Pharmaceutical Organic Chemistry By Dr Mehnaz Kamal Assistant

Pharmaceutical Organic Chemistry By Dr. Mehnaz Kamal Assistant Professor Pharmaceutical Chemistry Prince Sattam Bin Abdulaziz University
Heterocyclic Chemistry

1 -To know types of six membered heterocyclic ring 2 - To know sources of six membered heterocyclic compounds 3 - To know syntheses of six membered heterocyclic compounds 4 -To understand chemical reactions of six membered heterocyclic compounds
Six Membered Aromatic Heterocycles
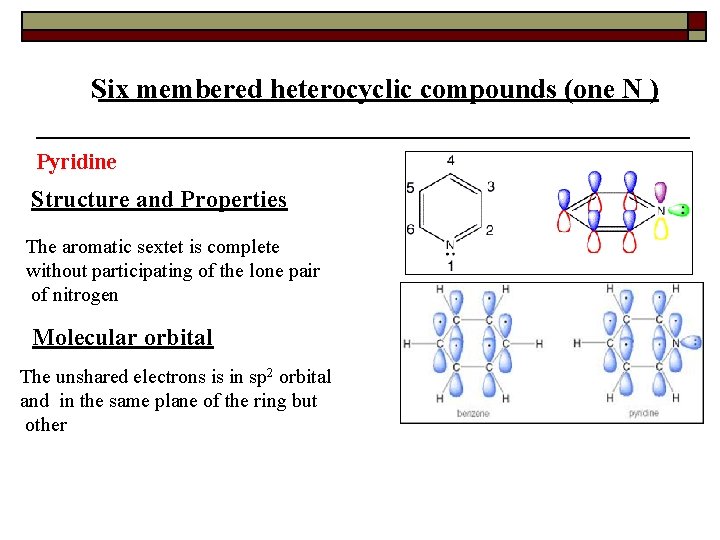
Six membered heterocyclic compounds (one N ) Pyridine Structure and Properties The aromatic sextet is complete without participating of the lone pair of nitrogen Molecular orbital The unshared electrons is in sp 2 orbital and in the same plane of the ring but other
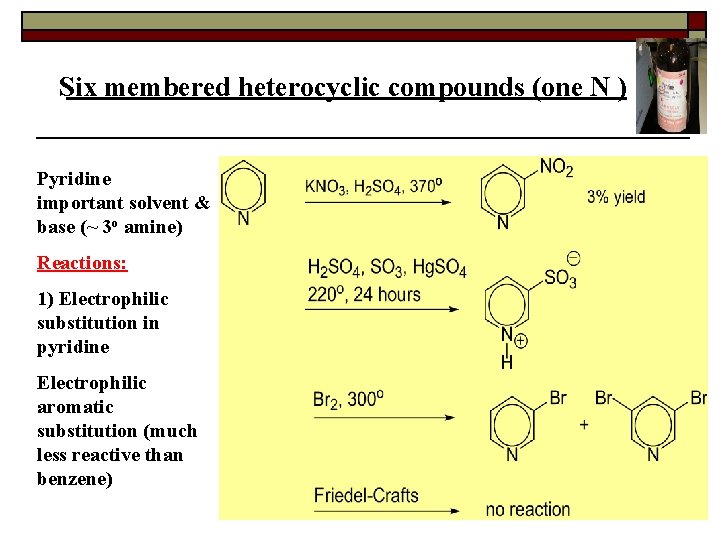
Six membered heterocyclic compounds (one N ) Pyridine important solvent & base (~ 3 o amine) Reactions: 1) Electrophilic substitution in pyridine Electrophilic aromatic substitution (much less reactive than benzene)

