Phase 2 Treatment Nave HIV Coinfection Sofosbuvir Peginterferon

Phase 2 Treatment Naïve HIV Coinfection Sofosbuvir + Peginterferon+ Ribavirin in HCV-HIV GT 1 -4 Rodriguez-Torres M, et al. J Acquir Immune Defic Syndr. 2015; 68: 543 -9. Hepatitis web study

Sofosbuvir + PEG + RBV for HCV-HIV Coinfection Study Features Sofosbuvir + PEG + RBV for HCV GT 1 -6 and HIV Coinfection: Features § Design: Open-label, single-arm, phase 2 trial of sofosbuvir plus peginterferon alfa-2 a plus ribavirin in HCV GT 1 -6 with HIV coinfection § Setting: single site in Peurto Rico § Entry Criteria - Chronic HIV coinfection; HCV genotype 1 -6 - Age ≥ 21 - HCV treatment naïve - On antiretroviral therapy for at least 8 weeks - CD 4 count greater than 200 cells/mm 3 - No cirrhosis § Patient Characteristics - N = 23 HCV-HIV coinfected patients - GT 1 (n=19); GT 2 (n=1); GT 3 (n=2); GT 4 (n=1) § Primary End-Points - Efficacy (SVR 12), safety/tolerability, and impact on HIV Source: Rodriguez-Torres M, et al. J Acquir Immune Defic Syndr. 2015; 68: 543 -9. Hepatitis web study
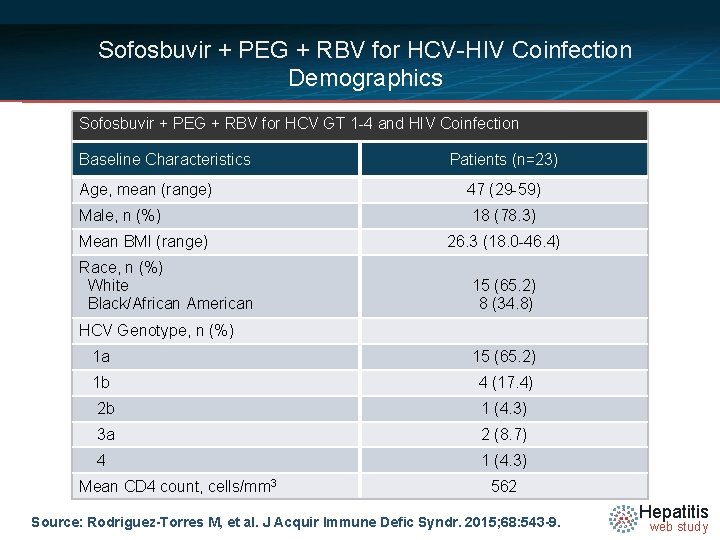
Sofosbuvir + PEG + RBV for HCV-HIV Coinfection Demographics Sofosbuvir + PEG + RBV for HCV GT 1 -4 and HIV Coinfection Baseline Characteristics Age, mean (range) Male, n (%) Mean BMI (range) Race, n (%) White Black/African American Patients (n=23) 47 (29 -59) 18 (78. 