Pharmaceutical Calculations Altering Product Strength Use of Stock




























- Slides: 29

Pharmaceutical Calculations: Altering Product Strength, Use of Stock Solutions, and Alligation Danielle Del. Villano, Pharm. D.

Objectives • Perform calculations for altering product strength by dilution, concentration, or fortification • Perform calculations for the preparation and use of stock solutions • Apply alligation medial and alligation alternate in problem solving

Keep in mind • Solution = solute + solvent – If w/v – solute in g / solution in m. L – If v/v – solute in m. L / solute+solvent in m. L – If w/w – solute in g / solute+solvent in g • Changing x% to fraction – X g / 100 m. L • Changing 1: x ratio strength to fraction – 1 g / x m. L

Altering Product Strength • Concentrated – Addition of active ingredient • Diluted – Addition of diluent • Fortification – Addition of a calculated quantity of additional therapeutic agent

Relationship between Strength and Total Quantity • If mixture is diluted to twice its original quantity, then strength will be reduced by one half – Start with 100 m. L of 5% solution (5 g drug) – Add 100 m. L, now 200 m. L of 2. 5% solution (5 g drug) • If a mixture is concentrated by evaporation to one half its original quantity, the strength will be doubled – Start with 100 m. L of 5% solution (5 g drug) – Evaporate, now 50 m. L of 10% solution (5 g drug)

Several Methods for Solving • Inverse proportions • Q 1 C 1 = Q 2 C 2 • Determine the quantity of active solute needed and then calculate the quantity of available solution to provide needed amount – “Box method”

Box method • Always keep it the same • When setting it up Total amount of solution Strength (% or ratio) Amount of solute – Fill in everything from the problem – Circle what you are trying to solve for – Keep your units

Problem 1 • If 500 m. L of a 15% v/v solution are diluted to 1500 m. L, what will be the percentage strength (v/v)? 500 m. L solution Dilute with 1000 1500 m. L solution m. L water 15% 5% 75 g drug 15 g 100 m. L x This stays the same = xg 500 m. L = 75 g drug xg 100 m. L x = 75 g 1500 m. L = 5%

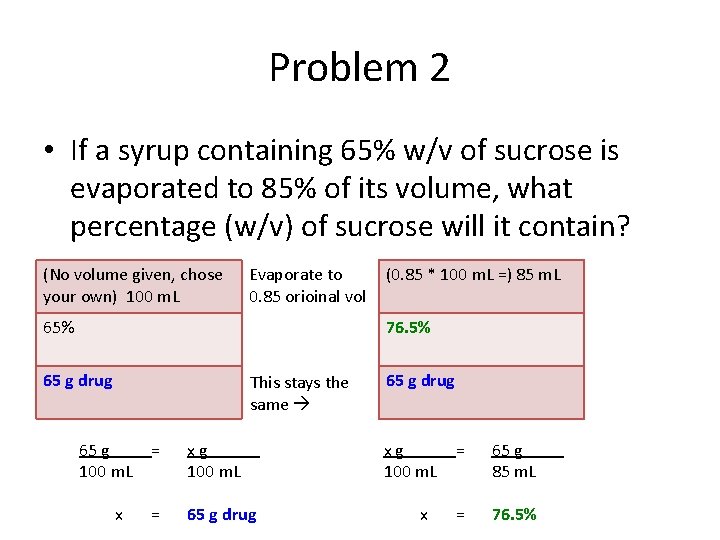
Problem 2 • If a syrup containing 65% w/v of sucrose is evaporated to 85% of its volume, what percentage (w/v) of sucrose will it contain? (No volume given, chose your own) 100 m. L Evaporate to 0. 85 orioinal vol 65% (0. 85 * 100 m. L =) 85 m. L 76. 5% 65 g drug This stays the same 65 g 100 m. L x = xg 100 m. L = 65 g drug xg 100 m. L x = 65 g 85 m. L = 76. 5%

Problem 3 • How many milliliters of a 1: 5000 w/v solution of the preservative laurakonium chloride can be made from 125 m. L of a 0. 2% solution? 125 m. L 1250 m. L 0. 2% 1: 5000 This stays the same 0. 25 g drug 0. 2 g 100 m. L x = xg 125 m. L = 0. 25 g drug 1 g = 5000 m. L x = 0. 25 g x m. L 1250 m. L

