Phase Diagrams Binary Eutectoid Systems IronCarbide Phase Diagram

Phase Diagrams Binary Eutectoid Systems Iron-Carbide Phase Diagram Steels and Cast Iron Weeks 7 - 8 1

What is Phase? • The term ‘phase’ refers to a separate and identifiable state of matter in which a given substance may exist. • Applicable to both crystalline and non-crystalline materials • An important refractory oxide silica is able to exist as three crystalline phases, quartz, tridymite and cristobalite, as well as a non-crystalline phase, silica glass, and as molten silica • Every pure material is considered to be a phase, so also is every solid, liquid, and gaseous solution • For example, the sugar–water syrup solution is one phase, and solid sugar is another 2

Introduction to Phase Diagram • There is a strong correlation between microstructure and mechanical properties, and the development of microstructure of an alloy is related to the characteristics of its phase diagram • It is a type of chart used to show conditions at which thermodynamically distinct phases can occur at equilibrium • Provides valuable information about melting, casting, crystallization, and other phenomena 3

ISSUES TO ADDRESS. . . • When we combine two elements. . . what equilibrium state do we get? • In particular, if we specify. . . --a composition (e. g. , wt% Cu - wt% Ni), and --a temperature (T ) then. . . How many phases do we get? What is the composition of each phase? How much of each phase do we get? Phase B Phase A Nickel atom Copper atom 4

Solubility Limit • At some specific temperature, there is a maximum concentration of solute atoms that may dissolve in the solvent to form a solid solution, which is called as Solubility Limit • The addition of solute in excess of this solubility limit results in the formation of another compound that has a distinctly different composition • This solubility limit depends on the temperature 5
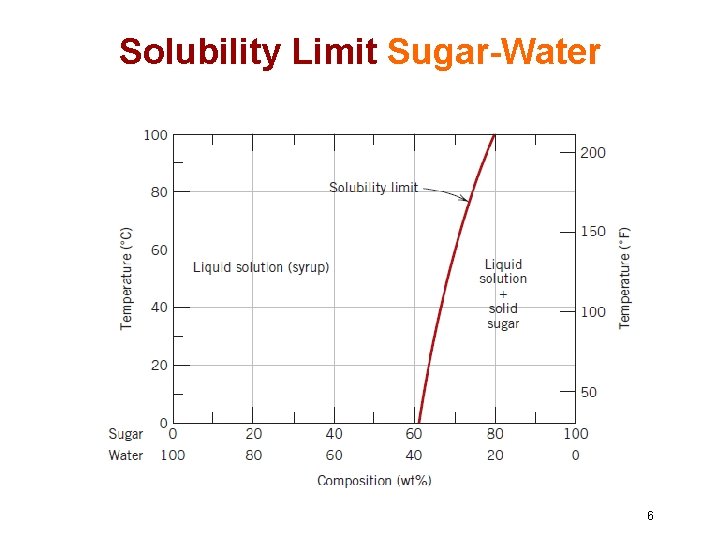
Solubility Limit Sugar-Water 6

Microstructure • the structure of a prepared surface of material as revealed by a microscope above 25× magnification • The microstructure of a material can strongly influence properties such as strength, toughness, ductility, hardness, corrosion resistance, high/low temperature behavior, wear resistance, etc 7

Components and Phases • Components: The elements or compounds which are present in the mixture (e. g. , Al and Cu) • Phases: The physically and chemically distinct material regions that result (e. g. , and ). Aluminum. Copper Alloy (lighter phase) (darker phase) 8
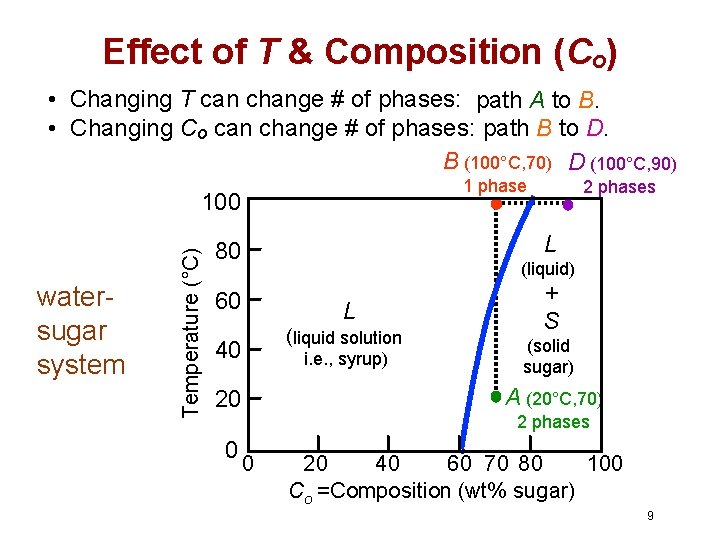
Effect of T & Composition (Co) • Changing T can change # of phases: path A to B. • Changing Co can change # of phases: path B to D. B (100°C, 70) D (100°C, 90) 1 phase watersugar system Temperature (°C) 100 L 80 (liquid) 60 L (liquid solution 40 i. e. , syrup) + S (solid sugar) A (20°C, 70) 20 0 2 phases 0 20 40 60 70 80 100 Co =Composition (wt% sugar) 9

PHASE EQUILIBRIA • Free Energy -> a function of the internal energy of a system, and also the disorder of the atoms or molecules (or entropy) • A system is at equilibrium if its free energy is at a minimum under some specified combination of temperature, pressure, and composition • A change in temperature, pressure, and/or composition for a system in equilibrium will result in an increase in the free energy • And in a possible spontaneous change to another state whereby the free energy is lowered 10

