Phase 2 b Treatment Experienced Telaprevir in Treatment

Phase 2 b Treatment Experienced Telaprevir in Treatment Experienced GT-1 PROVE 3 Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -303. Hepatitis web study

Telaprevir for Treatment-Experienced HCV Genotype 1 PROVE 3 Study: Study Design PROVE 3: Study Features § § § § Randomized, partially double-blind trial, placebo-controlled Phase 2 b trial All with HCV and lack of SVR with Peginterferon + Ribavirin Eligible if 18 to 70 years of age All with Genotype 1; 92% with HCV RNA > 800, 000 IU/ml N = 465 enrolled and 453 received at least 1 dose Setting: 53 international sites (41 in US) Randomized to one of 4 arms Drug Dosing Telaprevir = 1125 mg loading dose, then 750 mg every 8 hours Peginterferon alfa-2 a = 180 µg weekly Ribavirin = 1000 mg/d for wt < 75 kg; 1200 mg/d for wt > 75 kg Source: Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -303. Hepatitis web study
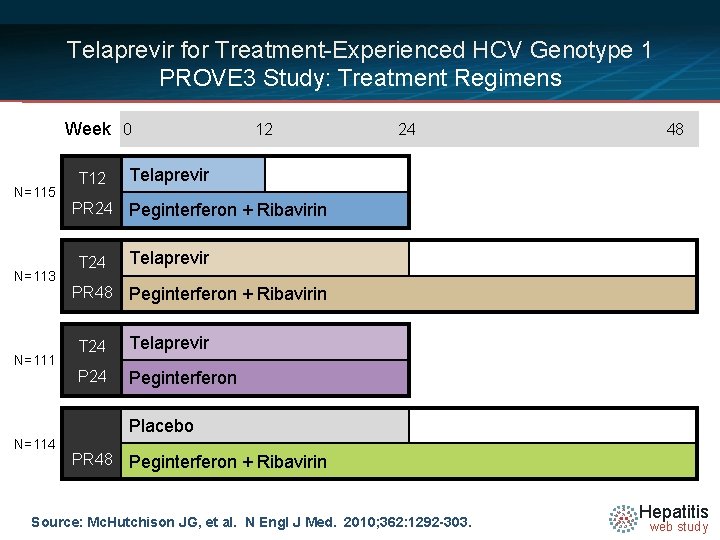
Telaprevir for Treatment-Experienced HCV Genotype 1 PROVE 3 Study: Treatment Regimens Week 0 N=115 N=113 N=111 T 12 PR 24 T 24 PR 48 12 24 48 Telaprevir Peginterferon + Ribavirin T 24 Telaprevir P 24 Peginterferon Placebo N=114 PR 48 Peginterferon + Ribavirin Source: Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -303. Hepatitis web study
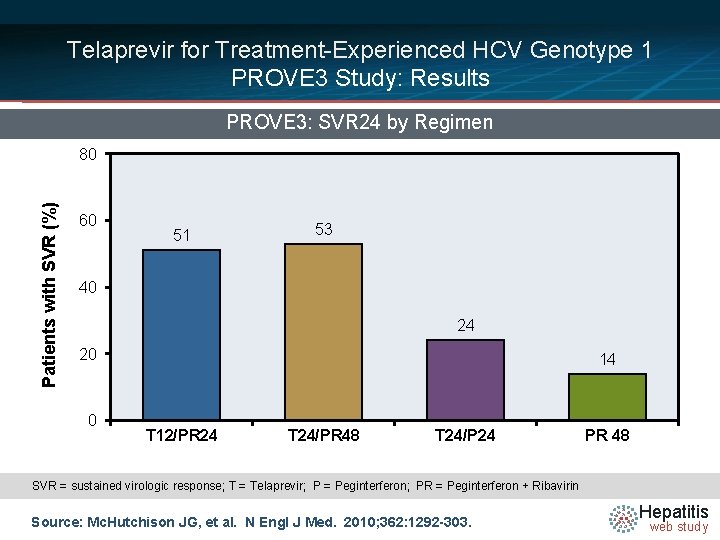