3) 26. 3 (18. 0 -46. 4) 15 (65. 2) 8 (34. 8) HCV Genotype, n (%) 1 a 15 (65. 2) 1 b 4 (17. 4) 2 b 1 (4. 3) 3 a 2 (8. 7) 4 1 (4. 3) Mean CD 4 count, cells/mm 3 562 Source: Rodriguez-Torres M, et al. J Acquir Immune Defic Syndr. 2015; 68: 543 -9. Hepatitis web study
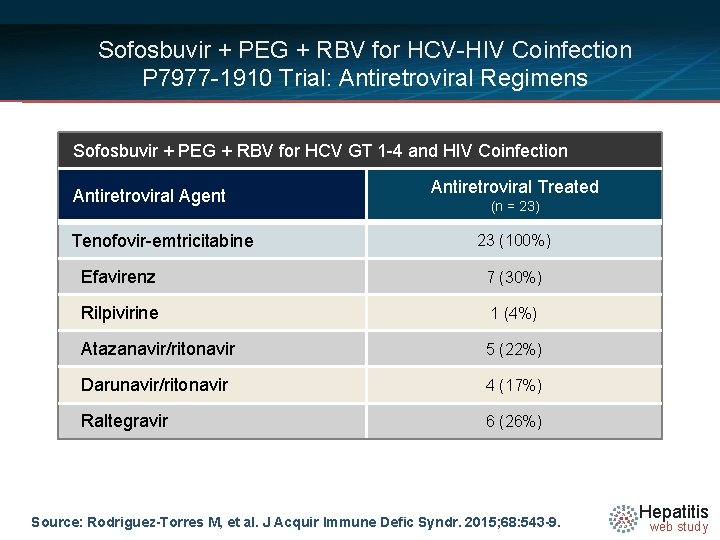
Sofosbuvir + PEG + RBV for HCV-HIV Coinfection P 7977 -1910 Trial: Antiretroviral Regimens Sofosbuvir + PEG + RBV for HCV GT 1 -4 and HIV Coinfection Antiretroviral Agent Tenofovir-emtricitabine Antiretroviral Treated (n = 23) 23 (100%) Efavirenz 7 (30%) Rilpivirine 1 (4%) Atazanavir/ritonavir 5 (22%) Darunavir/ritonavir 4 (17%) Raltegravir 6 (26%) Source: Rodriguez-Torres M, et al. J Acquir Immune Defic Syndr. 2015; 68: 543 -9. Hepatitis web study
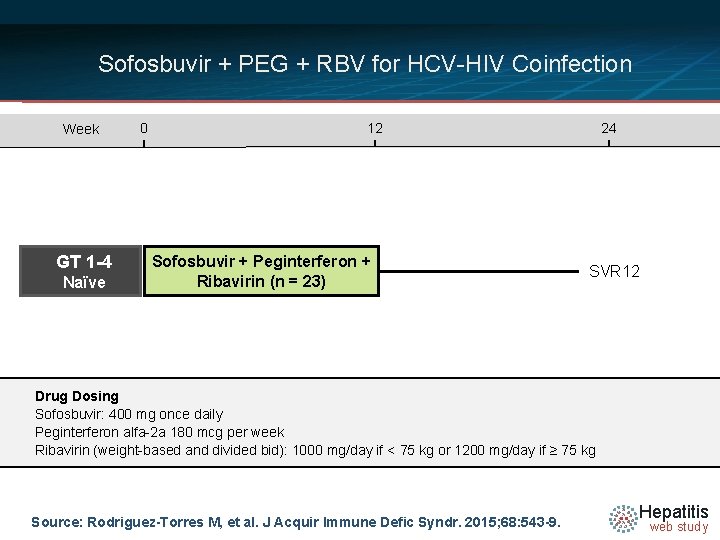
Sofosbuvir + PEG + RBV for HCV-HIV Coinfection Week GT 1 -4 Naïve 0 12 Sofosbuvir + Peginterferon + Ribavirin (n = 23) 24 SVR 12 Drug Dosing Sofosbuvir: 400 mg once daily Peginterferon alfa-2 a 180 mcg per week Ribavirin (weight-based and divided bid): 1000 mg/day if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Rodriguez-Torres M, et al. J Acquir Immune Defic Syndr. 2015; 68: 543 -9. Hepatitis web study
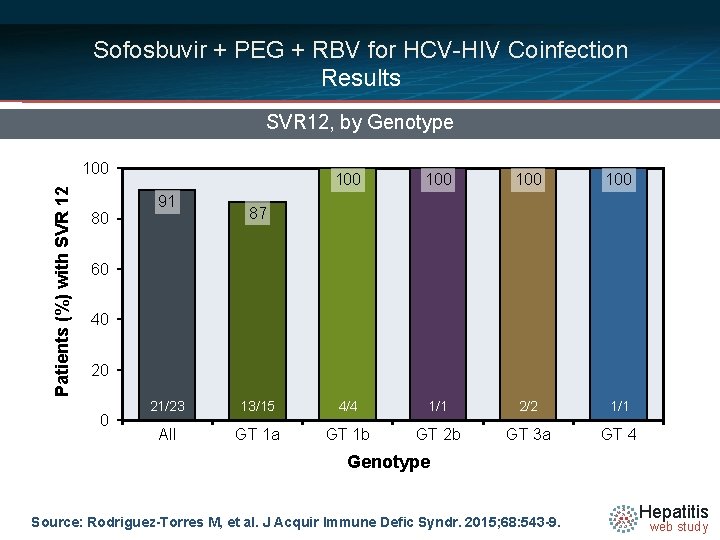
