Phase 3 Treatment Experienced Boceprevir in Treatment Experienced

Phase 3, Treatment Experienced Boceprevir in Treatment Experienced RESPOND-2 Hepatitis web study

Boceprevir for Retreatment of HCV Genotype 1 Infection RESPOND-2 Trial: Study Design RESPOND-2: Study Features § § § § § N = 403 HCV-monoinfected, treatment-experienced patients Randomized, double-blind, placebo-controlled, phase 3 study All with chronic HCV and genotype 1 Previously responded to treatment but did not obtain SVR Previous null responders excluded HCV RNA > 10, 000 IU/ml Phase III trial Age > 18 Randomized to 3 arms (1: 2: 2) Drug Dosing Boceprevir = 800 mg three times daily Peginterferon alfa-2 b = 1. 5 µg/kg once weekly Ribavirin = 600 -1400 mg/day (based on weight) Source: Bacon BR, et al. N Engl J Med. 2011; 364: 1207 -17. Hepatitis web study
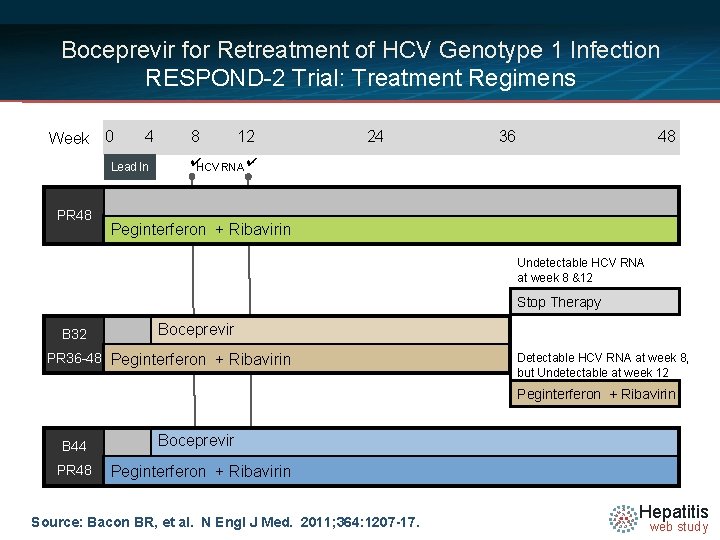
Boceprevir for Retreatment of HCV Genotype 1 Infection RESPOND-2 Trial: Treatment Regimens Week 0 4 Lead In PR 48 8 12 24 36 48 ✔HCV RNA ✔ Peginterferon + Ribavirin Undetectable HCV RNA at week 8 &12 Stop Therapy B 32 Boceprevir PR 36 -48 Peginterferon + Ribavirin Detectable HCV RNA at week 8, but Undetectable at week 12 Peginterferon + Ribavirin B 44 PR 48 Boceprevir Peginterferon + Ribavirin Source: Bacon BR, et al. N Engl J Med. 2011; 364: 1207 -17. Hepatitis web study
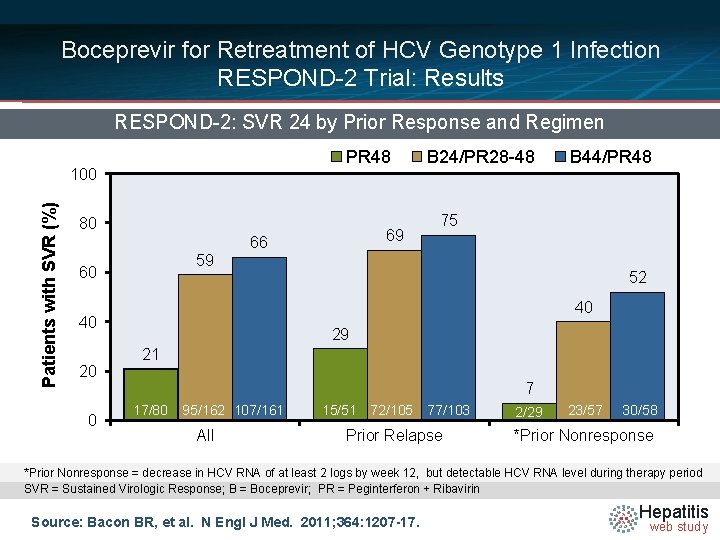
