Phage Biology Phage Therapy Basics Brief History Mario

Phage Biology & Phage Therapy Basics & Brief History Mario Vaneechoutte
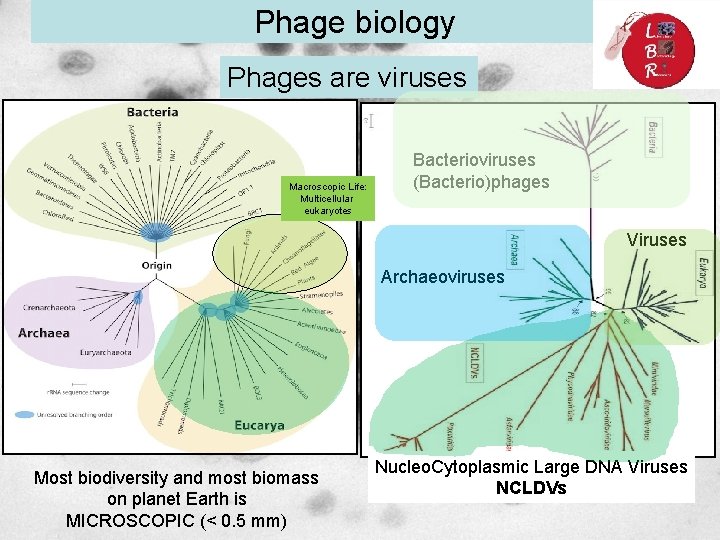
Phage biology Phages are viruses Macroscopic Life: Multicellular eukaryotes Bacterioviruses (Bacterio)phages Viruses Archaeoviruses Most biodiversity and most biomass on planet Earth is MICROSCOPIC (< 0. 5 mm) Nucleo. Cytoplasmic Large DNA Viruses NCLDVs
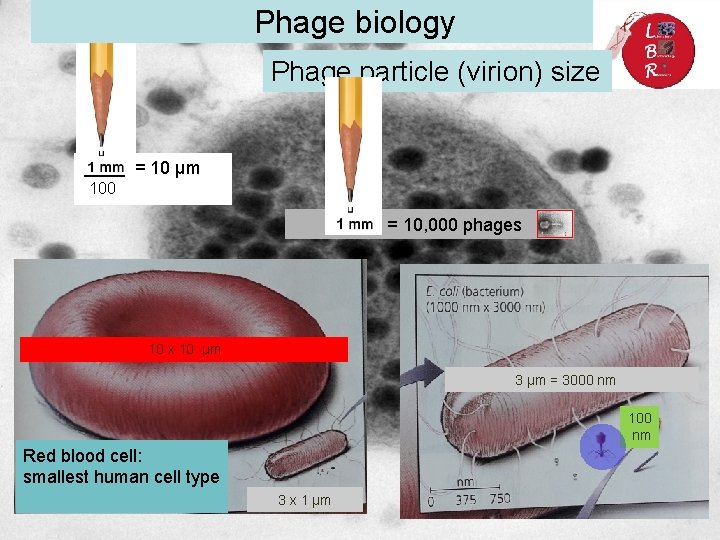
Phage biology Phage particle (virion) size == 10 µmµm 1000 1 mm = 10, 000 phages 10 x 10 µm 3 µm = 3000 nm 100 nm Red blood cell: smallest human cell type 3 x 1 µm
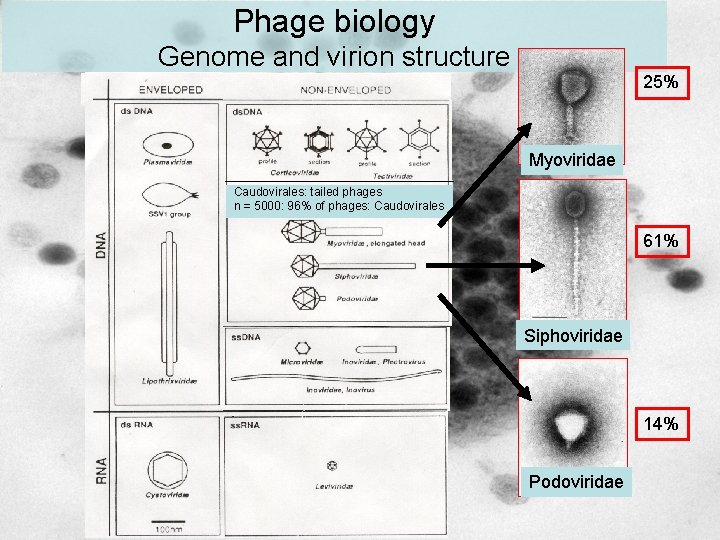
Phage biology Genome and virion structure 25% Myoviridae Caudovirales: tailed phages n = 5000: 96% of phages: Caudovirales 61% Siphoviridae 14% Podoviridae
