sn system 200 L+ L 100 0 L 183ºC TE 20 18. 3 Adapted from Fig. 9. 13, Callister & Rethwisch 8 e. 40 Micrograph of Pb-Sn eutectic microstructure : 97. 8 wt% Sn : 18. 3 wt%Sn 60 CE 61. 9 80 160 m Adapted from Fig. 9. 14, Callister & Rethwisch 8 e. 100 97. 8 C, wt% Sn 21
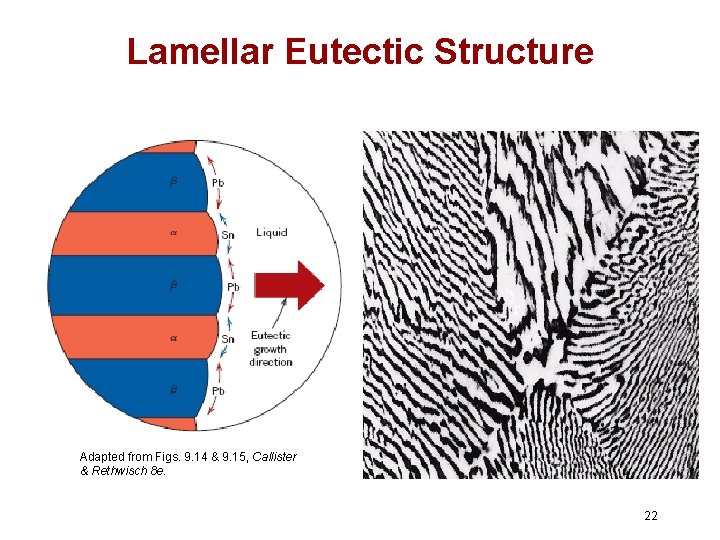
Lamellar Eutectic Structure Adapted from Figs. 9. 14 & 9. 15, Callister & Rethwisch 8 e. 22
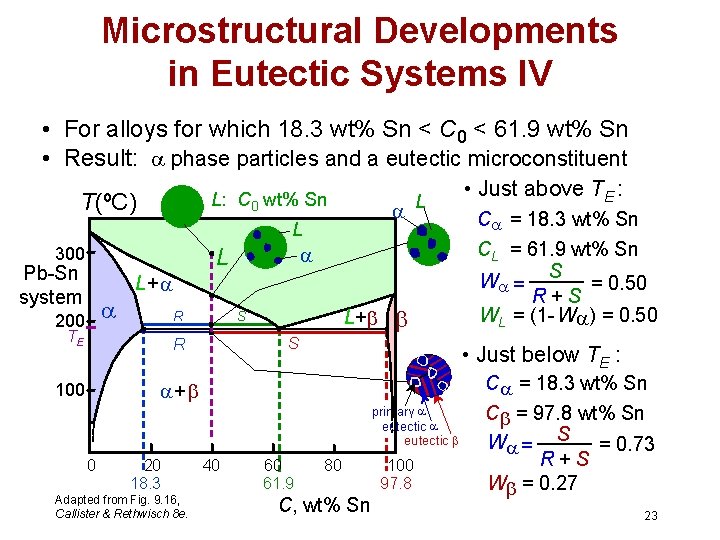
Microstructural Developments in Eutectic Systems IV • For alloys for which 18. 3 wt% Sn < C 0 < 61. 9 wt% Sn • Result: phase particles and a eutectic microconstituent T(ºC) L: C 0 wt% Sn L L 300 L Pb-Sn system 200 L+ R TE L+ S S R C = 18. 3 wt% Sn primary eutectic 0 20 18. 3 Adapted from Fig. 9. 16, Callister & Rethwisch 8 e. C = 18. 3 wt% Sn CL = 61. 9 wt% Sn W = S = 0. 50 R+S WL = (1 - W ) = 0. 50 • Just below TE : + 100 • Just above TE : 40 60 61. 9 80 C, wt% Sn 100 97. 8 C = 97. 8 wt% Sn W = S = 0. 73 R+S W = 0. 27 23
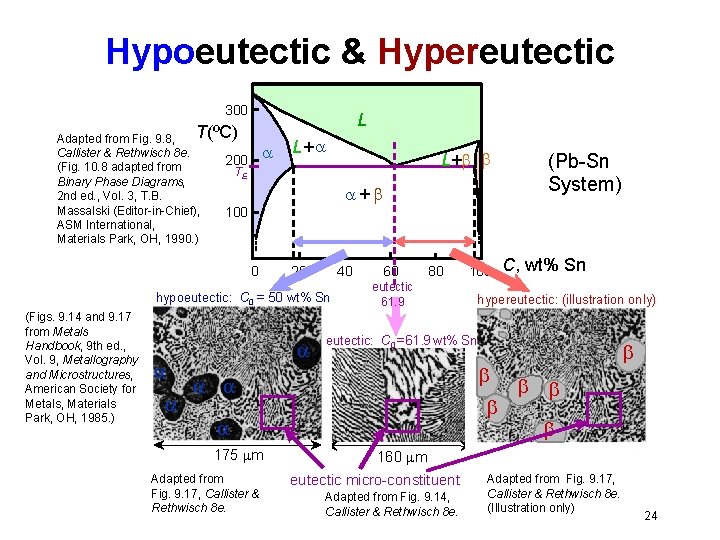
