Pharmaceutical Regulatory Affairs BPH 425 Chapter 1 Introduction

Pharmaceutical Regulatory Affairs BPH 425 Chapter 1: Introduction to RA


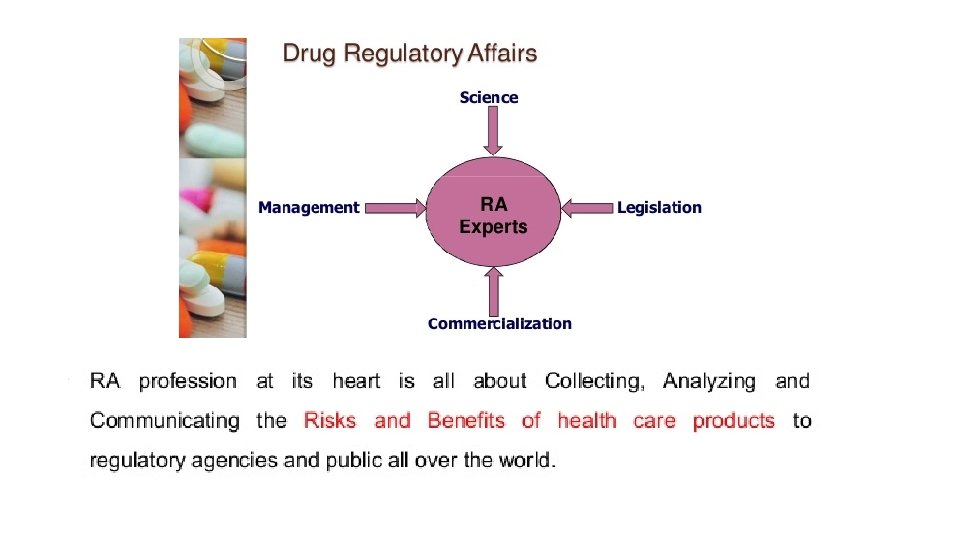
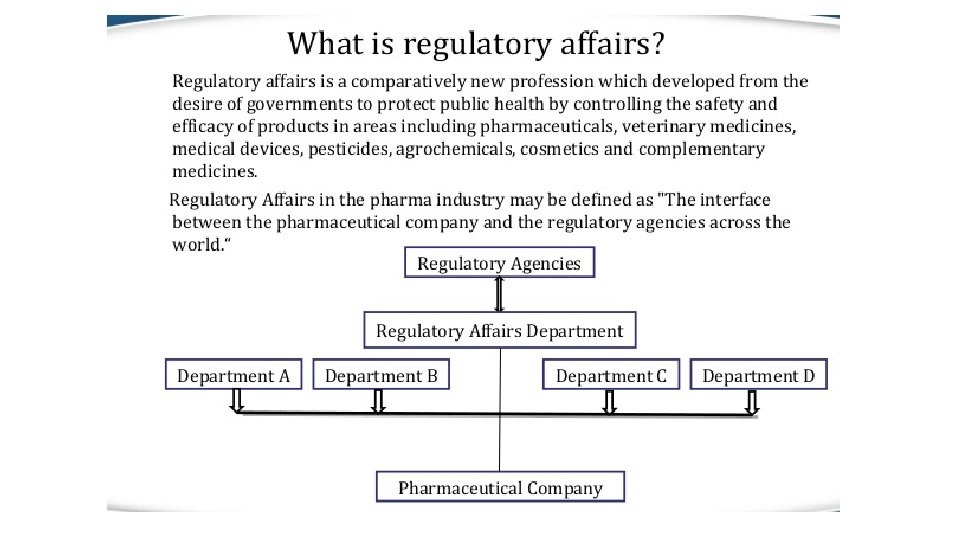
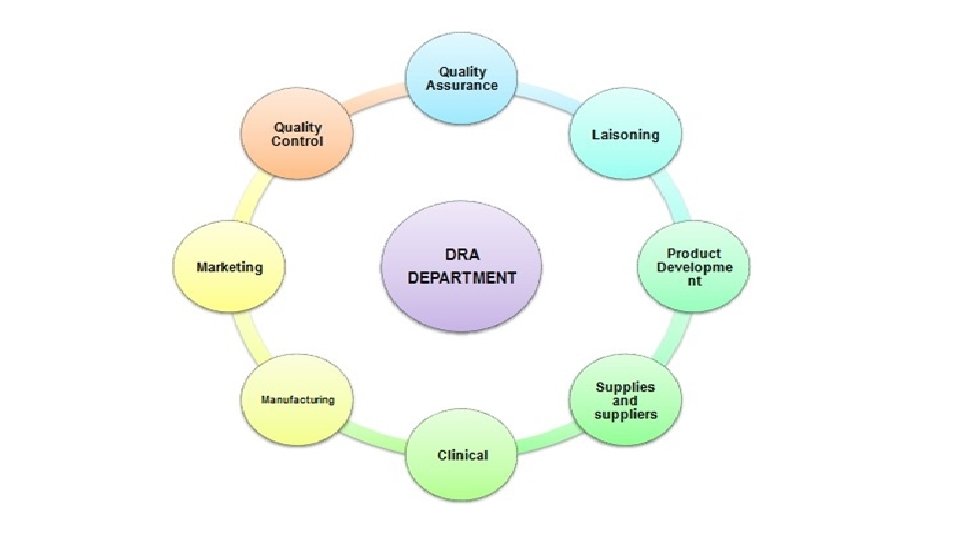
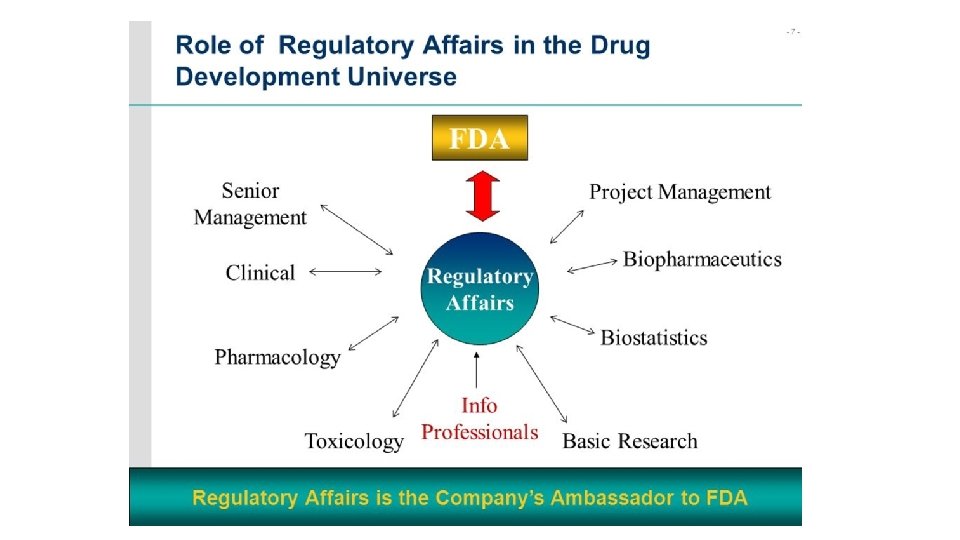
Regulatory affairs is a profession developed from the intention of governments to protect public health by controlling the safety and efficacy of products including pharmaceuticals, veterinary medicines, medical devices, cosmetics so that supplied products make a worthwhile contribution to public health and welfare. The drug development process is a lengthy, complex and extremely costly albeit necessary process. Regulatory Affairs plays a crucial role in the pharmaceutical industry and is involved in all stages of drug development and also after drug approval and marketing. Regulatory Affairs is a unique combination of science and management to achieve a commercially important goal within a drug-development organization. It gives strategic and technical advice at the highest level in their companies, right from the beginning of the development of a product, making an important contribution both commercially and scientifically to the success of a development program and the company as a whole.