Unary Phase Diagram • Three externally controllable parameters that will affect phase structure: temperature, pressure, and composition • The simplest type of phase diagram to understand is that for a one-component system, in which composition is held constant • Pure water exists in three phases: solid, liquid and vapor 11
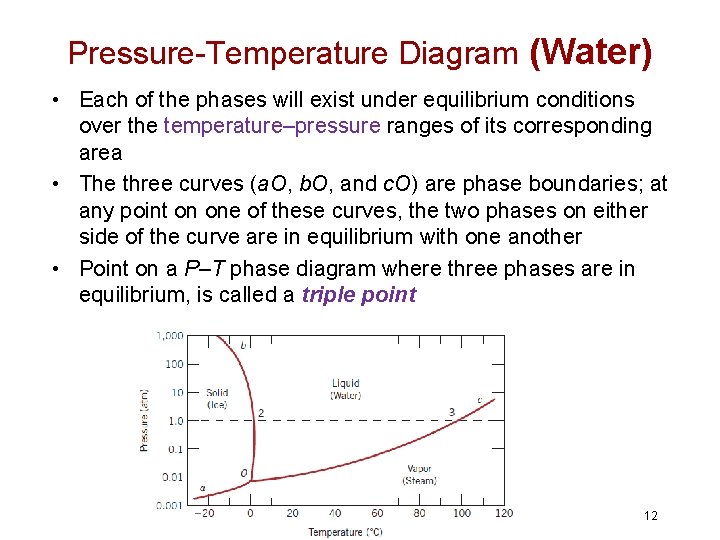
Pressure-Temperature Diagram (Water) • Each of the phases will exist under equilibrium conditions over the temperature–pressure ranges of its corresponding area • The three curves (a. O, b. O, and c. O) are phase boundaries; at any point on one of these curves, the two phases on either side of the curve are in equilibrium with one another • Point on a P–T phase diagram where three phases are in equilibrium, is called a triple point 12

Binary Phase Diagrams • A phase diagram in which temperature and composition are variable parameters, and pressure is held constant—normally 1 atm • Binary phase diagrams are maps that represent the relationships between temperature and the compositions and quantities of phases at equilibrium, which influence the microstructure of an alloy. • Many microstructures develop from phase transformations, the changes that occur when the temperature is altered 13

Phase Equilibria Simple solution system (e. g. , Ni-Cu solution) Crystal Structure electroneg r (nm) Ni FCC 1. 9 0. 1246 Cu FCC 1. 8 0. 1278 • Both have the same crystal structure (FCC) and have similar electronegativities and atomic radii (W. Hume – Rothery rules) suggesting high mutual solubility. • Ni and Cu are totally miscible in all proportions. 14
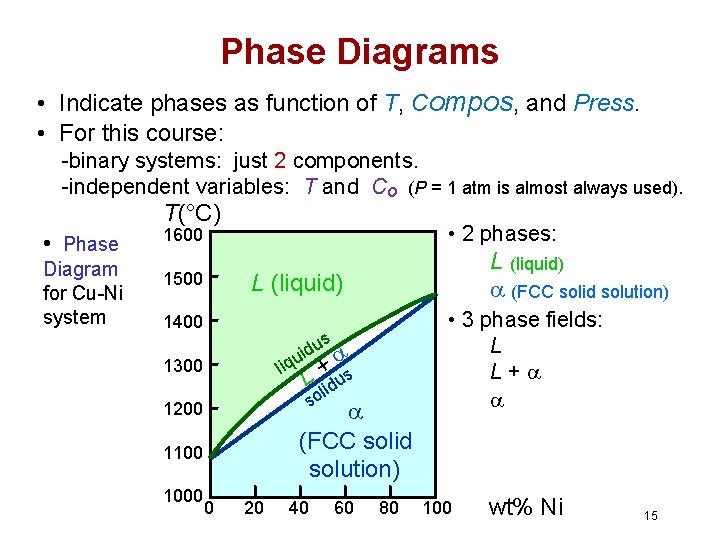