Hypoeutectic & Hypereutectic 300 L T(ºC) Adapted from Fig. 9. 8, Callister & Rethwisch 8 e. (Fig. 10. 8 adapted from Binary Phase Diagrams, 2 nd ed. , Vol. 3, T. B. Massalski (Editor-in-Chief), ASM International, Materials Park, OH, 1990. ) 200 L+ + 100 0 20 40 hypoeutectic: C 0 = 50 wt% Sn (Figs. 9. 14 and 9. 17 from Metals Handbook, 9 th ed. , Vol. 9, Metallography and Microstructures, American Society for Metals, Materials Park, OH, 1985. ) L+ TE 60 80 eutectic 61. 9 hypereutectic: (illustration only) Adapted from Fig. 9. 17, Callister & Rethwisch 8 e. C, wt% Sn eutectic: C 0 = 61. 9 wt% Sn 175 m 100 (Pb-Sn System) 160 m eutectic micro-constituent Adapted from Fig. 9. 14, Callister & Rethwisch 8 e. Adapted from Fig. 9. 17, Callister & Rethwisch 8 e. (Illustration only) 24
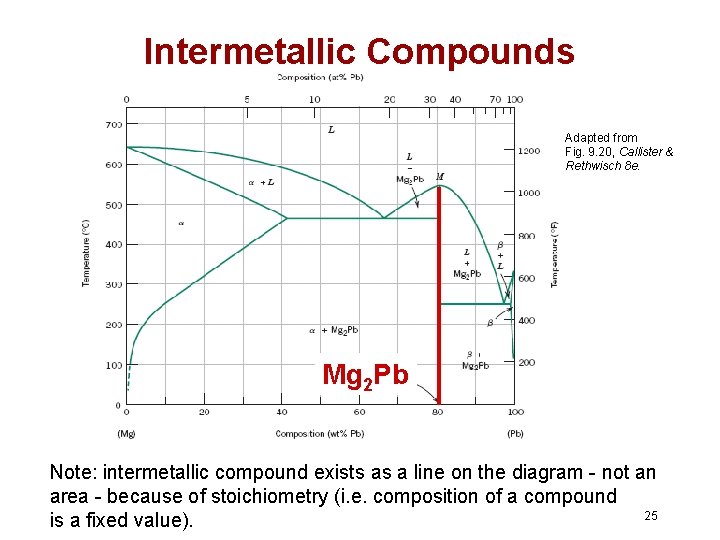
Intermetallic Compounds Adapted from Fig. 9. 20, Callister & Rethwisch 8 e. Mg 2 Pb Note: intermetallic compound exists as a line on the diagram - not an area - because of stoichiometry (i. e. composition of a compound 25 is a fixed value).

