Phase 3 Treatment Nave LedipasvirSofosbuvir Ribavirin for 8

Phase 3 Treatment Naïve Ledipasvir-Sofosbuvir +/- Ribavirin for 8 or 12 weeks in HCV GT 1 ION-3 Source: Kowdley K, et al. N Engl J Med. 2014; 370: 1879 -88. Hepatitis web study

Ledipasvir-Sofosbuvir for 8 or 12 Weeks in Treatment-Naïve HCV GT 1 ION-3 Study: Features ION-3 Trial § Design: Open-label, randomized, phase 3, using fixed-dose combination of ledipasvir-sofosbuvir +/- ribavirin in treatment-naïve patients with GT 1 HCV to explore feasibility of shortening treatment duration from 12 weeks to 8 weeks. § Setting: 58 sites in United States § Entry Criteria - Chronic HCV Genotype 1 (n = 647) - 18 years or older - No prior HCV treatment - Patients with cirrhosis were excluded § Primary End-Point: SVR 12 Source: Kowdley, K, et al. N Engl J Med. 2014; 370: 1879 -88. Hepatitis web study
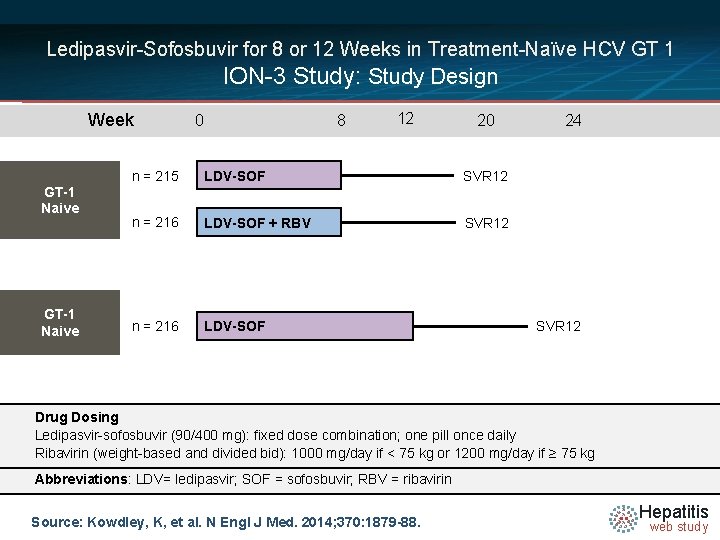
Ledipasvir-Sofosbuvir for 8 or 12 Weeks in Treatment-Naïve HCV GT 1 ION-3 Study: Study Design Week GT-1 Naive 0 8 12 20 n = 215 LDV-SOF SVR 12 n = 216 LDV-SOF + RBV SVR 12 n = 216 LDV-SOF 24 SVR 12 Drug Dosing N =14 Ledipasvir-sofosbuvir (90/400 mg): fixed dose combination; one pill once daily Ribavirin (weight-based and divided bid): 1000 mg/day if < 75 kg or 1200 mg/day if ≥ 75 kg Abbreviations: LDV= ledipasvir; SOF = sofosbuvir; RBV = ribavirin Source: Kowdley, K, et al. N Engl J Med. 2014; 370: 1879 -88. Hepatitis web study
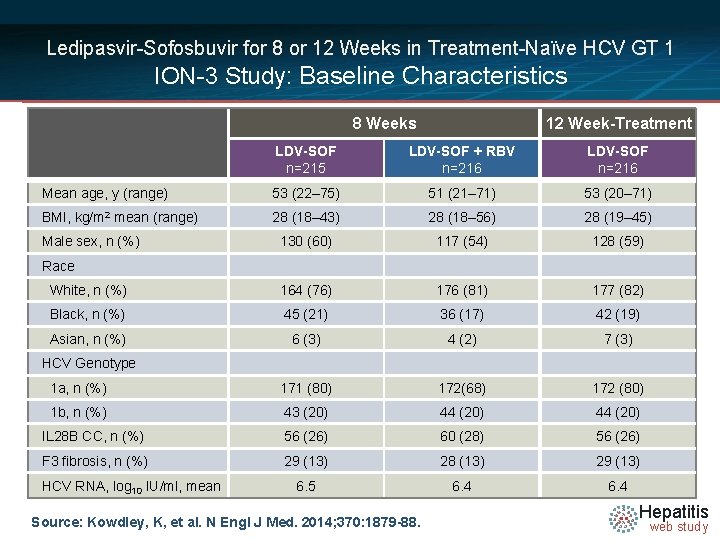
Ledipasvir-Sofosbuvir for 8 or 12 Weeks in Treatment-Naïve HCV GT 1 ION-3 Study: Baseline Characteristics 8 Weeks 12 Week-Treatment LDV-SOF n=215 LDV-SOF + RBV n=216 LDV-SOF n=216 Mean age, y (range) 53 (22– 75) 51 (21– 71) 53 (20– 71) BMI, kg/m 2 mean (range) 