Phase 3 Randomized Controlled Clinical Trial of Bictegravir

Phase 3 Randomized, Controlled, Clinical Trial of Bictegravir Coformulated With FTC/TAF in a Fixed-Dose Combination vs Dolutegravir + FTC/TAF in Treatment-Naïve HIV-1–Positive Adults: Week 48 Results Study Design Primary Endpoint 48 Week 0 n=320 Treatment-Naïve Adults § HIV-1 RNA ≥ 500 copies/m. L 96 144 B/F/TAF QD DTG + F/TAF Placebo QD 1: 1 § e. GFRCG ≥ 30 m. L/min DTG + F/TAF QD n=325 B/F/TAF Placebo QD § Phase 3, randomized, double-blind, active-controlled Study 1490 (Clinical. Trials. gov NCT 02607956) – Stratified by HIV-1 RNA, CD 4 cell count, and geographic region (USA vs ex-USA) – North America, Europe, Australia, and Latin America – Chronic hepatitis B/C virus (HBV/HCV) infection allowed § Primary endpoint: proportion with HIV-1 RNA <50 copies/m. L at Week 48 – Noninferiority margin of 12% based on US Food and Drug Administration-defined snapshot algorithm e. GFRCG, estimated glomerular filtration rate by Cockcroft-Gault equation. 1
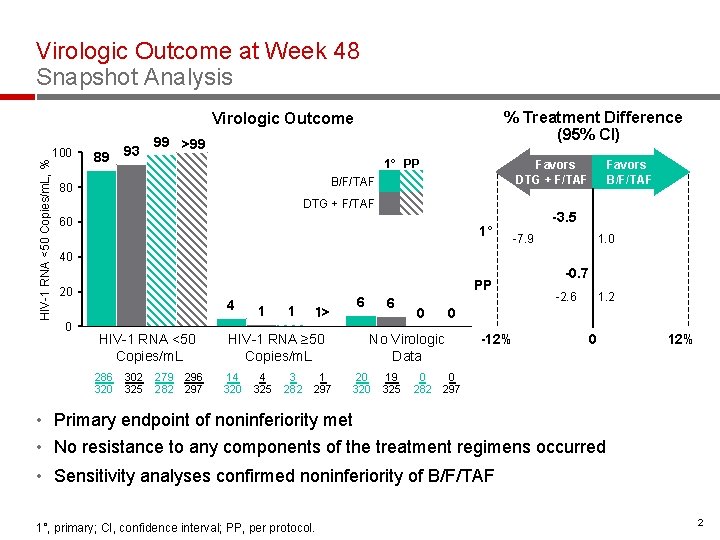
Virologic Outcome at Week 48 Snapshot Analysis % Treatment Difference (95% CI) Virologic Outcome HIV-1 RNA <50 Copies/m. L, % 100 89 93 99 >99 1° PP Favors DTG + F/TAF B/F/TAF 80 DTG + F/TAF 60 1° Favors B/F/TAF -3. 5 -7. 9 1. 0 40 PP 20 0 4 HIV-1 RNA <50 Copies/m. L 286 320 302 325 279 282 296 297 1 1 1> HIV-1 RNA ≥ 50 Copies/m. L 14 320 4 325 3 282 1 297 6 6 0 19 325 -2. 6 1. 2 0 No Virologic Data 20 320 -0. 7 -12% 0 0 282 297 • Primary endpoint of noninferiority met • No resistance to any components of the treatment regimens occurred • Sensitivity analyses confirmed noninferiority of B/F/TAF 1°, primary; CI, confidence interval; PP, per protocol. 2
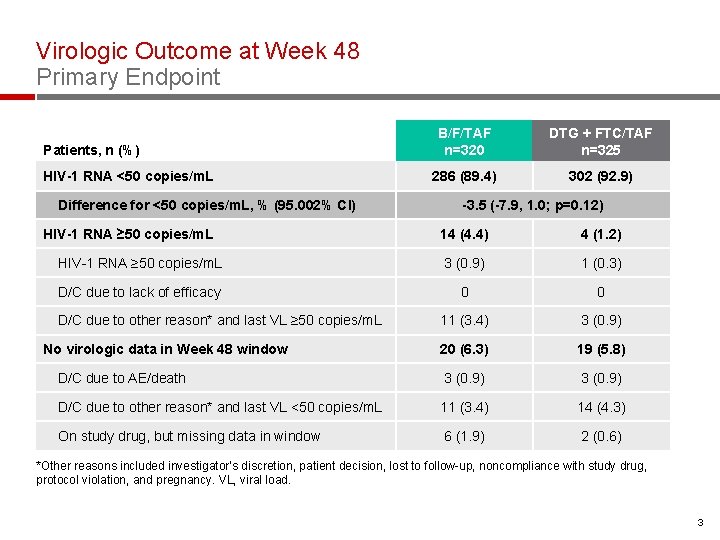
Virologic Outcome at Week 48 Primary Endpoint Patients, n (%) HIV-1 RNA <50 copies/m. L Difference for <50 copies/m. L, % (95. 002% CI) HIV-1 RNA ≥ 50 copies/m. L B/F/TAF n=320 DTG + FTC/TAF n=325 286 (89. 4) 302 (92. 9) -3. 5 (-7. 9, 1. 0; p=0. 12) 14 (4. 4) 4 (1. 2) HIV-1 RNA ≥ 50 copies/m. L 3 (0. 9) 1 (0. 3) D/C due to lack of efficacy 0 0 11 (3. 4) 3 (0. 9) 20 (6. 3) 19 (5. 8) D/C due to AE/death 3 (0. 9) D/C due to other reason* and last VL <50 copies/m. L 11 (3. 