Phase Changes Courtesy www labinitio com constant Temperature

Phase Changes Courtesy www. lab-initio. com
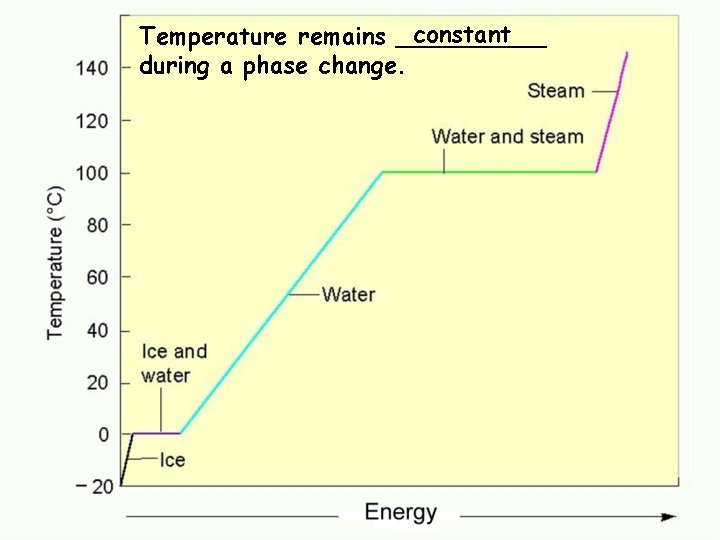
constant Temperature remains _____ during a phase change. Water phase changes

Kinetic Energy, Potential Energy and a Heating Curve. • Since Temperature is a measure of "Average Kinetic Energy", any change in temperature is a change in Kinetic Energy. • Since temperature does not change during a phase change, the energy that is gained or lost is Potential Energy. • Remember the 3 Ps. Plateau, Phase change and Potential Energy Change.