Boceprevir for Retreatment of HCV Genotype 1 Infection RESPOND-2 Trial: Results RESPOND-2: SVR 24 by Prior Response and Regimen PR 48 Patients with SVR (%) 100 80 59 60 0 B 44/PR 48 75 52 40 40 20 69 66 B 24/PR 28 -48 29 21 7 17/80 95/162 107/161 All 15/51 72/105 77/103 Prior Relapse 2/29 23/57 30/58 *Prior Nonresponse = decrease in HCV RNA of at least 2 logs by week 12, but detectable HCV RNA level during therapy period SVR = Sustained Virologic Response; B = Boceprevir; PR = Peginterferon + Ribavirin Source: Bacon BR, et al. N Engl J Med. 2011; 364: 1207 -17. Hepatitis web study
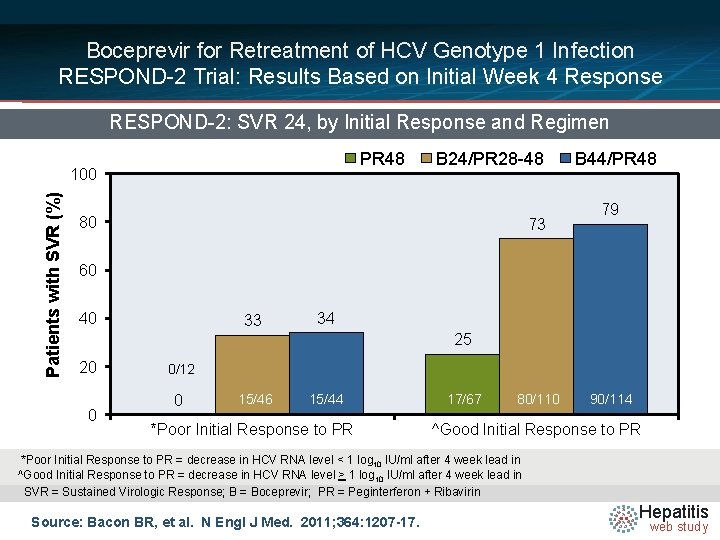
Boceprevir for Retreatment of HCV Genotype 1 Infection RESPOND-2 Trial: Results Based on Initial Week 4 Response RESPOND-2: SVR 24, by Initial Response and Regimen PR 48 Patients with SVR (%) 100 B 24/PR 28 -48 80 73 B 44/PR 48 79 60 40 33 34 25 20 0 0/12 0 15/46 15/44 *Poor Initial Response to PR 17/67 80/110 90/114 ^Good Initial Response to PR *Poor Initial Response to PR = decrease in HCV RNA level < 1 log 10 IU/ml after 4 week lead in ^Good Initial Response to PR = decrease in HCV RNA level > 1 log 10 IU/ml after 4 week lead in SVR = Sustained Virologic Response; B = Boceprevir; PR = Peginterferon + Ribavirin Source: Bacon BR, et al. N Engl J Med. 2011; 364: 1207 -17. Hepatitis web study

Boceprevir for Retreatment of HCV Genotype 1 Infection RESPOND-2 Trial: Conclusions: “The addition of boceprevir to peginterferon–ribavirin resulted in significantly higher rates of sustained virologic response in previously treated patients with chronic HCV genotype 1 infection, as compared with peginterferon–ribavirin alone. ” Source: Bacon BR, et al. N Engl J Med. 2011; 364: 1207 -17. Hepatitis web study
- Slides: 6