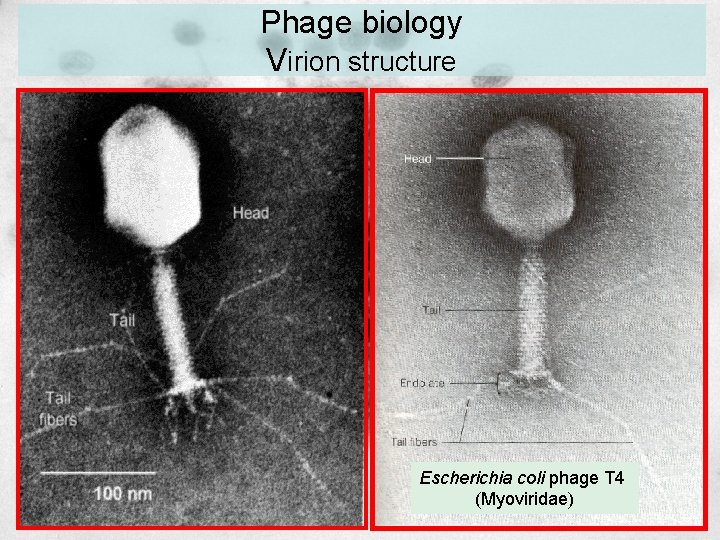
Phage biology Virion structure Escherichia coli phage T 4 (Myoviridae)
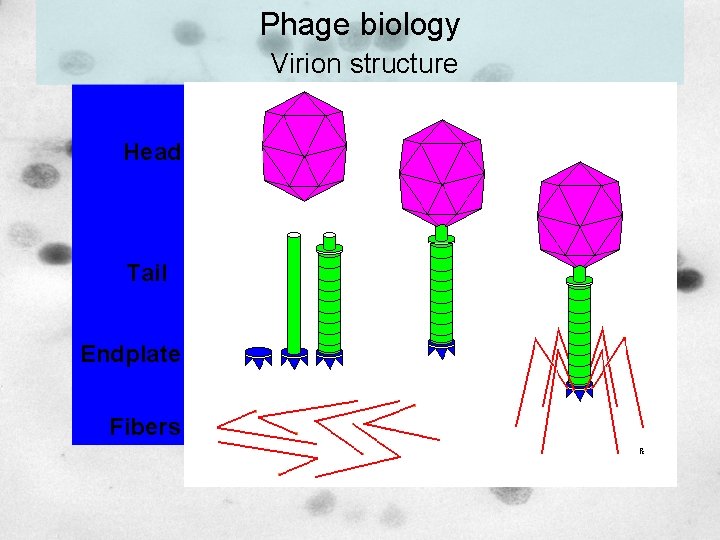
Phage biology Virion structure Head Tail Endplate Fibers
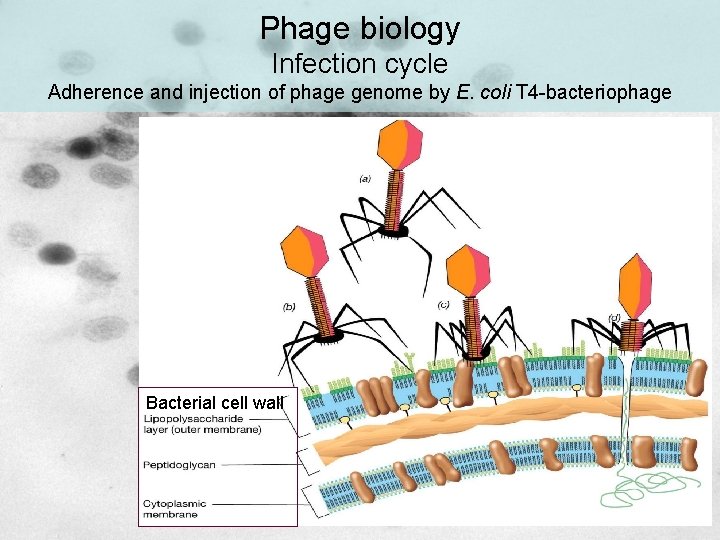
Phage biology Infection cycle Adherence and injection of phage genome by E. coli T 4 -bacteriophage Bacterial cell wall
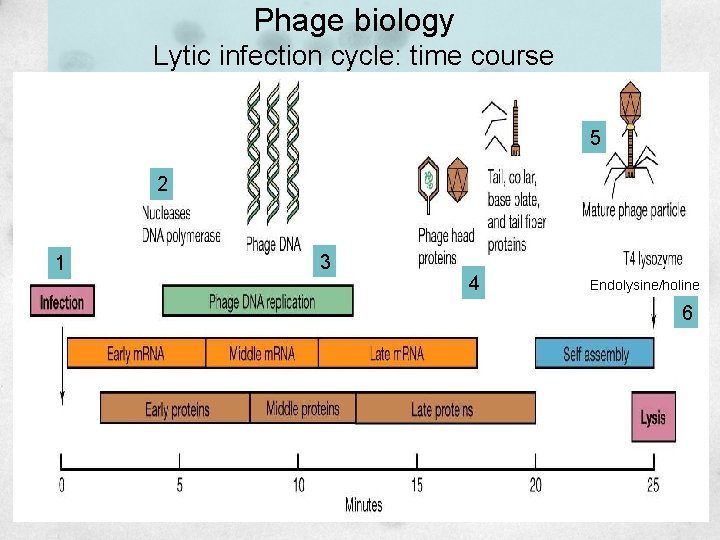
Phage biology Lytic infection cycle: time course 5 2 1 3 4 Endolysine/holine 6

Phage biology Infection cycles chronic, lytic, lysogenic Chronic phages (e. g. E. coli phage M 13): Phage particles (virions) are formed immediately after infection. Virions leave the host cell without lysis. Virulent phages (e. g. E. coli phage T 4): Phage particles (virions) are formed immediately after infection. The