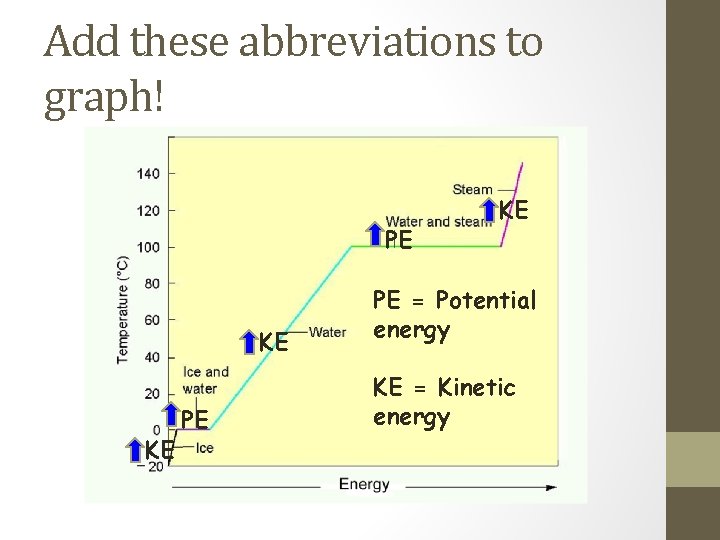
Add these abbreviations to graph! PE KE KE PE = Potential energy KE = Kinetic energy
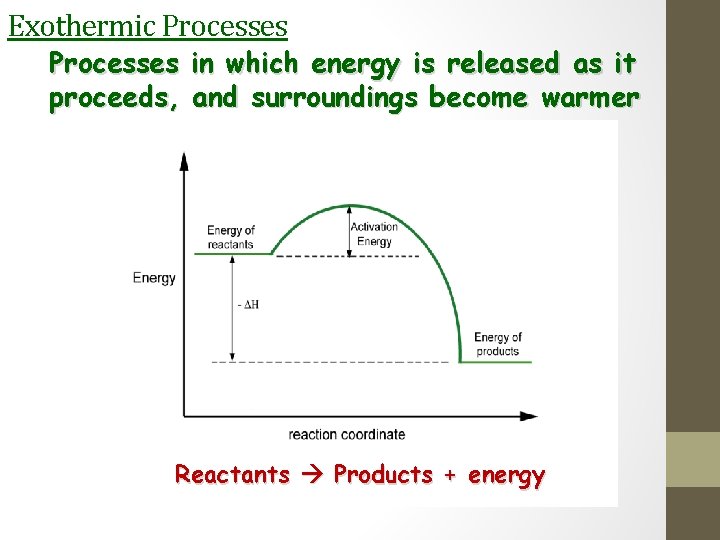
Exothermic Processes in which energy is released as it proceeds, and surroundings become warmer Reactants Products + energy
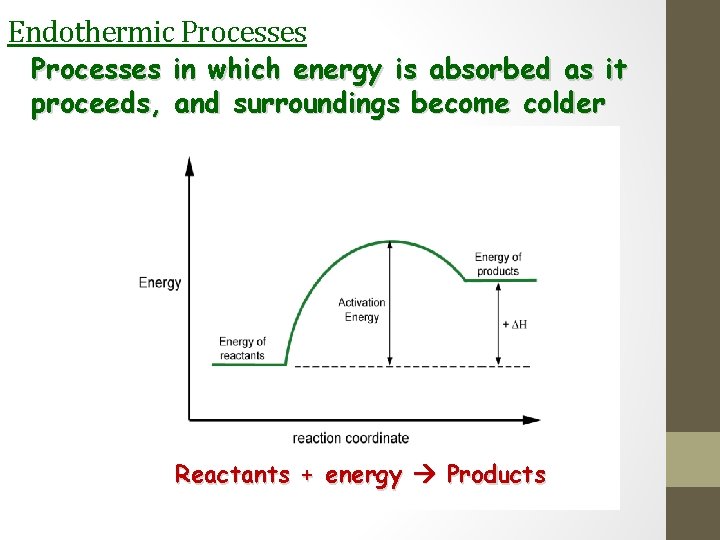
Endothermic Processes in which energy is absorbed as it proceeds, and surroundings become colder Reactants + energy Products