Eutectic, Eutectoid, & Peritectic • Eutectic - liquid transforms to two solid phases cool L heat + (For Pb-Sn, 183ºC, 61. 9 wt% Sn) • Eutectoid – one solid phase transforms to two other solid phases intermetallic compound - cementite S 2 S 1+S 3 cool + Fe 3 C (For Fe-C, 727ºC, 0. 76 wt% C) heat • Peritectic - liquid and one solid phase transform to a second solid phase S 1 + L S 2 +L cool heat (For Fe-C, 1493ºC, 0. 16 wt% C) 26
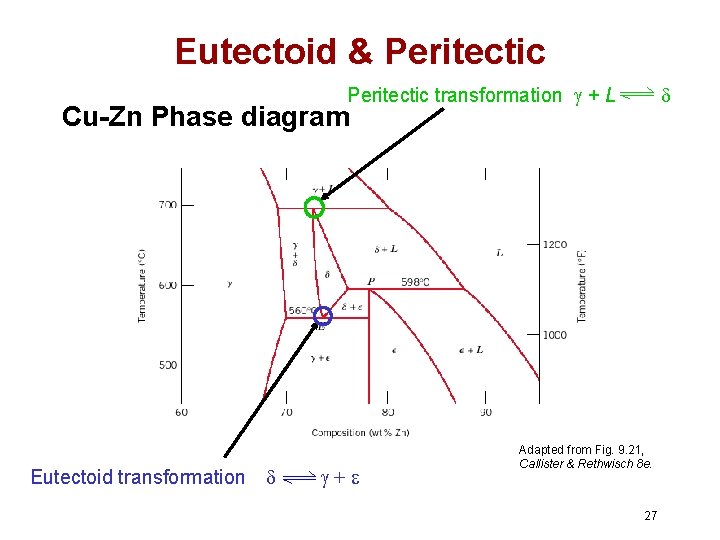
Eutectoid & Peritectic transformation + L Cu-Zn Phase diagram Eutectoid transformation + Adapted from Fig. 9. 21, Callister & Rethwisch 8 e. 27
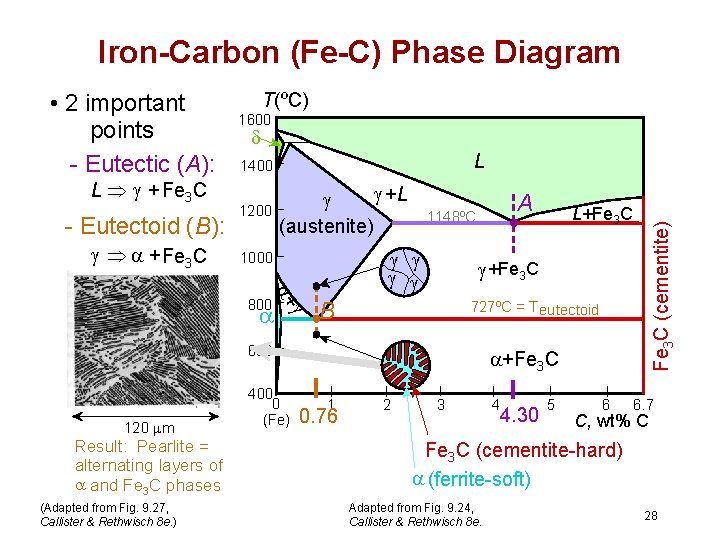
Iron-Carbon (Fe-C) Phase Diagram L Þ + Fe 3 C - Eutectoid (B): Þ + Fe 3 C T(ºC) 1600 L 1400 1200 +L (austenite) 1000 + 800 A 1148ºC +Fe 3 C B 727ºC = T eutectoid 600 120 m Result: Pearlite = alternating layers of and Fe 3 C phases (Adapted from Fig. 9. 27, Callister & Rethwisch 8 e. ) 400 0 (Fe) L+Fe 3 C (cementite) • 2 important points - Eutectic (A): +Fe 3 C 1 0. 76 2 3 4 4. 30 5 6 6. 7 C, wt% C Fe 3 C (cementite-hard) (ferrite-soft) Adapted from Fig. 9. 24, Callister & Rethwisch 8 e. 28
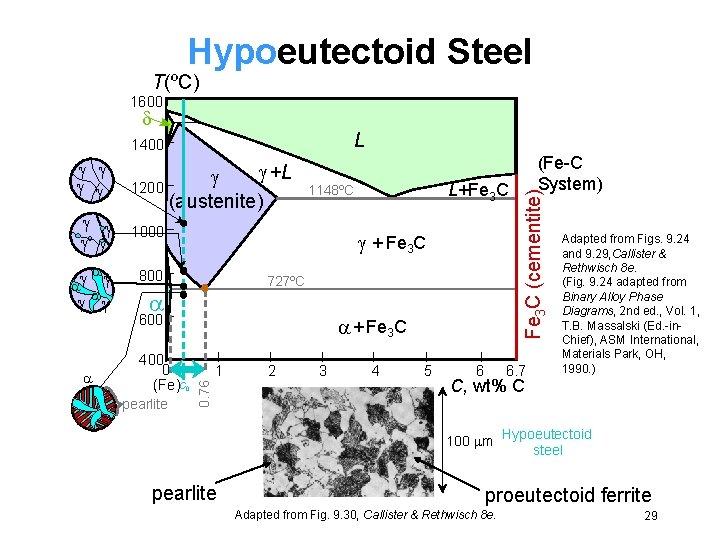