28 (18– 43) 28 (18– 56) 28 (19– 45) 130 (60) 117 (54) 128 (59) White, n (%) 164 (76) 176 (81) 177 (82) Black, n (%) 45 (21) 36 (17) 42 (19) Asian, n (%) 6 (3) 4 (2) 7 (3) 1 a, n (%) 171 (80) 172(68) 172 (80) 1 b, n (%) 43 (20) 44 (20) IL 28 B CC, n (%) 56 (26) 60 (28) 56 (26) F 3 fibrosis, n (%) 29 (13) 28 (13) 29 (13) 6. 5 6. 4 Male sex, n (%) Race HCV Genotype HCV RNA, log 10 IU/ml, mean Source: Kowdley, K, et al. N Engl J Med. 2014; 370: 1879 -88. Hepatitis web study
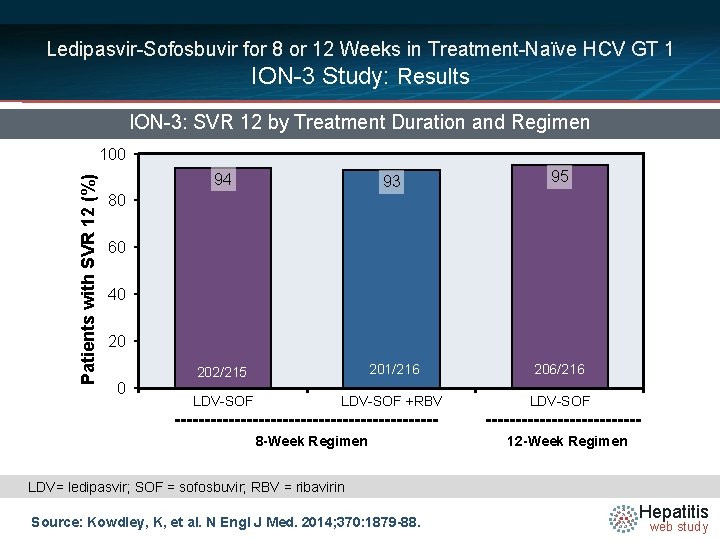
Ledipasvir-Sofosbuvir for 8 or 12 Weeks in Treatment-Naïve HCV GT 1 ION-3 Study: Results ION-3: SVR 12 by Treatment Duration and Regimen Patients with SVR 12 (%) 100 94 93 95 202/215 201/216 206/216 LDV-SOF +RBV LDV-SOF 80 60 40 20 0 8 -Week Regimen 12 -Week Regimen LDV= ledipasvir; SOF = sofosbuvir; RBV = ribavirin Source: Kowdley, K, et al. N Engl J Med. 2014; 370: 1879 -88. Hepatitis web study

Ledipasvir-Sofosbuvir for 8 or 12 Weeks in Treatment-Naïve HCV GT 1 ION-3 Study: Conclusions: “Ledipasvir-sofosbuvir for 8 weeks was associated with a high rate of sustained virologic response among previously untreated patients with HCV genotype 1 infection without cirrhosis. No additional benefit was associated with the inclusion of ribavirin in the regimen or with extension of the duration of treatment to 12 weeks. ” Source: Kowdley, K, et al. N Engl J Med. 2014; 370: 1879 -88. Hepatitis web study

This slide deck is from the University of Washington’s Hepatitis C Online and Hepatitis Web Study projects. Hepatitis C Online www. hepatitisc. uw. edu Hepatitis Web Study http: //depts. washington. edu/hepstudy/ Funded by a grant from the Centers for Disease Control and Prevention. Hepatitis web study
- Slides: 7