Pharmacogenomics Beatriz Prez Gonzlez 201718 Genomics Content 1

Pharmacogenomics Beatriz Pérez González 2017/18 Genomics

Content 1. Introduction 2. Clinical applications 3. Pharmacogenomics and cancer 4. The role in cancer treatment 5. Challenges and conclusions
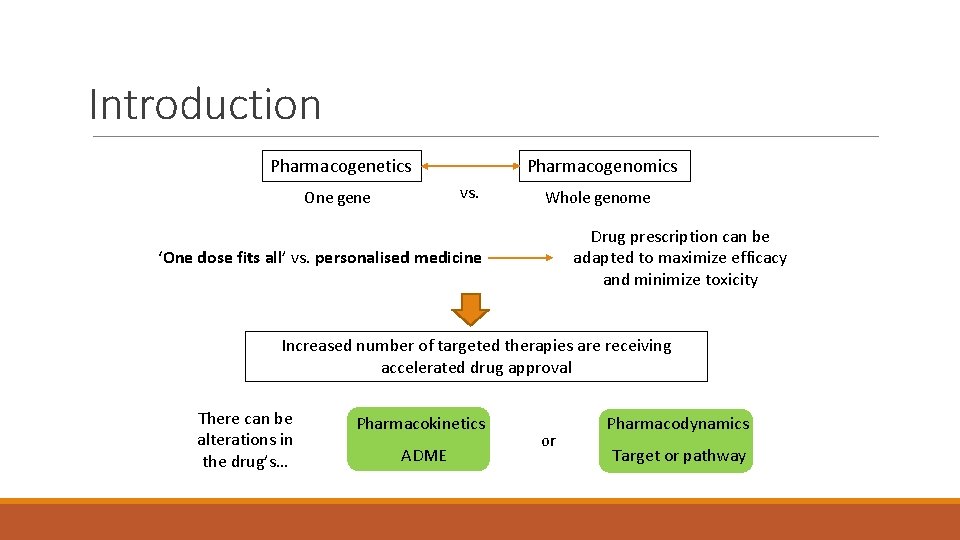
Introduction Pharmacogenomics Pharmacogenetics vs. One gene Whole genome Drug prescription can be adapted to maximize efficacy and minimize toxicity ‘One dose fits all’ vs. personalised medicine Increased number of targeted therapies are receiving accelerated drug approval There can be alterations in the drug’s… Pharmacokinetics ADME or Pharmacodynamics Target or pathway

Clinical applications Abacavir hypersensitivity HLA-B*5701 allele Inhibitor of reverse transcriptase Treatment HIV Exclude carriers of this allele 5 -8% cases of severe hypersensitivity Disappearance of the hypersensitivity syndrome caused by abacavir