Hypoeutectoid Steel T(ºC) 1600 L +L 1200 (austenite) + Fe 3 C 800 727ºC 600 400 0 (Fe)C 0 pearlite L+Fe 3 C 1148ºC 1000 + Fe 3 C 1 0. 76 2 3 4 5 6 6. 7 Adapted from Figs. 9. 24 and 9. 29, Callister & Rethwisch 8 e. (Fig. 9. 24 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in. Chief), ASM International, Materials Park, OH, 1990. ) C, wt% C 100 m pearlite (Fe-C System) Fe 3 C (cementite) 1400 Hypoeutectoid steel proeutectoid ferrite Adapted from Fig. 9. 30, Callister & Rethwisch 8 e. 29
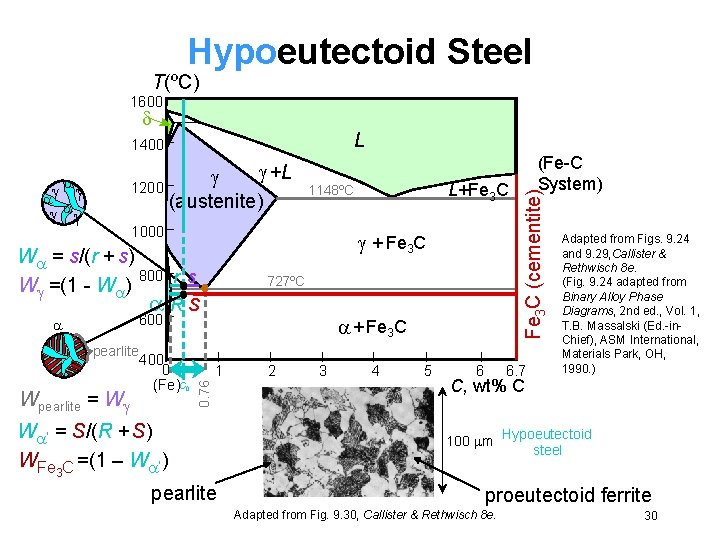
Hypoeutectoid Steel T(ºC) 1600 L +L 1200 (austenite) 1000 + Fe 3 C W = s/(r + s) 800 r s W =(1 - W ) 727ºC RS 600 pearlite Wpearlite = W 400 0 (Fe)C 0 + Fe 3 C 1 0. 76 L+Fe 3 C 1148ºC W ’ = S/(R + S) WFe 3 C =(1 – W ’) pearlite 2 3 4 5 6 (Fe-C System) Fe 3 C (cementite) 1400 6. 7 Adapted from Figs. 9. 24 and 9. 29, Callister & Rethwisch 8 e. (Fig. 9. 24 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in. Chief), ASM International, Materials Park, OH, 1990. ) C, wt% C 100 m Hypoeutectoid steel proeutectoid ferrite Adapted from Fig. 9. 30, Callister & Rethwisch 8 e. 30
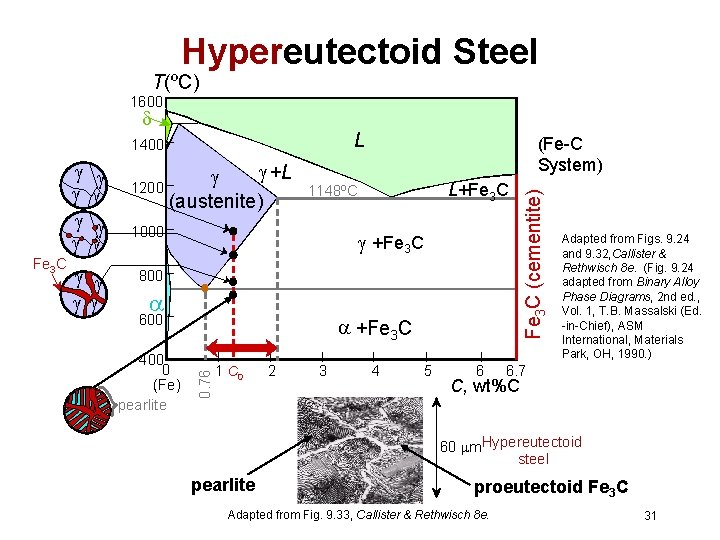
