Phase Changes Phase Changes Phase changes occur when
















- Slides: 19

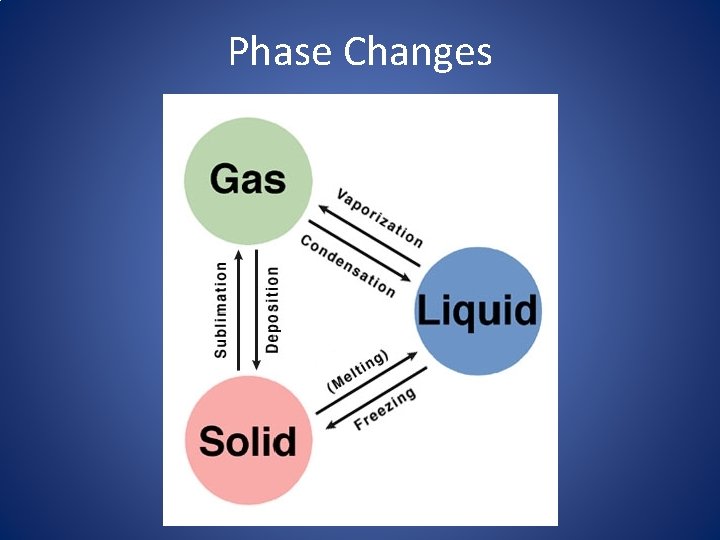
Phase Changes

Phase Changes • Phase changes occur when substances move from one state of matter to another. – Ex: ice cube melting • This is a physical change. – Why is it not considered a chemical change?

Phase Changes • The Kinetic Theory helps explain how and why phase changes take place. – Gaining energy/heat atoms move faster – Losing energy/heat atoms slow down • When a substance condenses from a gas back to liquid, has it absorbed or released heat?

Phase Changes

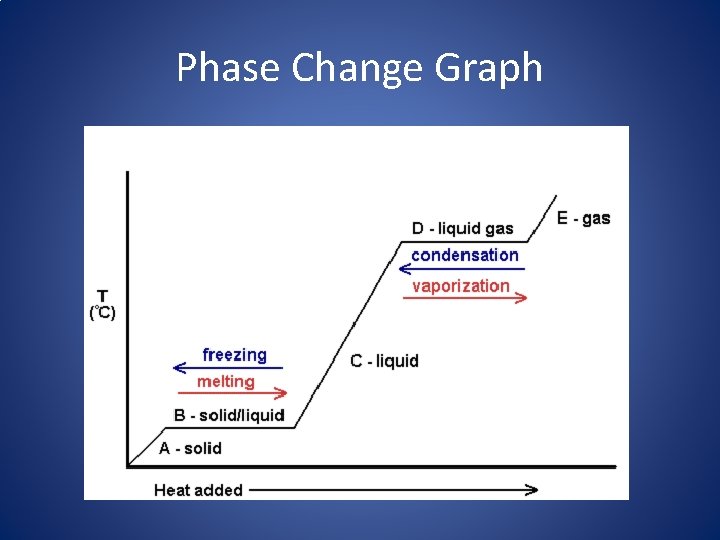
Phase Change Graph

Properties of Matter

Properties of Matter • Ways we describe matter • Two categories: – Physical – Chemical

Physical Properties • Observed with your senses • Can be observed without changing the material • Examples: – Color – Shape – Density

Chemical Properties • Cannot be observed without changing the matter • Describes how the matter reacts with other substances • Examples: – Flammability – Reactivity

Physical vs. Chemical Changes Physical Changes Chemical Changes • A change in size, shape, or state • No new substance is formed • Examples: • A change in physical and chemical properties • A new substance is formed • Examples: – Tearing paper – Crushing a rock – Ice melting – Burning paper – Frying an egg – Rusting

Signs of a Chemical Change • Chemical reactions either absorb or release energy. • Energy can be in many forms: – Heat – Light – Sound – Color – Bubbles

Density

Density • Physical property • Density is the amount of matter (mass) compared to the amount of space (volume) the object takes up.

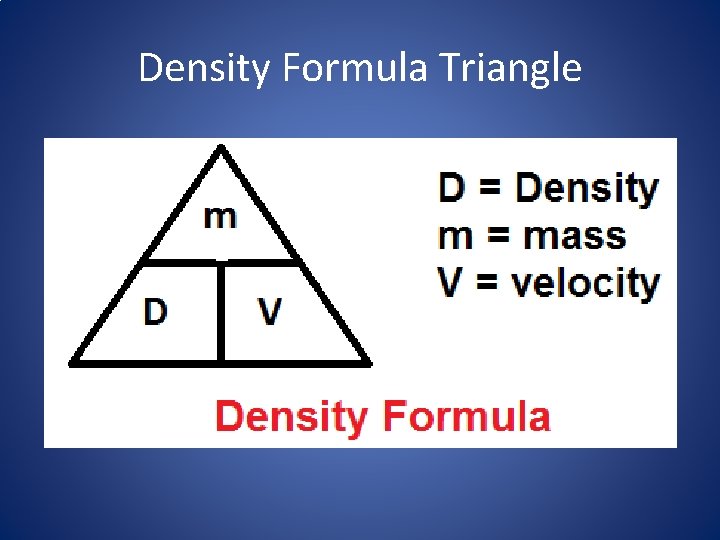
Density • Density is mass divided by volume –D = m/v • Our unit for density is g/ml or g/cm 3

Density Formula Triangle

Water and Density • Since one gram of water has a volume of one m. L, then the density of water will always be 1 g/m. L. • 50 m. L of water will have a mass of 50 grams, so again the density of water will be 1 g/m. L. • What would be the volume of 1000 m. L of water? What would be the density?

Floating and Sinking • Less dense materials will float on top of more dense materials. • Objects with a density of less than 1 g/m. L will float on top of water. • Objects with a density greater than 1 g/m. L will sink in water.

Comparing Densities • Where is the most dense object? • Where is the least dense object?

Density Practice • What is the density of a 16 gram pencil with a volume of 8 cm 3? • What is the volume of a substance that has a mass of 30 grams and a density of 3 g/m. L? • What is the mass of 40 m. L of milk if it has a density of 2 g/m. L?