Six membered heterocyclic compounds (one N) Substitution takes place at meta position
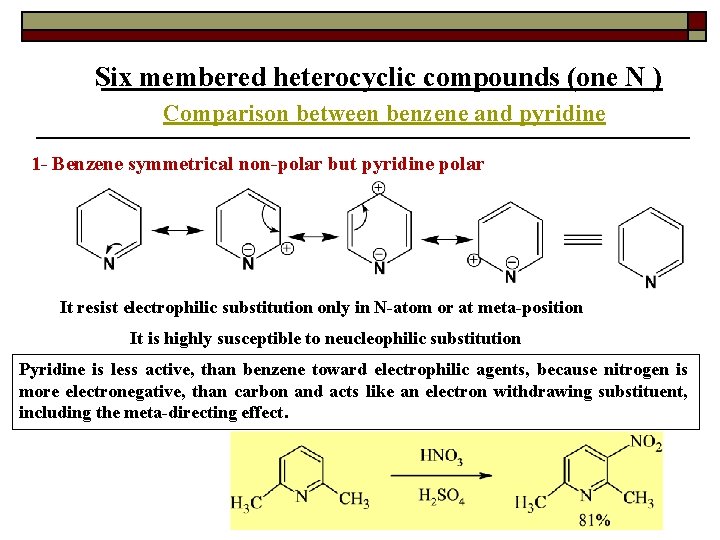
Six membered heterocyclic compounds (one N ) Comparison between benzene and pyridine 1 - Benzene symmetrical non-polar but pyridine polar It resist electrophilic substitution only in N-atom or at meta-position It is highly susceptible to neucleophilic substitution Pyridine is less active, than benzene toward electrophilic agents, because nitrogen is more electronegative, than carbon and acts like an electron withdrawing substituent, including the meta-directing effect.

Six membered heterocyclic compounds (one N ) Comparison between benzene and pyridine 2 - It is basic compound [its basicity less than aliphatic amine]
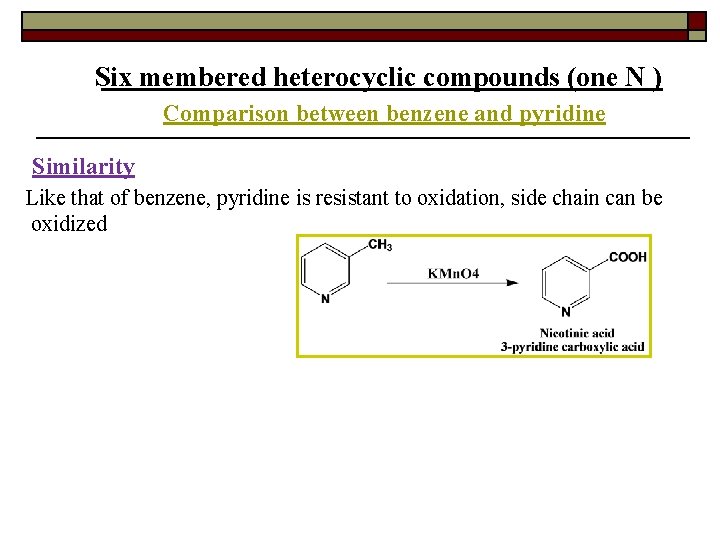
Six membered heterocyclic compounds (one N ) Comparison between benzene and pyridine Similarity Like that of benzene, pyridine is resistant to oxidation, side chain can be oxidized
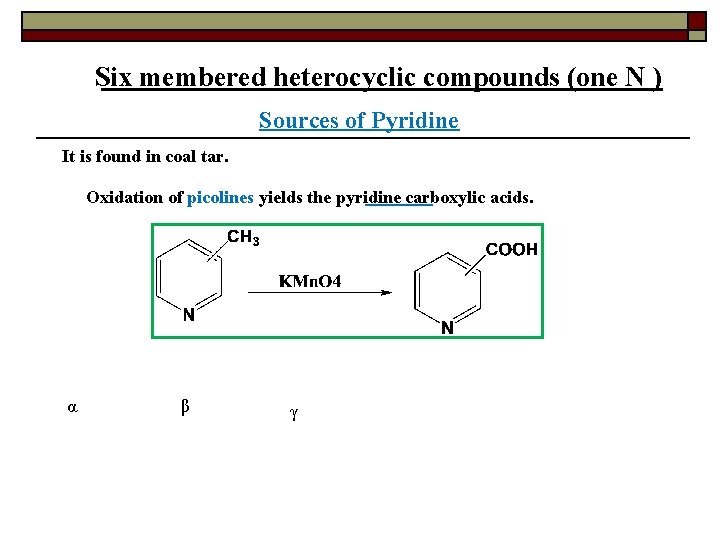
Six membered heterocyclic compounds (one N ) Sources of Pyridine It is found in coal tar. Oxidation of picolines yields the pyridine carboxylic acids. α β γ
Six membered heterocyclic compounds (one N ) α b g

Six membered heterocyclic compounds (one N ) The Nicotinic acid is a Vitamin. The Isonicotinic acid has been used in the form of its hydrazide in treatment of TB.