Phase Diagrams • Indicate phases as function of T, Compos, and Press. • For this course: -binary systems: just 2 components. -independent variables: T and Co (P = 1 atm is almost always used). T(°C) • Phase Diagram for Cu-Ni system • 2 phases: 1600 1500 L (liquid) 1400 us d i u liq + s L lidu so 1300 (FCC solid solution) 1200 1100 1000 L (liquid) (FCC solid solution) 0 20 40 60 80 • 3 phase fields: L L+ 100 wt% Ni 15
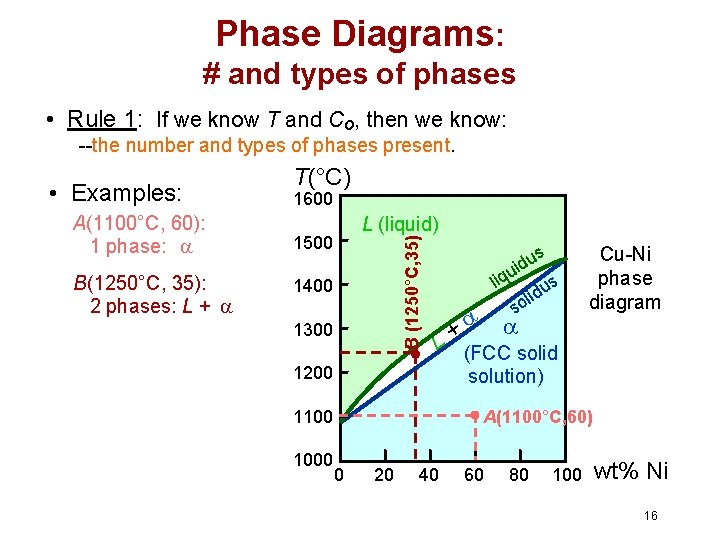
Phase Diagrams: # and types of phases • Rule 1: If we know T and Co, then we know: --the number and types of phases present. A(1100°C, 60): 1 phase: B(1250°C, 35): 2 phases: L + 1600 L (liquid) 1500 B (1250°C, 35) • Examples: T(°C) 1400 1300 + L 1200 1100 1000 us d i u liq us d i l so Cu-Ni phase diagram (FCC solid solution) A(1100°C, 60) 0 20 40 60 80 100 wt% Ni 16
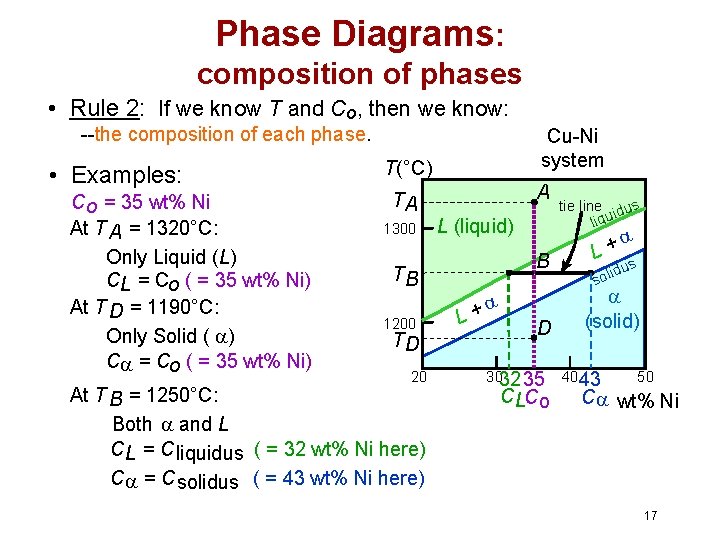
Phase Diagrams: composition of phases • Rule 2: If we know T and Co, then we know: --the composition of each phase. • Examples: T(°C) Cu-Ni system A TA Co = 35 wt% Ni 1300 L (liquid) At T A = 1320°C: Only Liquid (L) B TB CL = Co ( = 35 wt% Ni) At T D = 1190°C: + L 1200 D Only Solid ( ) TD C = Co ( = 35 wt% Ni) 20 3032 35 At T B = 1250°C: CLCo Both and L CL = C liquidus ( = 32 wt% Ni here) C = C solidus ( = 43 wt% Ni here) tie line dus i liqu L+ s idu l o s (solid) 4043 50 C wt% Ni 17
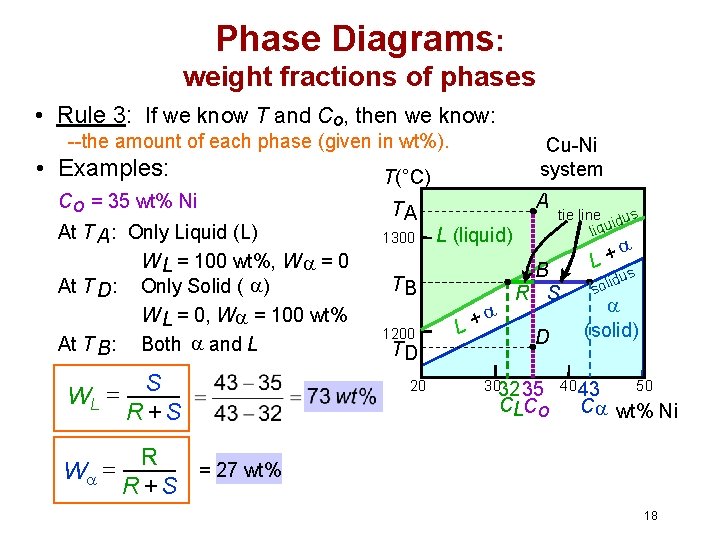
Phase Diagrams: weight fractions of phases • Rule 3: If we know T and Co, then we know: --the amount of each phase (given in wt%). • Examples: Co = 35 wt% Ni At T A : Only Liquid (L) W L = 100 wt%, W = 0 At T D: Only Solid ( ) W L = 0, W = 100 wt% At T B : Both and L WL = S R +S W = R = 27 wt% R +S Cu-Ni system T(°C) TA 1300 A L (liquid) TB 1200 TD 20 + L tie line dus i liqu B R S D 3032 35 CLCo + L s idu l o s (solid) 40 43 50 C wt% Ni 18

