Phase Changes Solids liquids and gases can be

Phase Changes § Solids, liquids, and gases can be converted from one phase to another by T & P changes. § 2 ways to represent these changes § Heating or cooling curve § Used when P is held constant § Phase diagram § Used when the effects of both P & T are of interest

Phase Changes § Heating and Cooling Curves § Starting with a solid material well below its melting point and adding heat at a constant rate will produce the following effects: § The T of the solid will increase at a constant rate until the solid starts to melt. § When melting begins, the T stops rising & remains constant until all of the solid is converted to liquid. § The T of the liquid starts increasing at a constant rate until boiling starts. § When boiling begins, the T stops rising & remains constant until all of the liquid has been converted to gas. § The T of the gas increases at a constant rate.
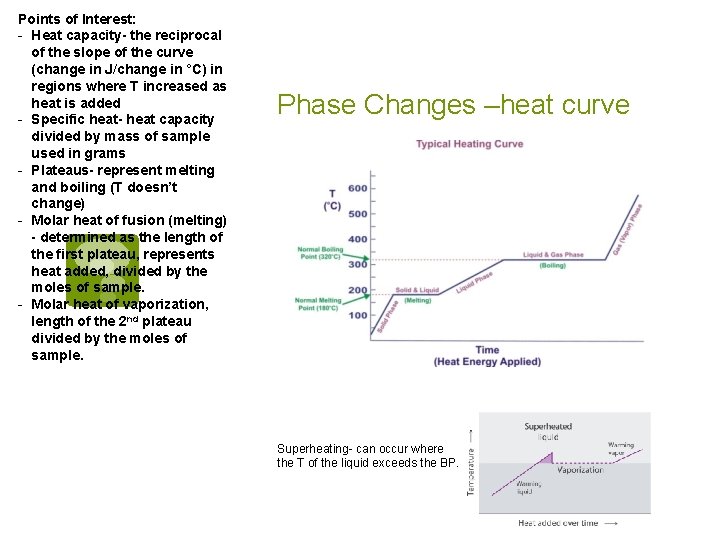
Points of Interest: - Heat capacity- the reciprocal of the slope of the curve (change in J/change in °C) in regions where T increased as heat is added - Specific heat- heat capacity divided by mass of sample used in grams - Plateaus- represent melting and boiling (T doesn’t change) - Molar heat of fusion (melting) - determined as the length of the first plateau, represents heat added, divided by the moles of sample. - Molar heat of vaporization, length of the 2 nd plateau divided by the moles of sample. Phase Changes –heat curve Superheating- can occur where the T of the liquid exceeds the BP.
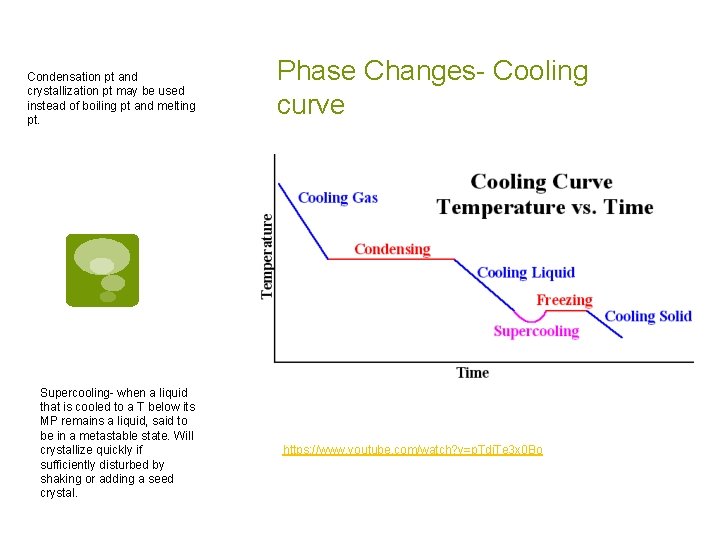
Condensation pt and crystallization pt may be used instead of boiling pt and melting pt. Supercooling- when a liquid that is cooled to a T below its MP remains a liquid, said to be in a metastable state. Will crystallize quickly if sufficiently disturbed by shaking or adding a seed crystal. Phase Changes- Cooling curve https: //www. youtube. com/watch? v=p. Tdi. Te 3 x 0 Bo
- Slides: 4