Six membered heterocyclic compounds (one N ) Synthesis of Pyridine Hantzsch pyridine synthesis The Hantzsch pyridine synthesis typically uses a 2: 1: 1 mixture of a β-keto acid (often acetoacetate), an aldehyde (often formaldehyde), and ammonia or its salt as the nitrogen donor. First, a double hydrogenated pyridine is obtained, which is then oxidized to the corresponding pyridine derivative.
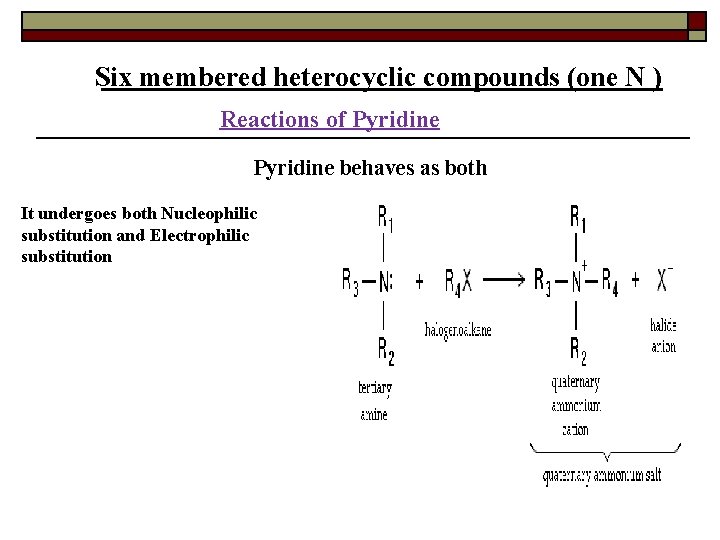
Six membered heterocyclic compounds (one N ) Reactions of Pyridine behaves as both It undergoes both Nucleophilic substitution and Electrophilic substitution
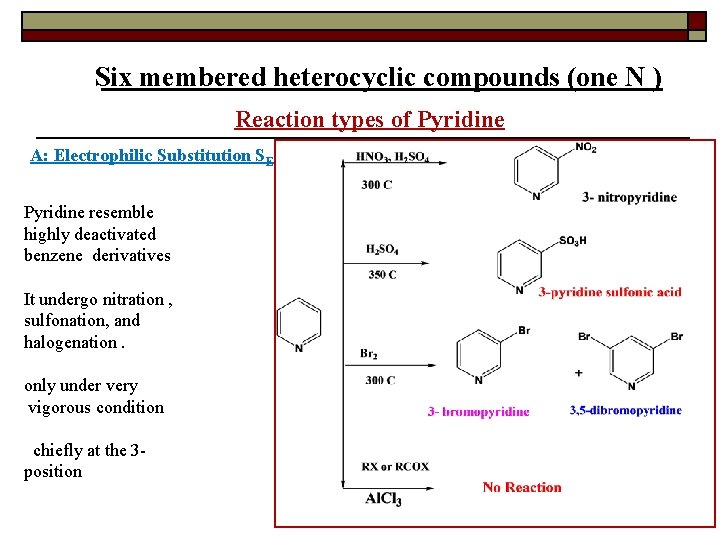
Six membered heterocyclic compounds (one N ) Reaction types of Pyridine A: Electrophilic Substitution SE Pyridine resemble highly deactivated benzene derivatives It undergo nitration , sulfonation, and halogenation. only under very vigorous condition chiefly at the 3 position

