Phase Changes Review 5 Phases of Matter Solid














- Slides: 18

Phase Changes

Review: 5 Phases of Matter • • • Solid Liquid Gas Plasma Bose-Einstein Condensate

What is a Phase Change? • Is a change from one state of matter (solid, liquid, gas) to another. • Phase changes are physical changes because: - It only affects physical appearance, not chemical make-up. - Reversible

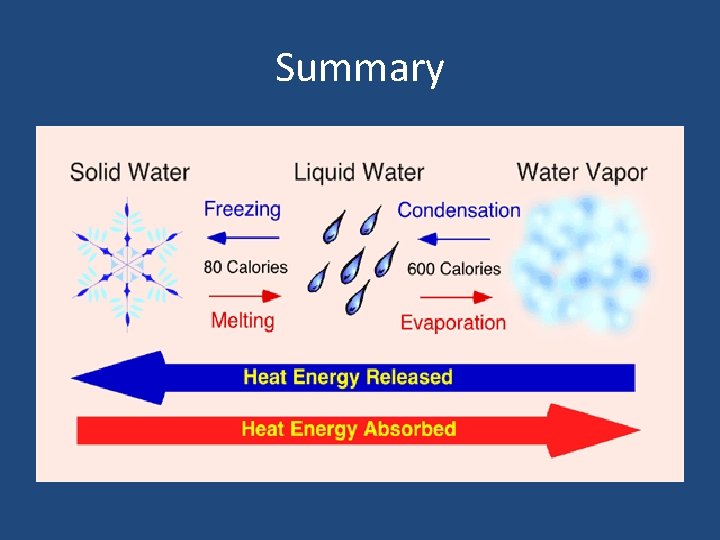
What happens during a phase change? • During a phase change, heat energy is either absorbed or released. • Heat energy is released as molecules slow down and move closer together. (Exothermic) • Heat energy is absorbed as molecules speed up and expand. (Endothermic)

Melting • Phase change from a solid to a liquid • Molecules speed up, move farther apart, and absorb heat energy

Freezing • Phase Change from a liquid to a solid • Molecule slow down, move closer together and release heat energy.

Vaporization (Boiling) • Phase change from a liquid to gas. It occurs at the boiling point of matter. • Molecules speed up, move farther apart, and absorb heat energy.

Evaporation • Phase change from a liquid to a gas on the surface of a liquid (occurs at all temperatures). • Molecules speed up, move farther apart, and absorb heat energy.

Condensation • Phase change from a gas to a liquid. • Molecule slow down, move closer together and release heat energy.

Sublimation • Phase change from a solid to a gas. • Molecules speed up, move farther apart, and absorb heat energy.

Deposition • Phase change from a gas to a solid. • Molecules slow down, move closer together and release heat energy.

Types of Phase Changes • Types of Phase Changes

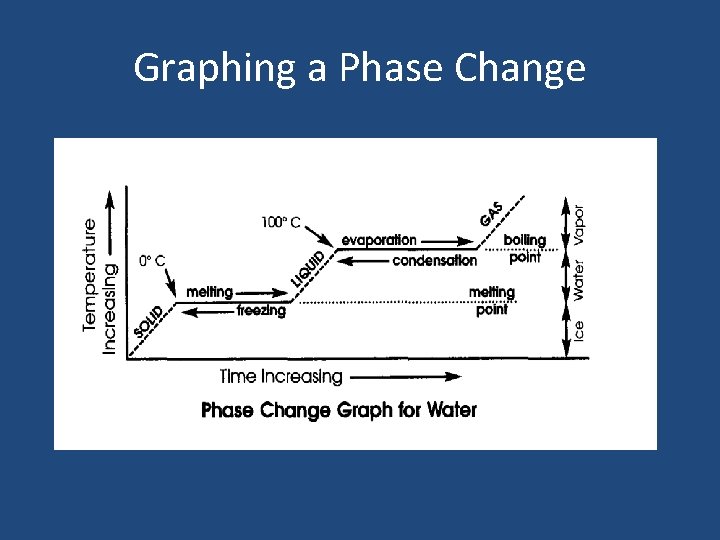
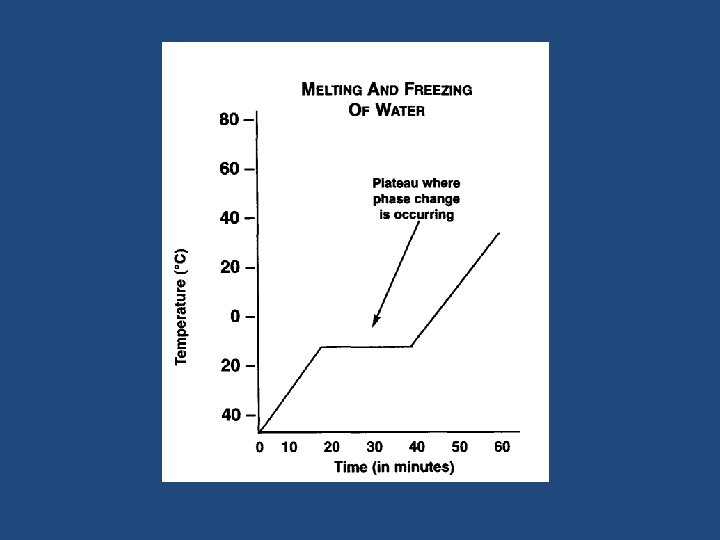
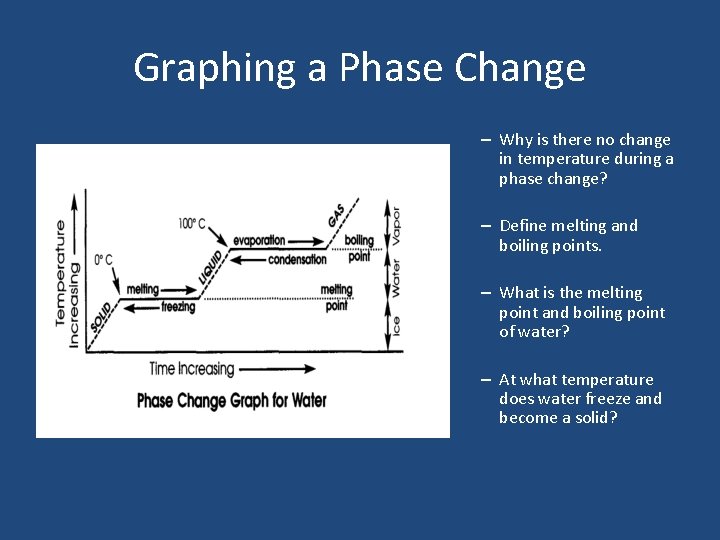
Graphing a Phase Change


Melting & Boiling Points • Melting Point: The temperature at which a solid changes into a liquid. • Boiling Point: The temperature at which a liquid changes into a gas. • What is a Freezing point? Compare the freezing and melting points of water.

Summary

Graphing a Phase Change – Why is there no change in temperature during a phase change? – Define melting and boiling points. – What is the melting point and boiling point of water? – At what temperature does water freeze and become a solid?

Phase Changes • http: //ket. pbslearningmedia. org/resource/64 d 228 a 1 -188 d-4641 -8 c 52 -759 d 5 d 8 ad 489/fastforward-teachable-moment-states-of-matter/ • https: //phet. colorado. edu/en/simulation/stat es-of-matter-basics