The Lever Rule • Tie line – connects the phases in equilibrium with each other - essentially an isotherm T(°C) 1300 tie line dus i liqu L (liquid) B TB 1200 20 + L M ML us id sol L+ R How much of each phase? Think of it as a lever (teeter-totter) S (solid) 30 C C 40 C L o R S 50 wt% Ni 19
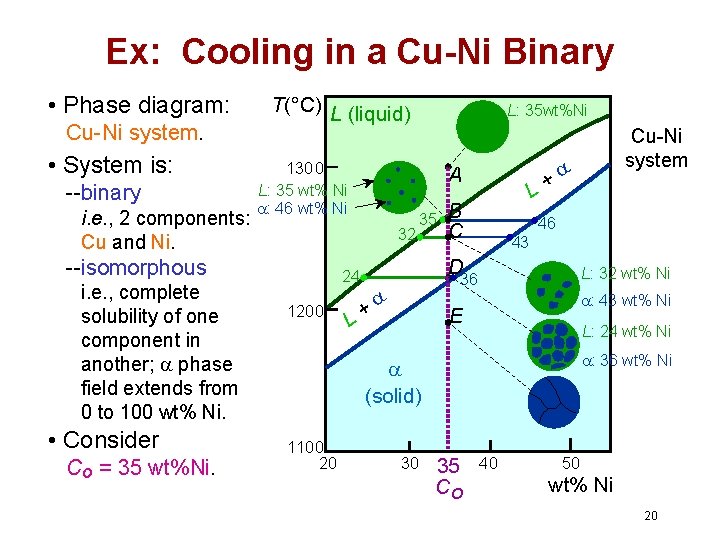
Ex: Cooling in a Cu-Ni Binary • Phase diagram: Cu-Ni system. • System is: --binary i. e. , 2 components: Cu and Ni. T(°C) L (liquid) 1300 L: 35 wt% Ni : 46 wt% Ni i. e. , complete solubility of one component in another; phase field extends from 0 to 100 wt% Ni. • Consider Co = 35 wt%Ni. A 32 --isomorphous L: 35 wt%Ni 35 B C 46 43 D 24 1200 L+ L: 32 wt% Ni 36 + L : 43 wt% Ni E L: 24 wt% Ni : 36 wt% Ni (solid) 1100 20 30 Cu-Ni system 35 Co 40 50 wt% Ni 20