Six membered heterocyclic compounds (pyridine ) Reaction types of Pyridine A: Electrophilic Substitution SE mainly at c-3 Why mainly at c-3 ? elec. (+ ve) par
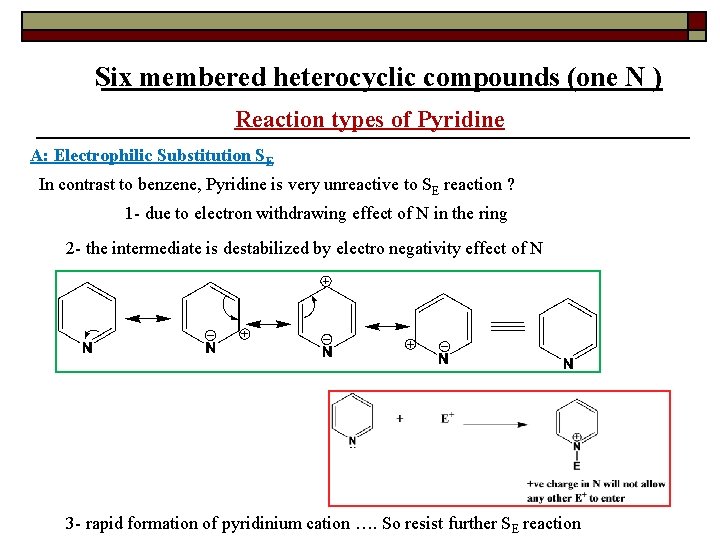
Six membered heterocyclic compounds (one N ) Reaction types of Pyridine A: Electrophilic Substitution SE In contrast to benzene, Pyridine is very unreactive to SE reaction ? 1 - due to electron withdrawing effect of N in the ring 2 - the intermediate is destabilized by electro negativity effect of N 3 - rapid formation of pyridinium cation …. So resist further SE reaction
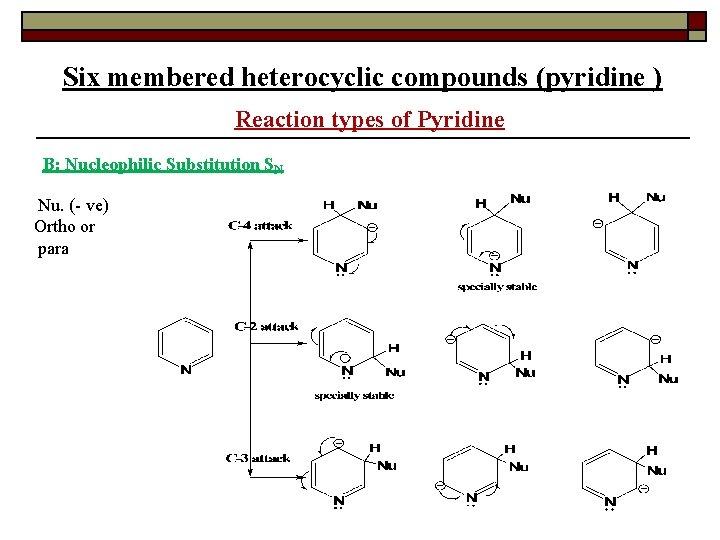
Six membered heterocyclic compounds (pyridine ) Reaction types of Pyridine B: Nucleophilic Substitution SN Nu. (- ve) Ortho or para