4) 14 (4. 3) On study drug, but missing data in window 6 (1. 9) 2 (0. 6) D/C due to other reason* and last VL ≥ 50 copies/m. L No virologic data in Week 48 window *Other reasons included investigator’s discretion, patient decision, lost to follow-up, noncompliance with study drug, protocol violation, and pregnancy. VL, viral load. 3
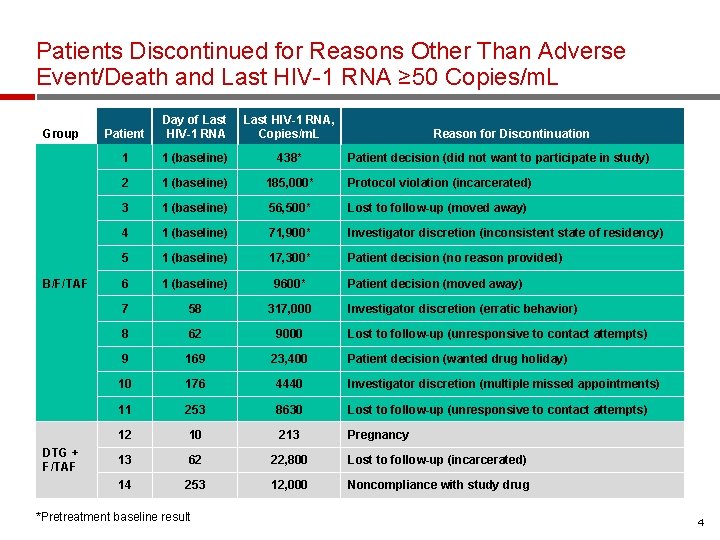
Patients Discontinued for Reasons Other Than Adverse Event/Death and Last HIV-1 RNA ≥ 50 Copies/m. L Group B/F/TAF DTG + F/TAF Patient Day of Last HIV-1 RNA, Copies/m. L 1 1 (baseline) 438* 2 1 (baseline) 185, 000* Protocol violation (incarcerated) 3 1 (baseline) 56, 500* Lost to follow-up (moved away) 4 1 (baseline) 71, 900* Investigator discretion (inconsistent state of residency) 5 1 (baseline) 17, 300* Patient decision (no reason provided) 6 1 (baseline) 9600* 7 58 317, 000 8 62 9000 9 169 23, 400 10 176 4440 Investigator discretion (multiple missed appointments) 11 253 8630 Lost to follow-up (unresponsive to contact attempts) 12 10 213 Pregnancy 13 62 22, 800 Lost to follow-up (incarcerated) 14 253 12, 000 Noncompliance with study drug *Pretreatment baseline result Reason for Discontinuation Patient decision (did not want to participate in study) Patient decision (moved away) Investigator discretion (erratic behavior) Lost to follow-up (unresponsive to contact attempts) Patient decision (wanted drug holiday) 4

Further Results and Conclusions § B/F/TAF was safe and well tolerated – Treatment discontinuations due to adverse events were rare in both arms § Lipid changes did not differ significantly between study arms § Less decrease in e. GFRCG was observed with B/F/TAF vs DTG + F/TAF – No treatment discontinuations due to renal adverse events § Further details of results in poster TUPDB 0201 LB Conclusions: § Virologic suppression at Week 48 was high in both arms, and B/F/TAF was noninferior to DTG + F/TAF in treatment-naïve adults – No patient discontinued due to lack of efficacy § No treatment-emergent resistance to any study medication was observed § These data reinforce the non-inferiority of B/F/TAF vs. DTG-based regimens seen in Study 1489 (abstract MOAB 0105 LB), presented yesterday 5
- Slides: 5