Clinical applications CYP 2 D 6 Metabolizes nearly 25% of used drugs and it has a large inter-individual variability Most important pharmacogenetic biomarker Different variants affect more than 41 drugs’ response CYP 2 D 6 genotype has robust effects on metabolizer phenotype Codeine poor intermediate extensive ultra-rapid tricyclic antidepressants
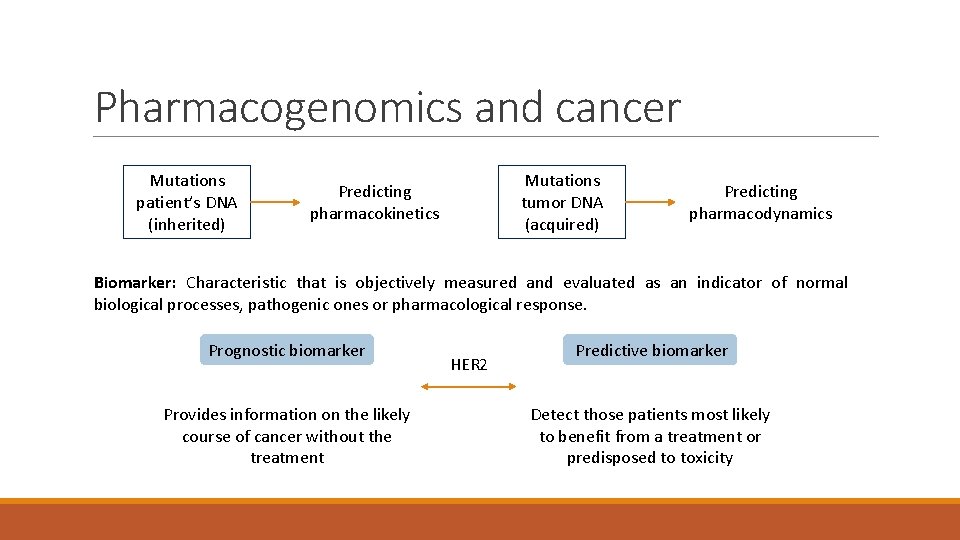
Pharmacogenomics and cancer Mutations patient’s DNA (inherited) Mutations tumor DNA (acquired) Predicting pharmacokinetics Predicting pharmacodynamics Biomarker: Characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic ones or pharmacological response. Prognostic biomarker Provides information on the likely course of cancer without the treatment HER 2 Predictive biomarker Detect those patients most likely to benefit from a treatment or predisposed to toxicity
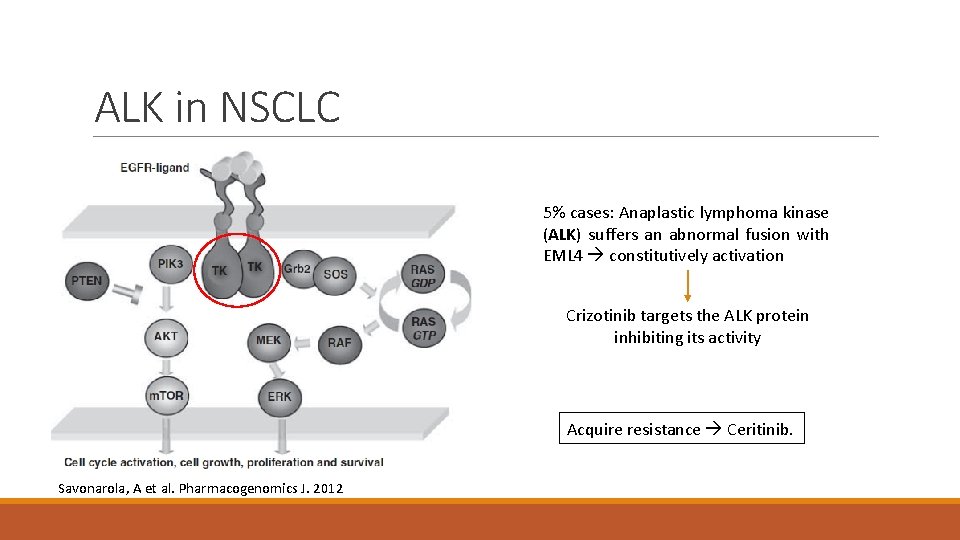
ALK in NSCLC 5% cases: Anaplastic lymphoma kinase (ALK) suffers an abnormal fusion with EML 4 constitutively activation Crizotinib targets the ALK protein inhibiting its activity Acquire resistance Ceritinib. Savonarola, A et al. Pharmacogenomics J. 2012
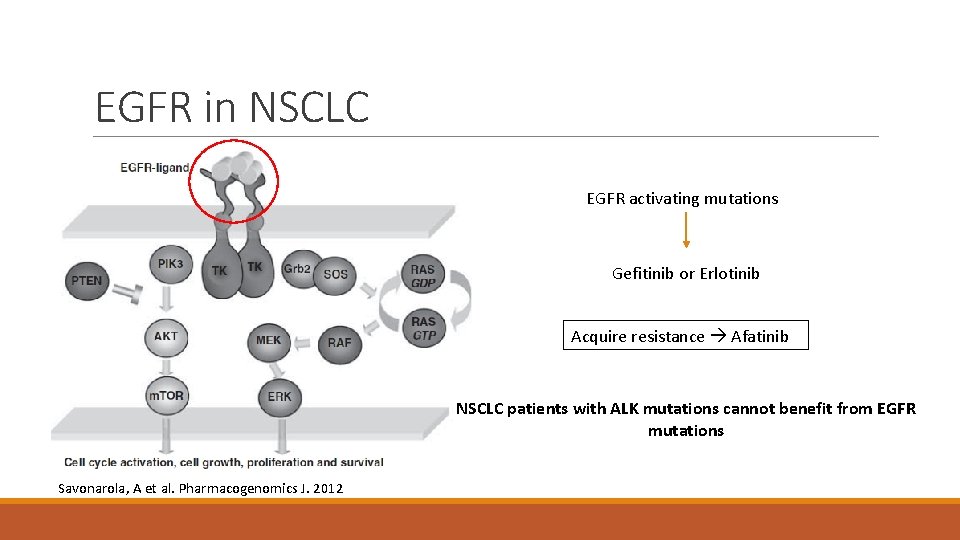
EGFR in NSCLC EGFR activating mutations Gefitinib or Erlotinib Acquire resistance Afatinib NSCLC patients with ALK mutations cannot benefit from EGFR mutations Savonarola, A et al. Pharmacogenomics J. 2012
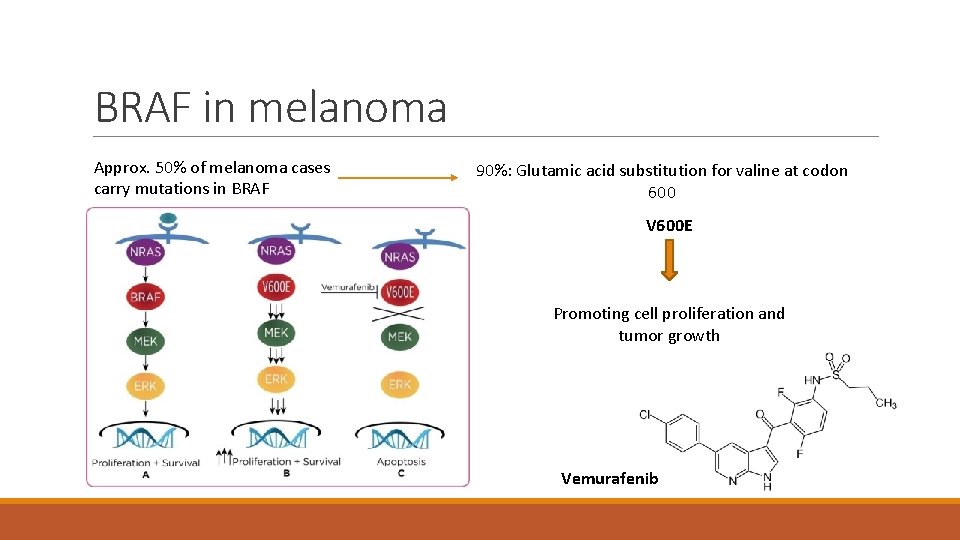
BRAF in melanoma Approx. 50% of melanoma cases carry mutations in BRAF 90%: Glutamic acid substitution for valine at codon 600 V 600 E Promoting cell proliferation and tumor growth Vemurafenib