Six membered heterocyclic compounds (one N ) Reaction of Pyridine Example 1 - Chichibabin Reaction
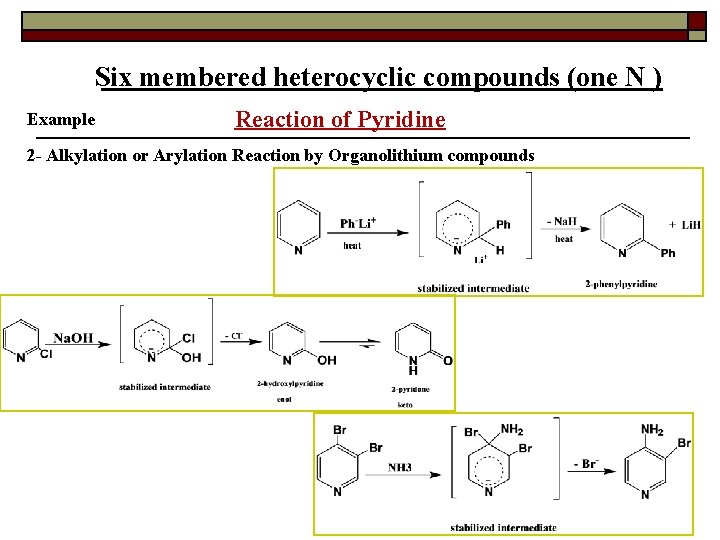
Six membered heterocyclic compounds (one N ) Example Reaction of Pyridine 2 - Alkylation or Arylation Reaction by Organolithium compounds
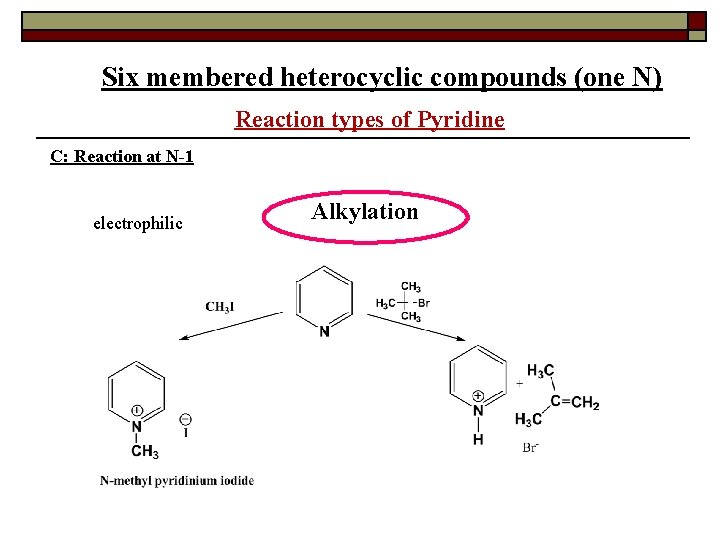
Six membered heterocyclic compounds (one N) Reaction types of Pyridine C: Reaction at N-1 electrophilic Alkylation
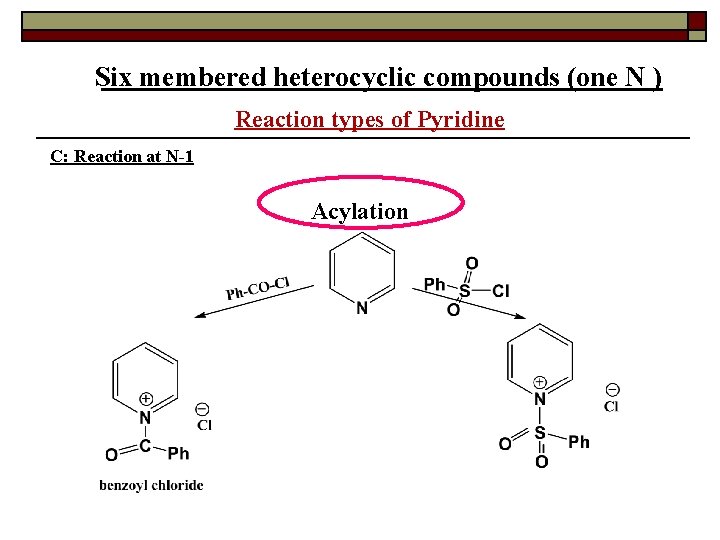
Six membered heterocyclic compounds (one N ) Reaction types of Pyridine C: Reaction at N-1 Acylation
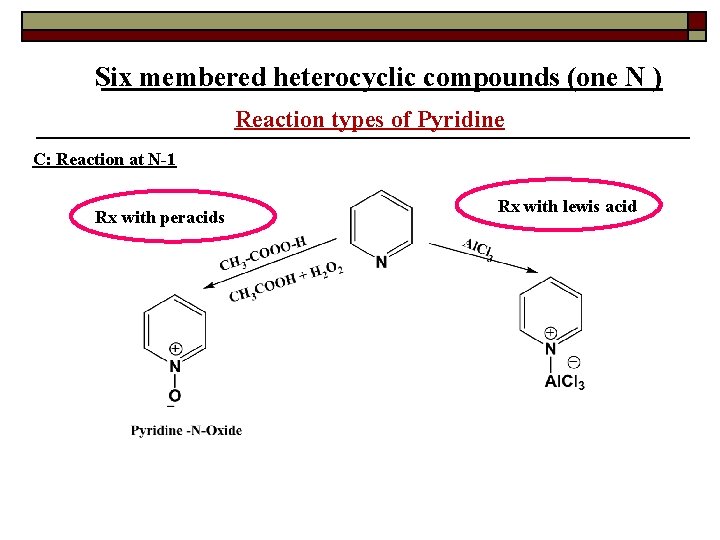
Six membered heterocyclic compounds (one N ) Reaction types of Pyridine C: Reaction at N-1 Rx with peracids Rx with lewis acid