Cored vs Equilibrium Phases • C changes as we solidify. • Cu-Ni case: First to solidify has C = 46 wt% Ni. Last to solidify has C = 35 wt% Ni. • Fast rate of cooling: Cored structure • Slow rate of cooling: Equilibrium structure First to solidify: 46 wt% Ni Last to solidify: < 35 wt% Ni Uniform C : 35 wt% Ni 21

Mechanical Properties: Cu-Ni System • Effect of solid solution strengthening on: --Ductility (%EL, %AR) 400 TS for pure Ni 300 TS for pure Cu 200 0 20 40 60 80 100 Cu Ni Composition, wt% Ni --Peak as a function of Co Elongation (%EL) Tensile Strength (MPa) --Tensile strength (TS) 60 %EL for pure Cu %EL for pure Ni 50 40 30 20 Cu 40 60 80 100 Ni Composition, wt% Ni --Min. as a function of Co 22

Eutectic System A eutectic system is a mixture of chemical compounds or elements that has a single chemical composition that solidifies at a lower temperature than any other composition 23
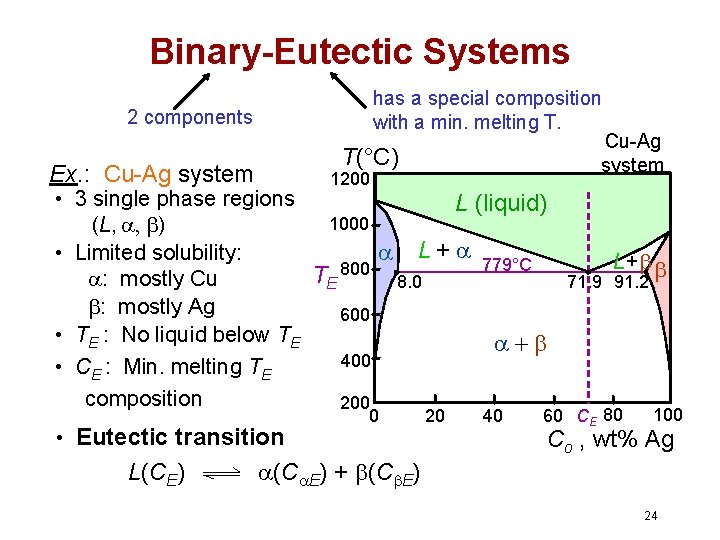
Binary-Eutectic Systems has a special composition with a min. melting T. 2 components Cu-Ag system T(°C) Ex. : Cu-Ag system 1200 • 3 single phase regions L (liquid) 1000 (L, , ) L + 779°C • Limited solubility: L+ 800 T : mostly Cu 8. 0 71. 9 91. 2 E : mostly Ag 600 • TE : No liquid below TE 400 • CE : Min. melting TE composition 200 • Eutectic transition L(CE) 0 (C E) + (C E) 20 40 60 CE 80 100 Co , wt% Ag 24
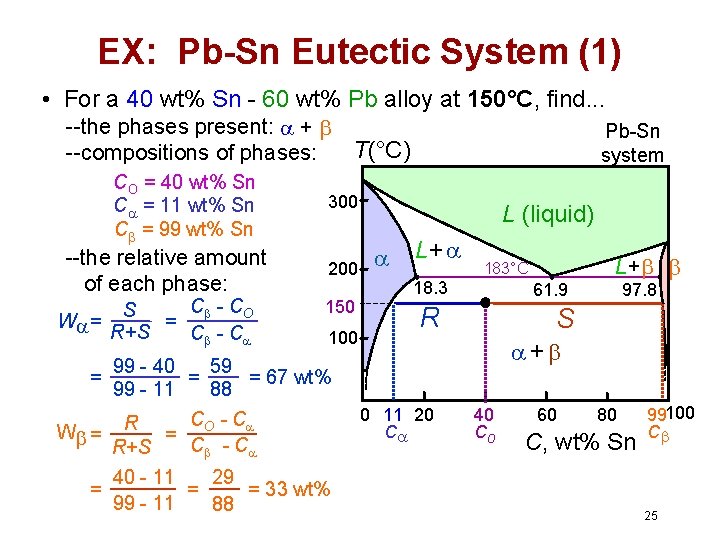
EX: Pb-Sn Eutectic System (1) • For a 40 wt% Sn - 60 wt% Pb alloy at 150°C, find. . . --the phases present: + T(°C) --compositions of phases: CO = 40 wt% Sn C = 11 wt% Sn C = 99 wt% Sn --the relative amount of each phase: W = C - CO S = R+S C - C Pb-Sn system 300 200 150 100 99 - 40 59 = = 67 wt% 99 - 11 88 C - C W = R = O C - C R+S L (liquid) L+ 18. 3 183°C 61. 9 R L+ 97. 8 S + = = 40 - 11 29 = = 33 wt% 99 - 11 88 0 11 20 C 40 Co 60 80 C, wt% Sn 99100 C 25
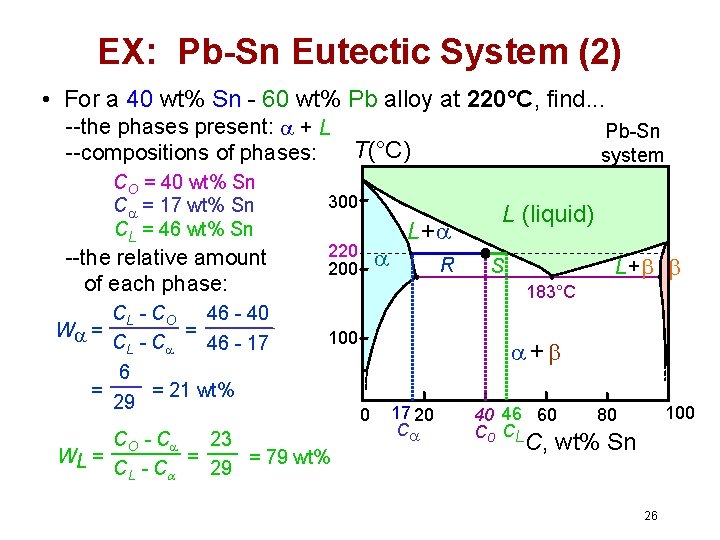
EX: Pb-Sn Eutectic System (2) • For a 40 wt% Sn - 60 wt% Pb alloy at 220°C, find. . . --the phases present: + L T(°C) --compositions of phases: CO = 40 wt% Sn C = 17 wt% Sn CL = 46 wt% Sn --the relative amount of each phase: CL - C O 46 - 40 = W = CL - C 46 - 17 6 = = 21 wt% 29 Pb-Sn system 300 220 200 L+ R L (liquid) L+ S 183°C 100 CO - C 23 = WL = = 79 wt% CL - C 29 + 0 17 20 C 40 46 60 Co CL 100 80 C, wt% Sn 26