Problem 4 • If a cough syrup contains in each teaspoonful, 1 mg of chlorpheniramine maleate and if a pharmacist desired to double the strength, how many milligrams of that ingredient would be needed to be added to a 60 m. L container of the syrup? Assume no increase in volume. 60 m. L 1 mg / 5 m. L Want to double this amnt = + x mg

Problem 5 • How many milliliters of a 1: 400 w/v stock solution should be used in preparing 1 gallon of a 1: 2000 w/v solution? Add x amount of (1 gallon) 3785 m. L diluent 1: 400 1: 2000 This stays the same

Problem 6 • How many milliliters of a 1: 16 solution of sodium hypochlorite should be used in preparing 5000 m. L of a 0. 5% solution of sodium hypochlorite for irrigation?

Problem 7 • How much drug should be used in preparing 50 m. L of a solution such that 5 m. L diluted to 500 m. L will yield a 1: 1000 solution?

Problem 8 • How many milliliters of a 17% concentrate of benzalkonium chloride should be used in preparing 300 m. L of a stock solution such that 15 m. L diluted to 1 liter will yield a 1: 5000 solution?

Problem 9 • How many milliliters of water should be added to a pint of 5% w/v solution to make a 2% w/v solution?

Problem 10 • How many milliliters of 85% w/w phosphoric acid having a specific gravity of 1. 71 should be used in preparing 1 gallon of ¼% w/v phosphoric acid solution to be used for bladder irrigation?

Problem 11 • How many grams of 20% benzocaine ointment and how many grams of ointment base (diluent) should be used in preparing 5 lbs of 2. 5% benzocaine ointment?

Problem 12 • How many grams of coal tar should be added to 3200 g of 5% coal tar ointment to prepare an ointment containing 20% coal tar?

Alligation Medial • “Weighted average” percentage strength of a mixture of two or more substances can be calculated • Percentage strength of compound is multiplied by its corresponding quantity • Sum is divided by total quantity of the mixture to give percentage strength of the mixture

Problem 13 • What is the percentage of zinc oxide in an ointment prepared by mixing 200 g of 10% ointment, 50 g of 20% ointment, and 100 g of 5% ointment? 0. 10 g * 0. 20 * 0. 05 * TOTALS 200 g = 50 g = 100 g = 350 g 20 g 10 g 5 g 35 g x g = 35 g 100 g 350 g x = 10%

Problem 14 • What is the percentage strength of alcohol in a mixture of 500 m. L of a solution containing 40% v/v alcohol, 400 m. L of a second solution containing 21% v/v alcohol, and a sufficient quantity of a nonalcoholic third solution to make a total of 1000 m. L?

Alligation Alternate

Problem 15 • In what proportion should 20% benzocaine ointment be mixed with an ointment base to produce a 2. 5% benzocaine ointment? 20% 2. 5 parts 2. 5% 0% 17. 5 parts 20 total parts The 20% benzocaine and ointment base shoule be mixed at a 2. 5: 17. 5 ratio, or 1: 7.

Problem 16 • A hospital pharmacist wants to use three lots of zinc oxide containing, respectively, 50%, 20% and 5% zinc oxide. In what proportion should they be mixed to prepare a 10% zinc oxide ointment? 50% 5 parts The 50%, 20% and 5% lots should be mixed in a 5: 5: 50 proportion (or 1: 1: 10) 5 parts 20% 10% 5% 10 parts + 40 parts = 50 parts 60 parts total ointment

Problem 17 • How many milliliters of 50% w/v dextrose solution and how many milliliters of 5% w/v dextrose solution are required to prepare 4500 m. L of a 10% w/v solution?

Problem 18 • In what proportion must glycerin with a specific gravity of 1. 25 and water be mixed to prepare a liquid having a specific gravity of 1. 10?

Questions

Reference • Ansel, H. C. (2009) Phamaceutical Calculations (13 th Ed. ). Philadelphia: Lippincott Williams & Wilkins, and Wolters Kluwer Publishers