Six membered heterocyclic compounds (one N ) Reaction types of Pyridine Electrophilic ( Nitration)
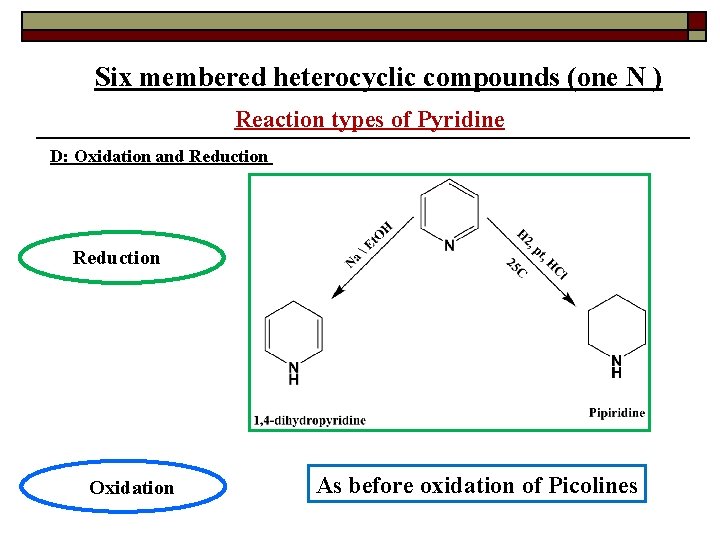
Six membered heterocyclic compounds (one N ) Reaction types of Pyridine D: Oxidation and Reduction Oxidation As before oxidation of Picolines
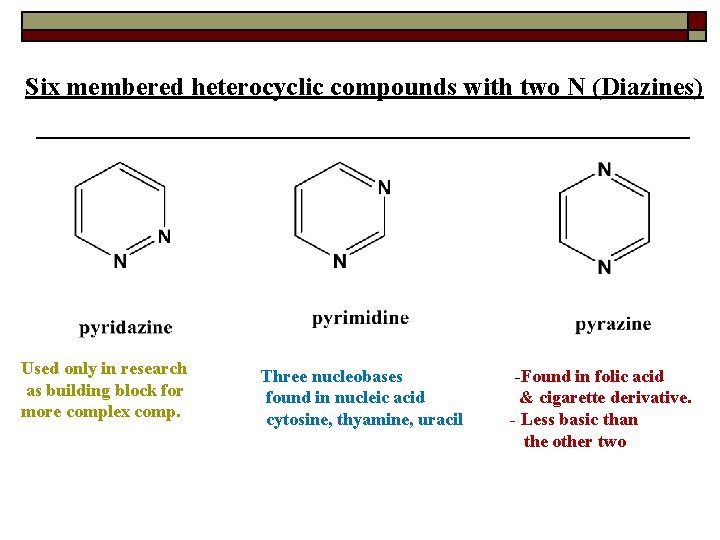
Six membered heterocyclic compounds with two N (Diazines) Used only in research as building block for more complex comp. Three nucleobases found in nucleic acid cytosine, thyamine, uracil -Found in folic acid & cigarette derivative. - Less basic than the other two
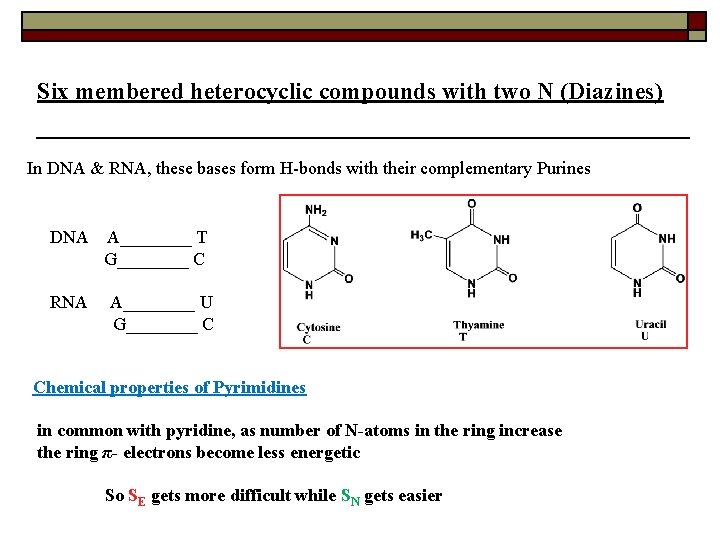
Six membered heterocyclic compounds with two N (Diazines) In DNA & RNA, these bases form H-bonds with their complementary Purines DNA RNA A____ T G____ C A____ U G____ C Chemical properties of Pyrimidines in common with pyridine, as number of N-atoms in the ring increase the ring π- electrons become less energetic So SE gets more difficult while SN gets easier
![Six membered heterocyclic compounds (pyridine ) Fused ring Quinoline Benzo[b]pyridine 1 -azanaphthaline Isoquinoline Benzo[c]pyridine Six membered heterocyclic compounds (pyridine ) Fused ring Quinoline Benzo[b]pyridine 1 -azanaphthaline Isoquinoline Benzo[c]pyridine](http://slidetodoc.com/presentation_image_h2/9d11337177688872ca0f57dcd1547549/image-29.jpg)
Six membered heterocyclic compounds (pyridine ) Fused ring Quinoline Benzo[b]pyridine 1 -azanaphthaline Isoquinoline Benzo[c]pyridine 2 -azanaphthaline Both Quinoline and Isoquinoline are weak bases. Both undergo SE more easly than pyridine in position 5 and 8 on benzoid ring. Like pyridine , N-ring of quinoline and isoquinoline undergo SN at α position
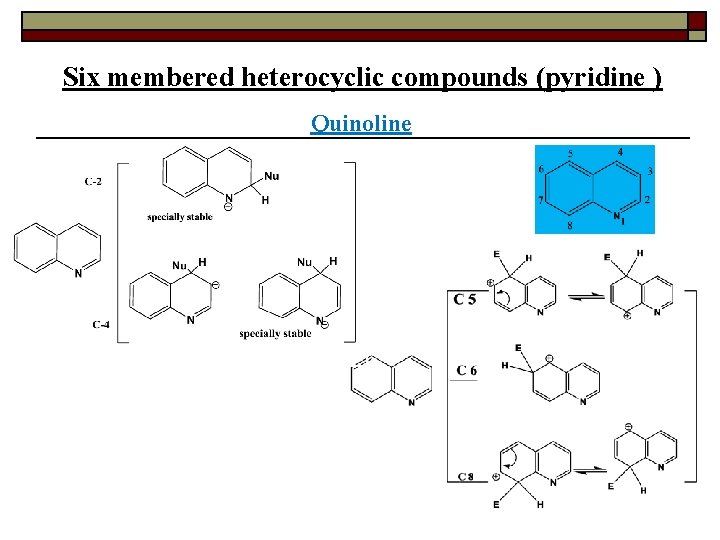
Six membered heterocyclic compounds (pyridine ) Quinoline