UGT 1 A 1 and Irinotecan is a topoisomerase I inhibitor, prodrug used for metastatic colorectal cancer. Irinotecan Inactivated by uridine diphosphate glucuronosyl transferase 1 A 1 (UGT 1 A 1) Metabolism SN-38 (active) Inactivation UGT 1 A 1*28 allele related to reduced expression and activity SN-38 G Severe toxicity It is also an example of the potential under-dosing of the homozygous or heterozygous patients for the WT allele

Conclusions • Increased targeted therapies mean greater reliance on biomarkers to help decide the most beneficial treatment for a patient. • Sometimes the number of patients in studies to identify genetic markers is not enough. • GWAS is a promising method to identify novel common genetic variations in pharmacogenomics. • Cost-effectiveness analysis are difficult to determine. “With the continuing decline in cost of sequencing, in a not-too-distant future, every individual will have their entire inherited genome sequenced early in life for clinical purposes” Relling, M. V et al. Nature. 2015

References • Lu, D. -Y. , Lu, T. -R. , Xu, B. & Ding, J. Pharmacogenetics of cancer therapy: breakthroughs from beyond? Futur. Sci. OA 1, fso. 15. 80 (2015). • Pirmohamed, M. Pharmacogenetics and pharmacogenomics. Br. J. Clin. Pharmacol. 52, 345– 347 (2001). • Patel, J. N. Cancer pharmacogenomics, challenges in implementation, and patient-focused perspectives. Pharmgenomics. Pers. Med. 9, 65– 77 (2016). • Savonarola, A. , Palmirotta, R. , Guadagni, F. & Silvestris, F. Pharmacogenetics and pharmacogenomics: Role of mutational analysis in anti-cancer targeted therapy. Pharmacogenomics J. 12, 277– 286 (2012). • Relling, M. V and Evans, W. E. Pharmacogenomics in the clinic. Nature 526, 343– 350 (2015). • Low, S. K. , Takahashi, A. , Mushiroda, T. & Kubo, M. Genome-Wide Association Study: A Useful Tool to Identify Common Genetic Variants Associated with Drug Toxicity and Efficacy in Cancer Pharmacogenomics. Clin. Cancer Res. 20, 2541– 2552 (2014). • Dickmann, L. J. & Ware, J. A. Pharmacogenomics in the age of personalized medicine. Drug Discov. Today Technol. 21 – 22, 11– 16 (2016). • Becquemont, L. HLA: a pharmacogenomics success story. Pharmacogenomics 11, 277– 81 (2010). • Wang, D. , Papp, A. C. & Sun, X. Functional characterization of CYP 2 D 6 enhancer polymorphisms. Hum. Mol. Genet. 24, 1556– 1562 (2015).

Thank you for your attention!
- Slides: 13