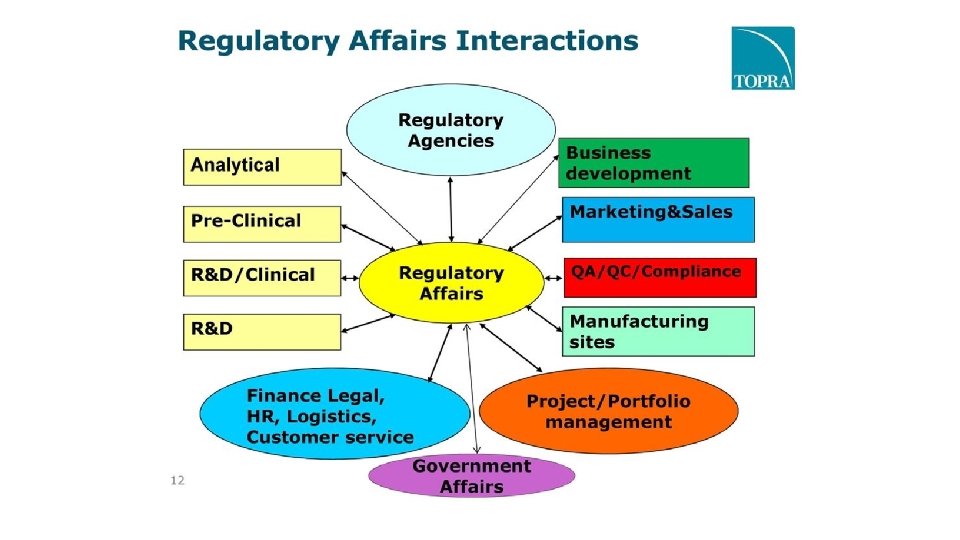

SCOPE OF REGULATORY AFFAIRS Regulatory affairs professionals are employed in industry, government regulatory authorities and academics. The wide range of regulatory professionals includes in these areas: • Pharmaceuticals • Medical devices • In vitro diagnostics • Biologics and biotechnology • Nutritional Products • Cosmetics • Veterinary Products


Ø 1. Keeping track of the ever-changing legislation in all the regions in which a company wishes to distribute its products. Ø 2. Advising on legal and scientific restraints and requirements. Ø 3. Collecting, collating and evaluating scientific data. Ø 4. Presenting registration documents to regulatory agencies and carrying out any subsequent negotiations necessary to obtain or maintain marketing authorization for the products concerned.

Ø 5. Giving strategic and technical advice at the highest level in their companies, making an important contribution both commercially and scientifically to the success of a development programme and the company as a whole. Ø 6. Helping the company avoid problems caused by badly kept records, inappropriate scientific thinking or poor presentation of data. Ø 7. Preparation of organized and Ensure adherence and compliance with all the applicable CGMP, ICH, GCP, GLP guidelines regulations and laws. Ø 8. A regulatory affair plays a crucial role in the industry and is involved in all stages of drug development and also after drug approval and marketing.


Job description of an RA executive: v Assisting in Responding Queries for various regulatory authorities. v Analyzing and arranging dossier in CTD/e. CTD formats for regulatory submissions. v Up to date maintenance of Documentation Data base available with RA. v Coordination with cross functional departments like QA, QC, Production, Warehouse, Distribution and Marketing for dossier preparation, Post approval supplement filling and respond to queries. v Routine Interaction with QA-documentation for updating Master Files. v Review of Artworks (Labels and Package Insert). v Good knowledge of Module 3. v Knowledge of ICH Guidelines. v Review and approval of QC spec, STP, method validation inline with pharmacopoeia.

v Review and approval of R&D/ Products document in line with pharmacopoeia and regulatory requirements v Coordinate with cross functional departments such as Operations, QA, QC, Engineering etc, and support to regulatory Preparation and review of Master safety Data sheet, TSE & BSE documents v Regulatory Compliance, for change control activities monitoring the impact of changes in regulatory submissions. v Assess and review documentation to ensure compliance with product licences. v Monitoring the progress of registration submissions, responding to queries and ensuring that registration approvals are granted without undue delays.


- Slides: 19