Hypereutectoid Steel T(ºC) 1600 L Fe 3 C +L 1200 (austenite) 1000 L+Fe 3 C 1148ºC +Fe 3 C 800 600 400 0 (Fe) pearlite +Fe 3 C 0. 76 (Fe-C System) 1 C 0 2 3 4 5 6 Fe 3 C (cementite) 1400 Adapted from Figs. 9. 24 and 9. 32, Callister & Rethwisch 8 e. (Fig. 9. 24 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in-Chief), ASM International, Materials Park, OH, 1990. ) 6. 7 C, wt%C 60 m. Hypereutectoid steel pearlite proeutectoid Fe 3 C Adapted from Fig. 9. 33, Callister & Rethwisch 8 e. 31
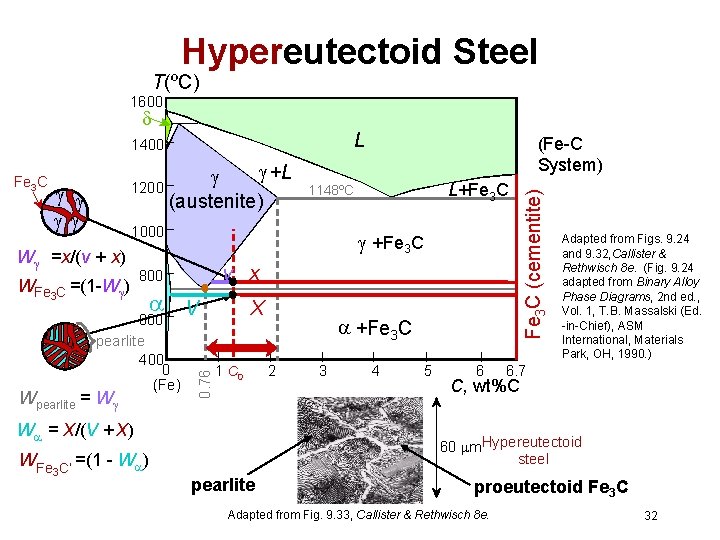
Hypereutectoid Steel T(ºC) 1600 L +L 1200 (austenite) W =x/(v + x) WFe 3 C =(1 -W ) +Fe 3 C v x 800 V 600 pearlite 400 0 (Fe) Wpearlite = W X 1 C 0 W = X/(V + X) WFe 3 C’ L+Fe 3 C 1148ºC 1000 0. 76 Fe 3 C (Fe-C System) +Fe 3 C 2 3 4 5 6 Fe 3 C (cementite) 1400 Adapted from Figs. 9. 24 and 9. 32, Callister & Rethwisch 8 e. (Fig. 9. 24 adapted from Binary Alloy Phase Diagrams, 2 nd ed. , Vol. 1, T. B. Massalski (Ed. -in-Chief), ASM International, Materials Park, OH, 1990. ) 6. 7 C, wt%C 60 m. Hypereutectoid steel =(1 - W ) pearlite proeutectoid Fe 3 C Adapted from Fig. 9. 33, Callister & Rethwisch 8 e. 32