Microstructures in Eutectic Systems: I • Co < 2 wt% Sn • Result: --at extreme ends --polycrystal of grains i. e. , only one solid phase. T(°C) L: Co wt% Sn 400 L L 300 200 L+ (Pb-Sn System) : Co wt% Sn TE 100 + 0 Co 10 20 30 Co, wt% Sn 2 (room T solubility limit) 27
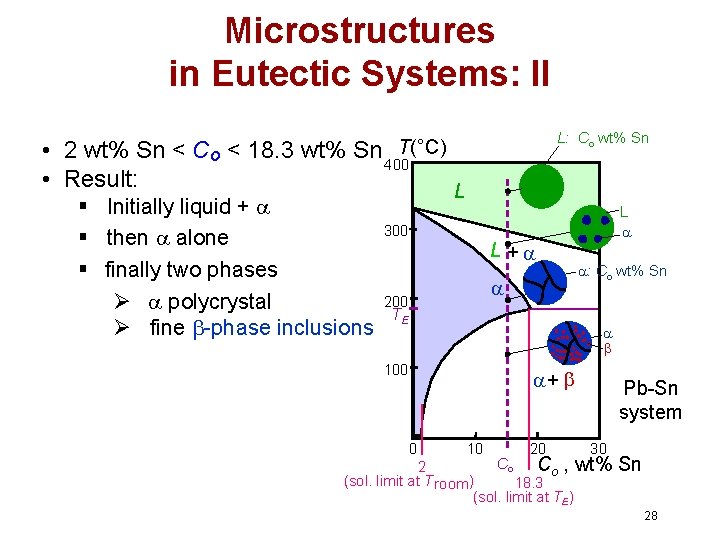
Microstructures in Eutectic Systems: II L: Co wt% Sn T(°C) • 2 wt% Sn < Co < 18. 3 wt% Sn 400 • Result: § Initially liquid + § then alone § finally two phases Ø polycrystal Ø fine -phase inclusions L L 300 L + 200 TE : Co wt% Sn 100 + 0 10 20 Pb-Sn system 30 Co Co , wt% 2 (sol. limit at T room ) 18. 3 (sol. limit at TE) Sn 28
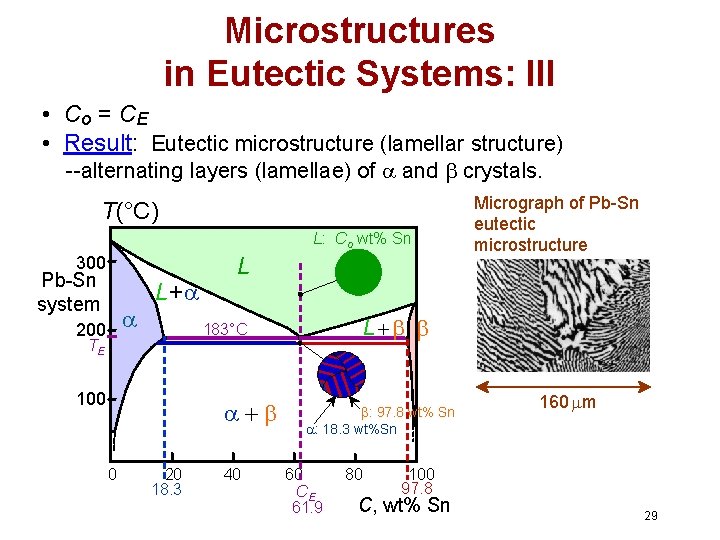
Microstructures in Eutectic Systems: III • Co = CE • Result: Eutectic microstructure (lamellar structure) --alternating layers (lamellae) of and crystals. T(°C) L: Co wt% Sn 300 Pb-Sn system 200 L+ L 100 0 L 183°C TE 20 18. 3 40 Micrograph of Pb-Sn eutectic microstructure : 97. 8 wt% Sn : 18. 3 wt%Sn 60 CE 61. 9 80 160 m 100 97. 8 C, wt% Sn 29
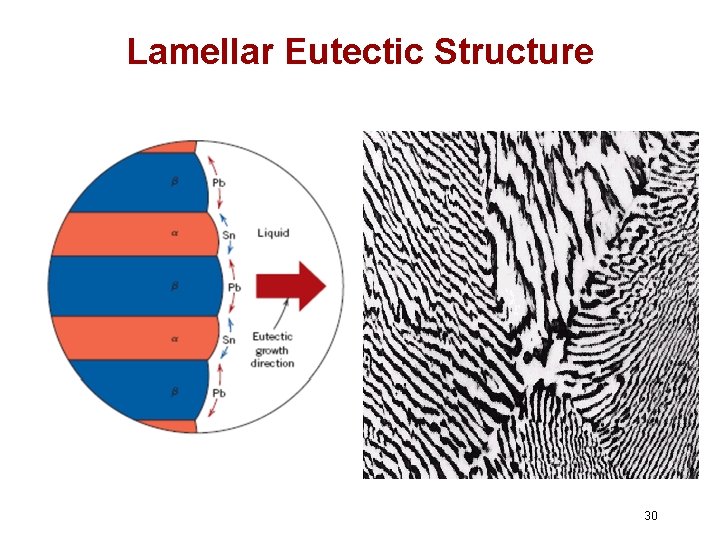
Lamellar Eutectic Structure 30
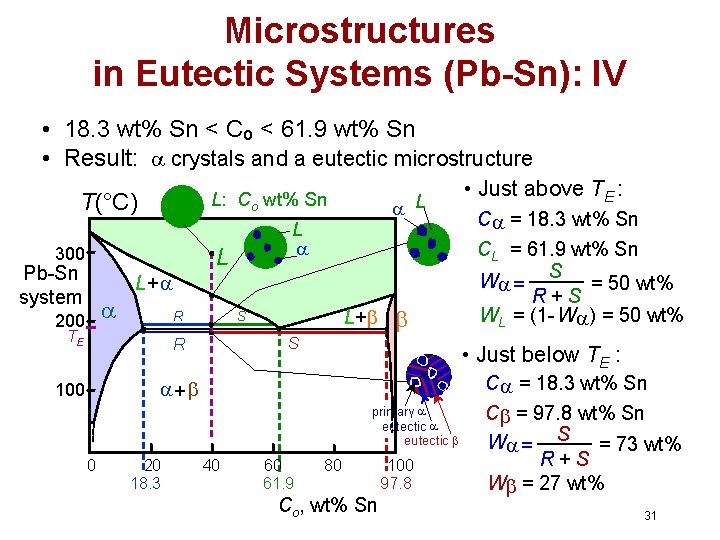
Microstructures in Eutectic Systems (Pb-Sn): IV • 18. 3 wt% Sn < Co < 61. 9 wt% Sn • Result: crystals and a eutectic microstructure T(°C) L: Co wt% Sn 300 L L Pb-Sn system 200 L L+ R TE L+ S S R C = 18. 3 wt% Sn primary eutectic 0 20 18. 3 C = 18. 3 wt% Sn CL = 61. 9 wt% Sn W = S = 50 wt% R+S WL = (1 - W ) = 50 wt% • Just below TE : + 100 • Just above TE : 40 60 61. 9 80 Co, wt% Sn 100 97. 8 C = 97. 8 wt% Sn W = S = 73 wt% R+S W = 27 wt% 31
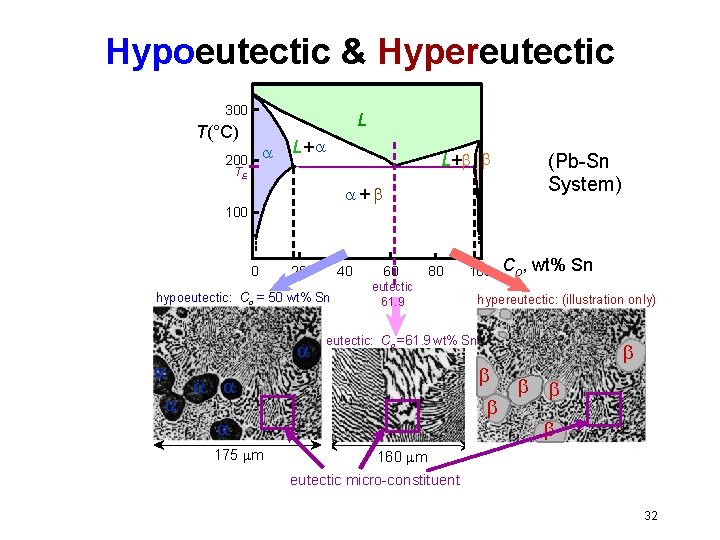
Hypoeutectic & Hypereutectic 300 L T(°C) 200 L+ TE + 100 0 20 40 hypoeutectic: Co = 50 wt% Sn 60 80 eutectic 61. 9 Co, wt% Sn hypereutectic: (illustration only) eutectic: Co = 61. 9 wt% Sn 175 m 100 (Pb-Sn System) 160 m eutectic micro-constituent 32