Standard Temperature equals: q 273 Kelvin (273 K) q 0 C

Converting Celsius to Kelvin Many problems involving temperature require that the temperature be in KELVINS! Kelvins = C + 273 °C = Kelvins - 273

Melting point • Melting - change from solid to liquid • Melting point - SPECIFIC temperature when melting occurs. • Each pure substance has a SPECIFIC melting point. • M. P. of Water = 0°C (32°F)

Boiling Point • Boiling Point – temperature at which a liquid boils • Each pure substance has a SPECIFIC boiling point. • Examples: • B. P. of Water = 100°C (212°F)
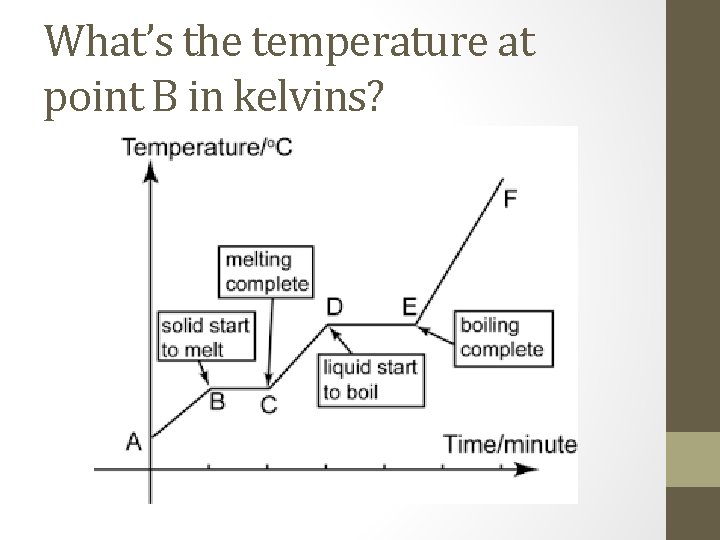
What’s the temperature at point B in kelvins?
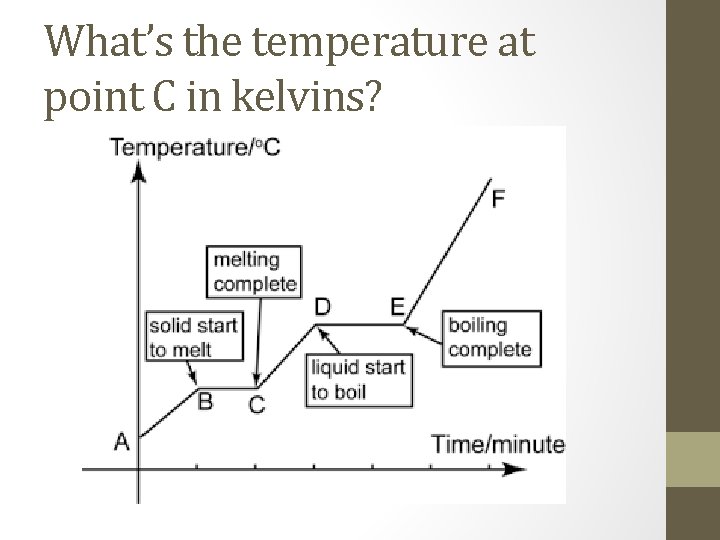
What’s the temperature at point C in kelvins?
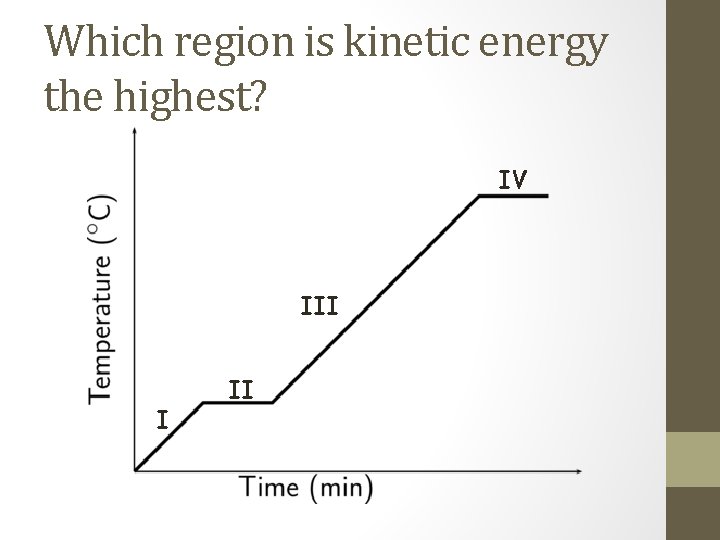
Which region is kinetic energy the highest? IV III I II
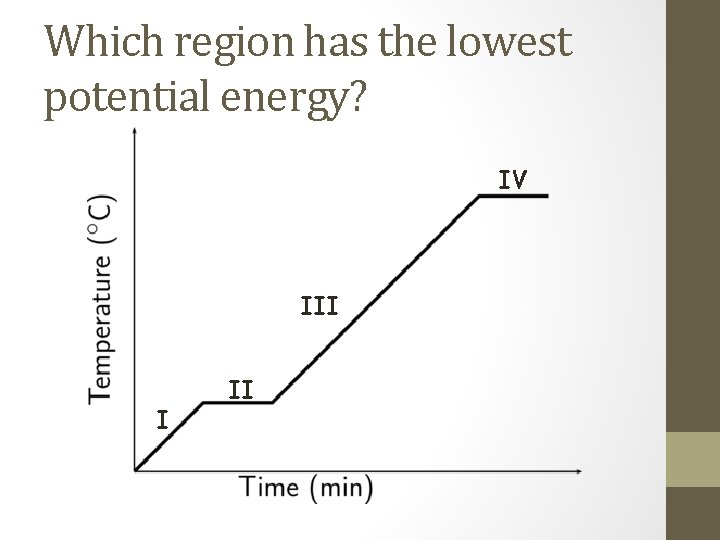
Which region has the lowest potential energy? IV III I II
- Slides: 14