Example Problem For a 99. 6 wt% Fe-0. 40 wt% C steel at a temperature just below the eutectoid, determine the following: a) The compositions of Fe 3 C and ferrite ( ). b) The amount of cementite (in grams) that forms in 100 g of steel. c) The amounts of pearlite and proeutectoid ferrite ( ) in the 100 g. 33
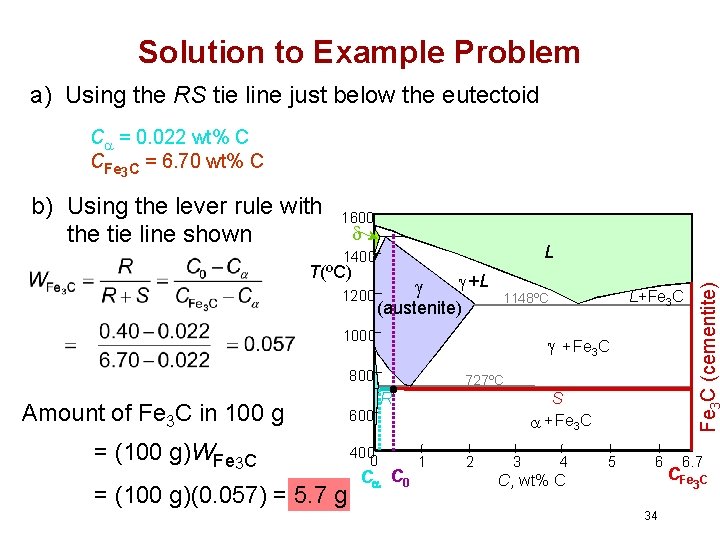
Solution to Example Problem a) Using the RS tie line just below the eutectoid C = 0. 022 wt% C CFe 3 C = 6. 70 wt% C 1600 L 1400 T(ºC) 1200 +L (austenite) + Fe 3 C 800 Amount of Fe 3 C in 100 g = (100 g)WFe 3 C = (100 g)(0. 057) = 5. 7 g 727ºC R S + Fe 3 C 600 400 0 L+Fe 3 C 1148ºC 1000 Fe C (cementite) b) Using the lever rule with the tie line shown C C 0 1 2 3 4 5 6 C , wt% C 6. 7 CFe 3 C 34
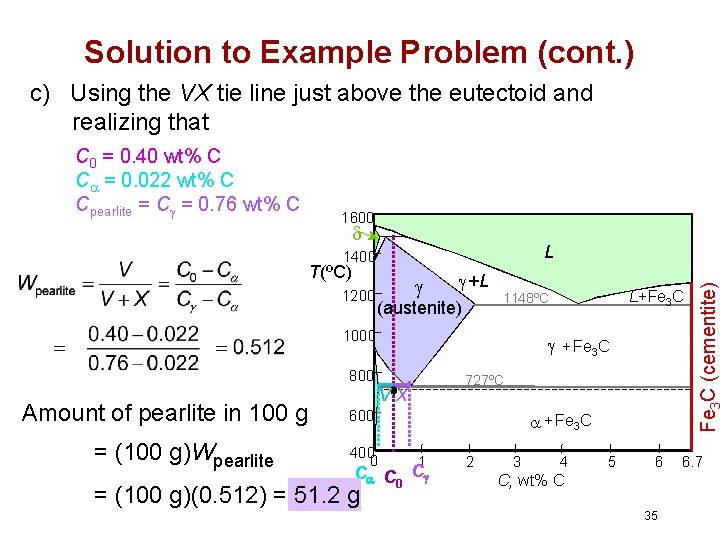
Solution to Example Problem (cont. ) c) Using the VX tie line just above the eutectoid and realizing that 1600 L 1400 T(ºC) 1200 +L (austenite) 1000 + Fe 3 C 800 Amount of pearlite in 100 g = (100 g)Wpearlite 727ºC VX 600 400 0 + Fe 3 C 1 C C 0 C = (100 g)(0. 512) = 51. 2 g L+Fe 3 C 1148ºC 2 3 4 5 6 C, wt% C 35 Fe C (cementite) C 0 = 0. 40 wt% C C = 0. 022 wt% C Cpearlite = C = 0. 76 wt% C 6. 7