Telaprevir for Treatment-Experienced HCV Genotype 1 PROVE 3 Study: Results PROVE 3: SVR 24 by Regimen Patients with SVR (%) 80 60 51 53 40 24 20 0 14 T 12/PR 24 T 24/PR 48 T 24/P 24 PR 48 SVR = sustained virologic response; T = Telaprevir; P = Peginterferon; PR = Peginterferon + Ribavirin Source: Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -303. Hepatitis web study
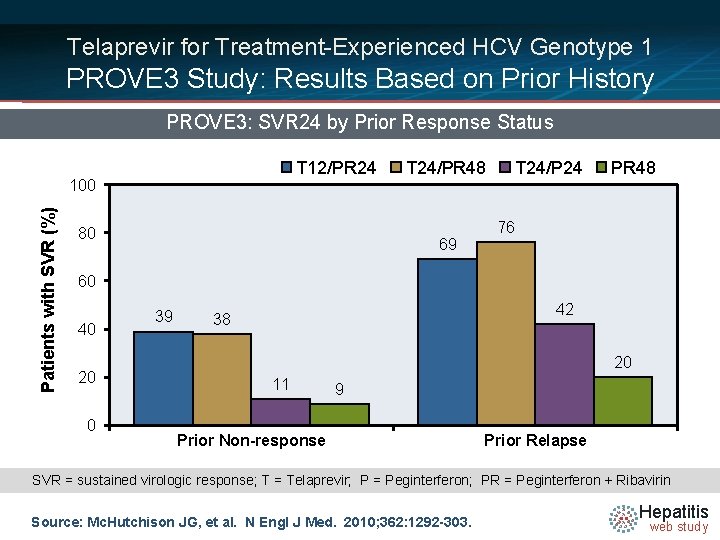
Telaprevir for Treatment-Experienced HCV Genotype 1 PROVE 3 Study: Results Based on Prior History PROVE 3: SVR 24 by Prior Response Status T 12/PR 24 Patients with SVR (%) 100 80 T 24/PR 48 69 T 24/P 24 PR 48 76 60 40 20 0 39 42 38 20 11 9 Prior Non-response Prior Relapse SVR = sustained virologic response; T = Telaprevir; P = Peginterferon; PR = Peginterferon + Ribavirin Source: Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -303. Hepatitis web study
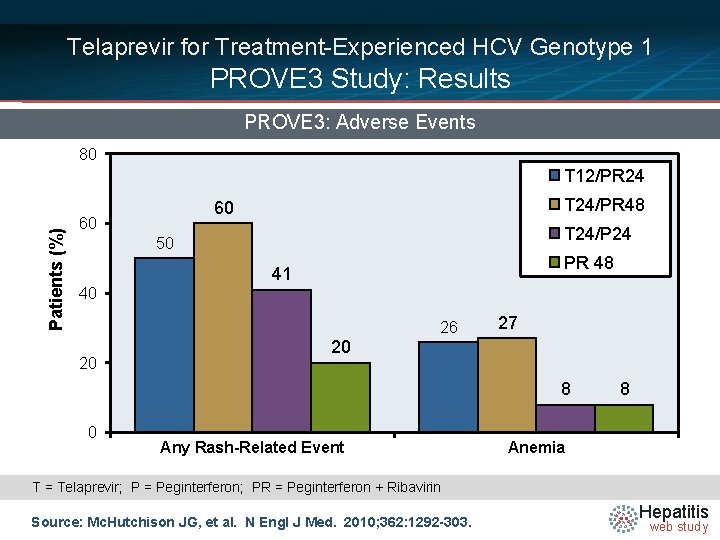
Telaprevir for Treatment-Experienced HCV Genotype 1 PROVE 3 Study: Results PROVE 3: Adverse Events 80 Patients (%) T 12/PR 24 T 24/PR 48 60 60 T 24/P 24 50 PR 48 41 40 26 20 27 20 8 0 Any Rash-Related Event 8 Anemia T = Telaprevir; P = Peginterferon; PR = Peginterferon + Ribavirin Source: Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -303. Hepatitis web study

Telaprevir for Treatment-Experienced HCV Genotype 1 PROVE 3 Study: Conclusions: “In HCV-infected patients in whom initial peginterferon alfa and ribavirin treatment failed, retreatment with telaprevir in combination with peginterferon alfa-2 a and ribavirin was more effective than retreatment with peginterferon alfa-2 a and ribavirin alone. ” Source: Mc. Hutchison JG, et al. N Engl J Med. 2010; 362: 1292 -303. Hepatitis web study
- Slides: 7