Eutectoid & Peritectic • Eutectic - liquid in equilibrium with two solids L cool + heat • Eutectoid - solid phase in equation with two solid phases intermetallic compound S 2 S 1+S 3 - cementite cool + Fe 3 C (727ºC) heat • Peritectic - liquid + solid 1 solid 2 • S 1 + L S 2 cool + L heat (1493ºC) 33
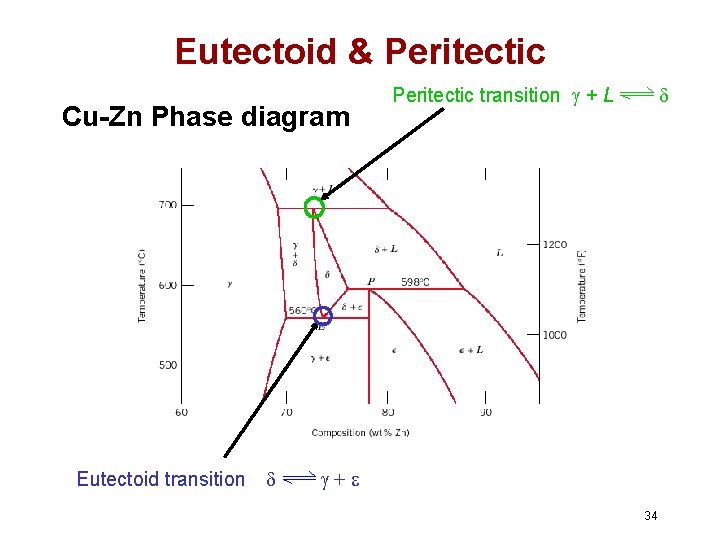
Eutectoid & Peritectic Cu-Zn Phase diagram Eutectoid transition Peritectic transition + L + 34
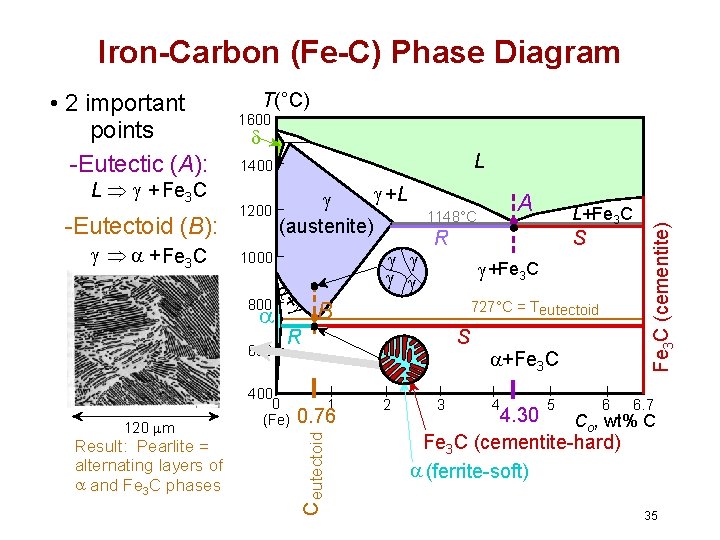
Iron-Carbon (Fe-C) Phase Diagram L Þ + Fe 3 C -Eutectoid (B): Þ + Fe 3 C T(°C) 1600 L 1400 1200 +L (austenite) + 800 600 Result: Pearlite = alternating layers of and Fe 3 C phases S +Fe 3 C 727°C = Teutectoid R 400 0 (Fe) S 1 0. 76 L+Fe 3 C R B C eutectoid 120 m 1000 A 1148°C 2 3 +Fe 3 C 4 5 6 Fe 3 C (cementite) • 2 important points -Eutectic (A): 6. 7 4. 30 Co, wt% C Fe 3 C (cementite-hard) (ferrite-soft) 35
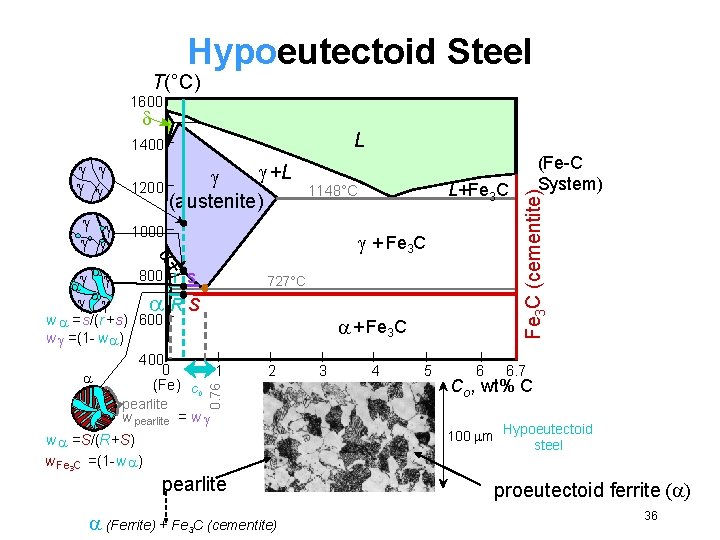
Hypoeutectoid Steel T(°C) 1600 L 800 + Fe 3 C r s 727°C RS w =s/(r +s) 600 w =(1 - w ) 400 0 (Fe) pearlite + Fe 3 C 1 C 0 2 w pearlite = w w =S/(R+S) w Fe 3 C =(1 -w ) L+Fe 3 C 1148°C 1000 + +L 1200 (austenite) 0. 76 pearlite (Ferrite) + Fe 3 C (cementite) 3 4 5 6 (Fe-C System) Fe 3 C (cementite) 1400 6. 7 Co , wt% C 100 m Hypoeutectoid steel proeutectoid ferrite ( ) 36
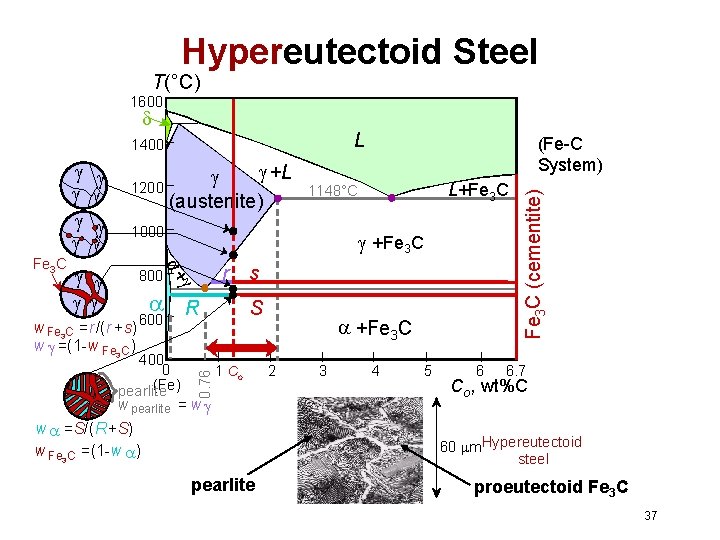
Hypereutectoid Steel T(°C) 1600 L +L 1200 (austenite) 1000 800 w Fe 3 C =r/(r +s) w =(1 -w Fe 3 C ) r R 600 400 0 (Fe) pearlite L+Fe 3 C 1148°C +Fe 3 C + Fe 3 C 0. 76 (Fe-C System) s S 1 Co w pearlite = w w =S/(R+S) w Fe 3 C =(1 -w ) +Fe 3 C 2 3 4 5 6 Fe 3 C (cementite) 1400 6. 7 Co , wt%C 60 m. Hypereutectoid steel pearlite proeutectoid Fe 3 C 37