VMSE: Interactive Phase Diagrams Microstructure, phase compositions, and phase fractions respond interactively Change alloy composition 36
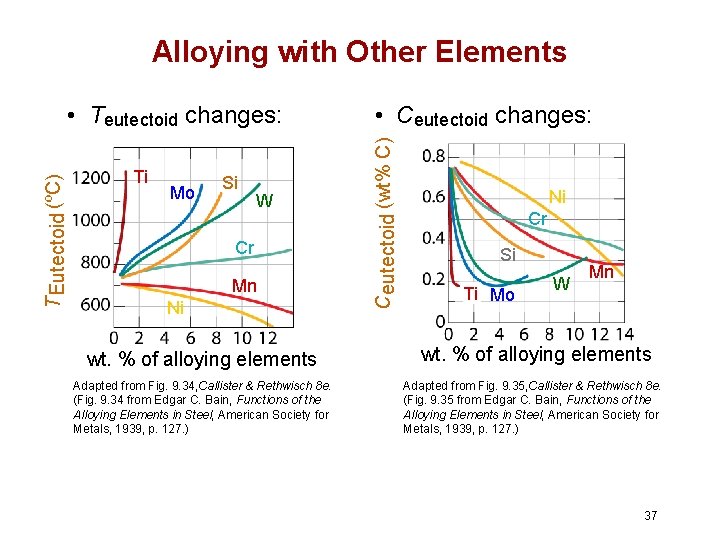
Alloying with Other Elements Ti Mo Si W Cr Mn Ni wt. % of alloying elements Adapted from Fig. 9. 34, Callister & Rethwisch 8 e. (Fig. 9. 34 from Edgar C. Bain, Functions of the Alloying Elements in Steel, American Society for Metals, 1939, p. 127. ) • Ceutectoid changes: Ceutectoid (wt% C) T Eutectoid (ºC) • Teutectoid changes: Ni Cr Si Ti Mo W Mn wt. % of alloying elements Adapted from Fig. 9. 35, Callister & Rethwisch 8 e. (Fig. 9. 35 from Edgar C. Bain, Functions of the Alloying Elements in Steel, American Society for Metals, 1939, p. 127. ) 37

Summary • Phase diagrams are useful tools to determine: -- the number and types of phases present, -- the composition of each phase, -- and the weight fraction of each phase given the temperature and composition of the system. • The microstructure of an alloy depends on -- its composition, and -- whether or not cooling rate allows for maintenance of equilibrium. • Important phase diagram phase transformations include eutectic, eutectoid, and peritectic. 38
- Slides: 38