PHASE CHANGES Phase Change the reversible physical change

PHASE CHANGES

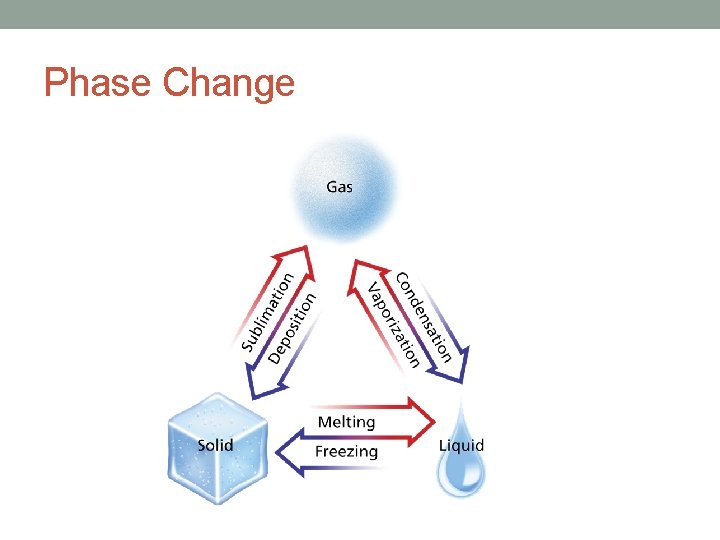
Phase Change • the reversible physical change that occurs when a substance changes from one state of matter to another • Examples: - Melting - Condensation - Freezing - Sublimation - Vaporization - Deposition

Energy and Phase Changes • Energy is transferred between a substance and its surroundings during a phase change • Energy is either absorbed or released • Energy being absorbed is endothermic • Melting • Energy being released is exothermic • Freezing and Burning

Melting • Endothermic • Phase change from solid to a liquid • Kinetic energy of molecules increase • Examples: - Chocolate - Tar - Grease - wax - butter - ice/snow

Freezing • Exothermic • Phase change from liquid to a solid • Kinetic energy of molecules decrease • Examples - Chocolate - Tar - Grease - wax - butter - ice/snow

Vaporization • Substance changes from a liquid into a gas • Endothermic process • Heat of Vaporization- amount of heat needed to change 1 g of a substance from a liquid to a gas • varies from substance to substance • water - 2261 J/g

Evaporation • Change from a liquid to a gas at temperatures below the substance’s boiling point.

Boiling • The temperature at which water boils • Boiling takes place throughout a liquid • Depends on atmospheric pressure • boiling point of water at sea level is 100°C • At higher elevations • the atmospheric pressure is lower • the boiling point is lower than 100°C

Condensation • substance changes from a gas or vapor to a liquid • exothermic process • Examples: • morning dew & “fog” on a shower mirror

Sublimation • phase change from a solid to a gas • endothermic change • Dry ice • solid form of carbon dioxide • does not form a liquid

Deposition • gas or vapor changes directly into a solid • skips liquid phase • exothermic phase change • is the reverse of sublimation • frost is an example
Phase Change
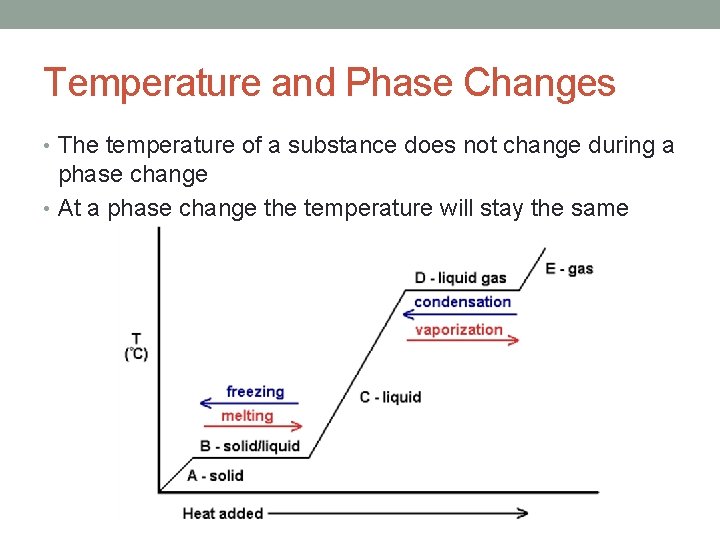
Temperature and Phase Changes • The temperature of a substance does not change during a phase change • At a phase change the temperature will stay the same
- Slides: 13