Case Study For a 99. 6 wt% Fe-0. 40 wt% C at a temperature just below the eutectoid, determine the following a) composition of Fe 3 C and ferrite ( ) b) the amount of carbide (cementite) in grams that forms per 100 g of steel c) the amount of pearlite and proeutectoid ferrite ( ) 38
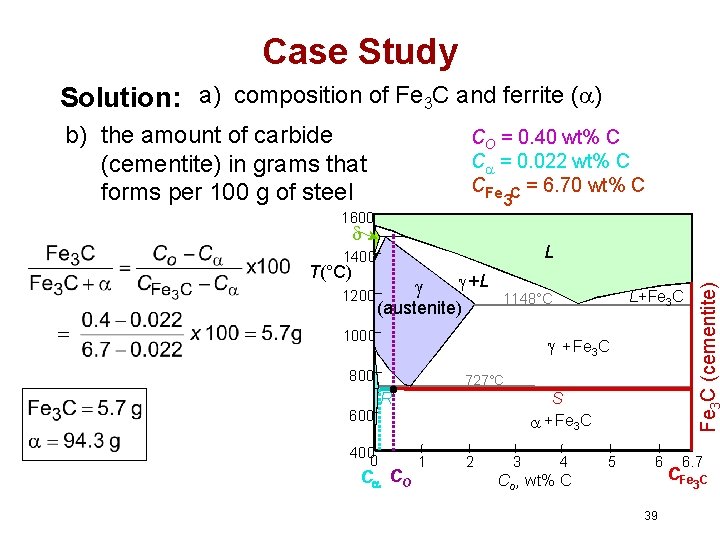
Case Study Solution: a) composition of Fe 3 C and ferrite ( ) b) the amount of carbide (cementite) in grams that forms per 100 g of steel CO = 0. 40 wt% C C = 0. 022 wt% C CFe C = 6. 70 wt% C 3 1600 T(°C) 1200 +L (austenite) + Fe 3 C 800 727°C R S + Fe 3 C 600 400 0 L+Fe 3 C 1148°C 1000 Fe C (cementite) L 1400 C CO 1 2 3 4 5 6 Co , wt% C 6. 7 CFe 3 C 39
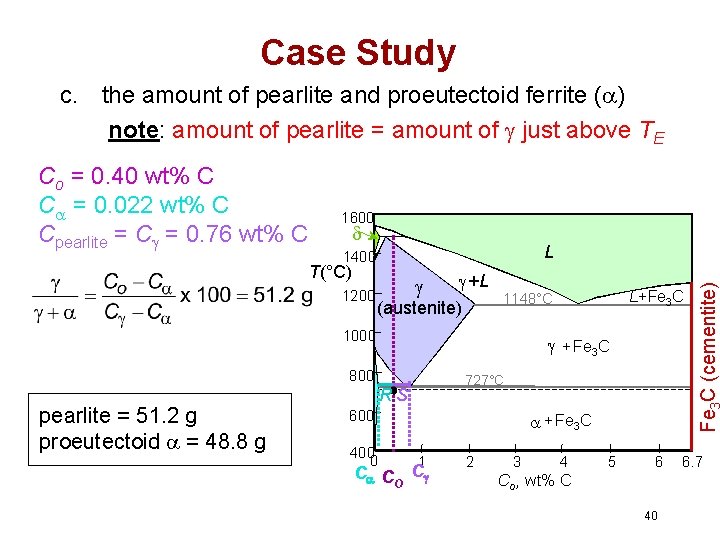
Case Study c. the amount of pearlite and proeutectoid ferrite ( ) note: amount of pearlite = amount of just above TE 1600 L 1400 T(°C) 1200 +L (austenite) 1000 + Fe 3 C 800 pearlite = 51. 2 g proeutectoid = 48. 8 g 727°C RS 600 400 0 L+Fe 3 C 1148°C + Fe 3 C 1 C CO C 2 3 4 5 6 Co , wt% C 40 Fe C (cementite) Co = 0. 40 wt% C C = 0. 022 wt% C Cpearlite = C = 0. 76 wt% C 6. 7
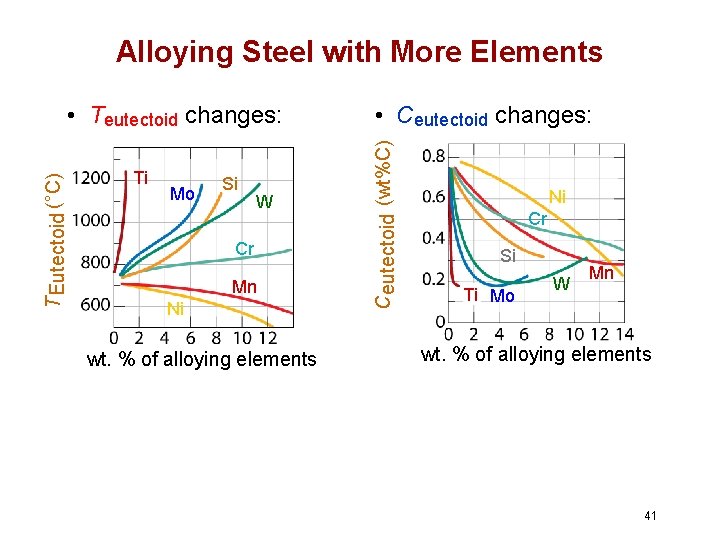
Alloying Steel with More Elements Ti Mo Si W Cr Mn Ni wt. % of alloying elements • Ceutectoid changes: Ceutectoid (wt%C) T Eutectoid (°C) • Teutectoid changes: Ni Cr Si Ti Mo W Mn wt. % of alloying elements 41

Self-Study Task • Applications of Phase Diagrams of Alloys in Mechanical and Manufacturing Engineering 42

Summary • Phase diagrams are useful tools to determine: --the number and types of phases, --the wt% of each phase, --and the composition of each phase for a given T and composition of the system. • Alloying to produce a solid solution usually --increases the tensile strength (TS) --decreases the ductility. • Binary eutectics and binary eutectoids allow for a range of microstructures. 43
- Slides: 43