Six membered heterocyclic compounds (pyridine ) Isoquinoline
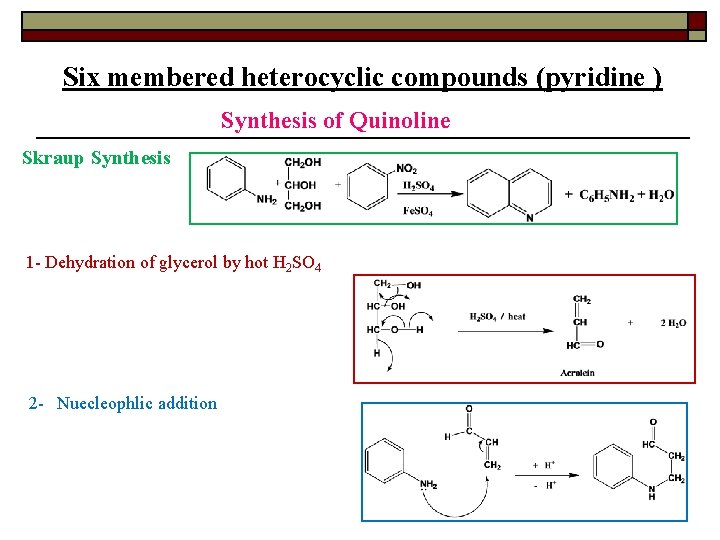
Six membered heterocyclic compounds (pyridine ) Synthesis of Quinoline Skraup Synthesis 1 - Dehydration of glycerol by hot H 2 SO 4 2 - Nuecleophlic addition
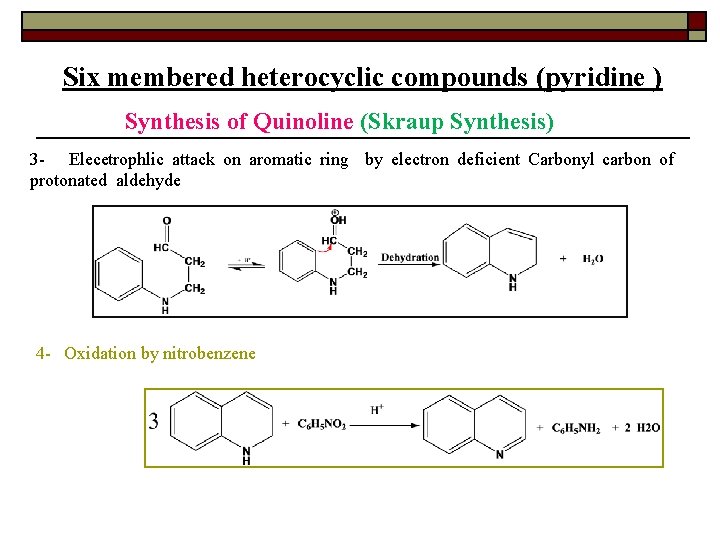
Six membered heterocyclic compounds (pyridine ) Synthesis of Quinoline (Skraup Synthesis) 3 - Elecetrophlic attack on aromatic ring by electron deficient Carbonyl carbon of protonated aldehyde 4 - Oxidation by nitrobenzene