Sofosbuvir + PEG + RBV for HCV-HIV Coinfection Results SVR 12, by Genotype Patients (%) with SVR 12 100 80 91 100 100 4/4 1/1 2/2 1/1 87 60 40 20 0 21/23 95/112 All 13/15 GT 1 a 22/25 GT 1 b 94/106 GT 2 b GT 3 a 26/31 GT 4 Genotype Source: Rodriguez-Torres M, et al. J Acquir Immune Defic Syndr. 2015; 68: 543 -9. Hepatitis web study
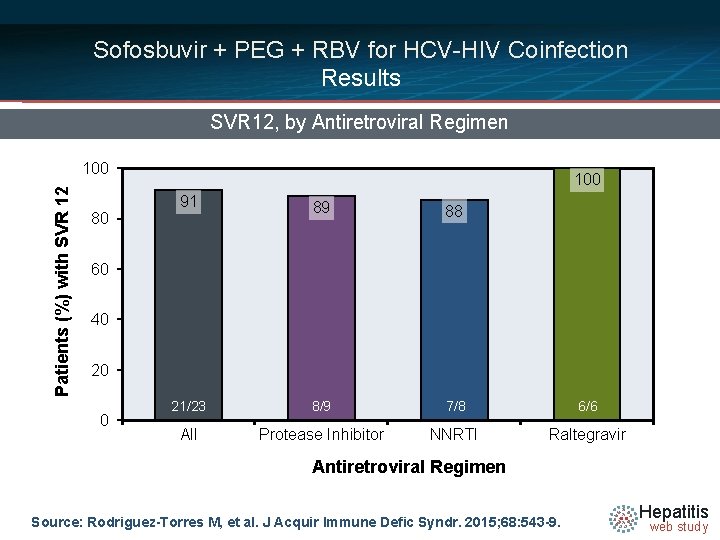
Sofosbuvir + PEG + RBV for HCV-HIV Coinfection Results SVR 12, by Antiretroviral Regimen Patients (%) with SVR 12 100 80 100 91 89 88 21/23 8/9 7/8 6/6 22/25 Protease Inhibitor 94/106 NNRTI 26/31 Raltegravir 60 40 20 0 95/112 All Antiretroviral Regimen Source: Rodriguez-Torres M, et al. J Acquir Immune Defic Syndr. 2015; 68: 543 -9. Hepatitis web study

Sofosbuvir + PEG + RBV for HCV-HIV Coinfection Results: Interpretation: “Sofosbuvir may be coadministered safely with many commonly used antiretrovirals. The addition of sofosbuvir to peginterferon–ribavirin was highly effective as assessed by SVR in HCV/HIV-coinfected patients. ” Source: Rodriguez-Torres M, et al. J Acquir Immune Defic Syndr. 2015; 68: 543 -9. Hepatitis web study

This slide deck is from the University of Washington’s Hepatitis C Online and Hepatitis Web Study projects. Hepatitis C Online www. hepatitisc. uw. edu Hepatitis Web Study http: //depts. washington. edu/hepstudy/ Funded by a grant from the Centers for Disease Control and Prevention. Hepatitis web study
- Slides: 9