Six membered heterocyclic compounds (pyridine ) Synthesis of Quinoline (Bischler Nepieralski Synthesis)

Six membered heterocyclic compounds (pyridine ) Reactions of Quinoline Electrophilic Substitution Nitration Sulfonation Halogenation

Six membered heterocyclic compounds (pyridine ) Reactions of Quinoline Nucleophilic Substitution Hydroxylation Alkylation Or Arylation with organo -metallic compounds Amination

Six membered heterocyclic compounds (pyridine ) Reactions of Quinoline Oxidation & Reduction Oxidation

Fused Six membered heterocyclic compounds with more than one N Purines Contain pyrimidine fused to imidazole ring

Fused Six membered heterocyclic compounds with more than one N
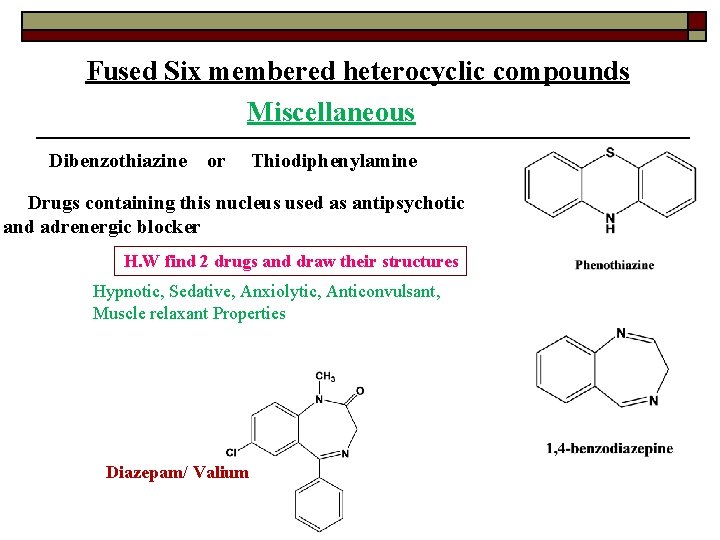
Fused Six membered heterocyclic compounds Miscellaneous Dibenzothiazine or Thiodiphenylamine Drugs containing this nucleus used as antipsychotic and adrenergic blocker H. W find 2 drugs and draw their structures Hypnotic, Sedative, Anxiolytic, Anticonvulsant, Muscle relaxant Properties Diazepam/ Valium

- Slides: 41