bacterial cell is lysed and virions are released. only lytic cycle = Therapeutic phages Temperate phages (e. g. E. coli phage Lambda): Phages can ‘choose’ between a lytic cycle: lytic phase or The phage genome can be incorporated into the bacterial genome as a prophage and duplicated together with the bacterial genome: lysogenic phase
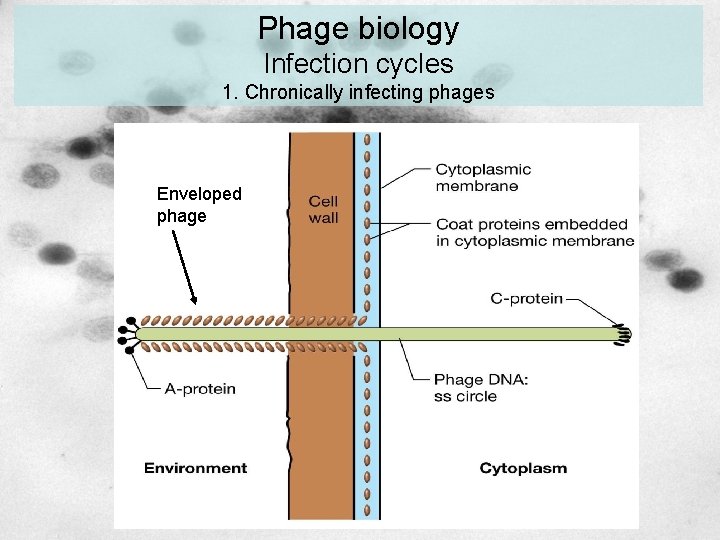
Phage biology Infection cycles 1. Chronically infecting phages Enveloped phage
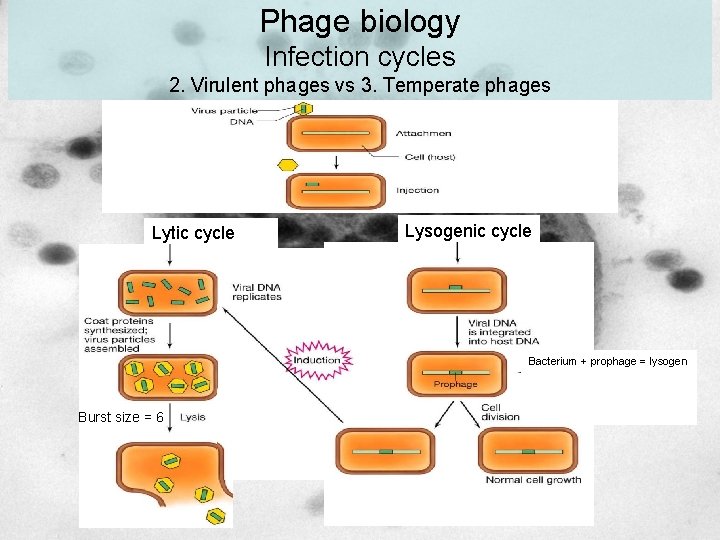
Phage biology Infection cycles 2. Virulent phages vs 3. Temperate phages Lytic cycle Lysogenic cycle Bacterium + prophage = lysogen Burst size = 6
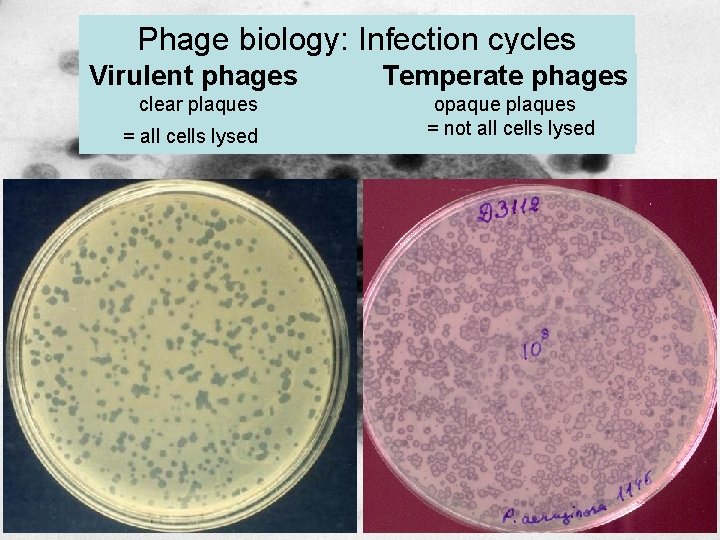
Phage biology: Infection cycles Virulent phages Temperate phages clear plaques opaque plaques = not all cells lysed = all cells lysed
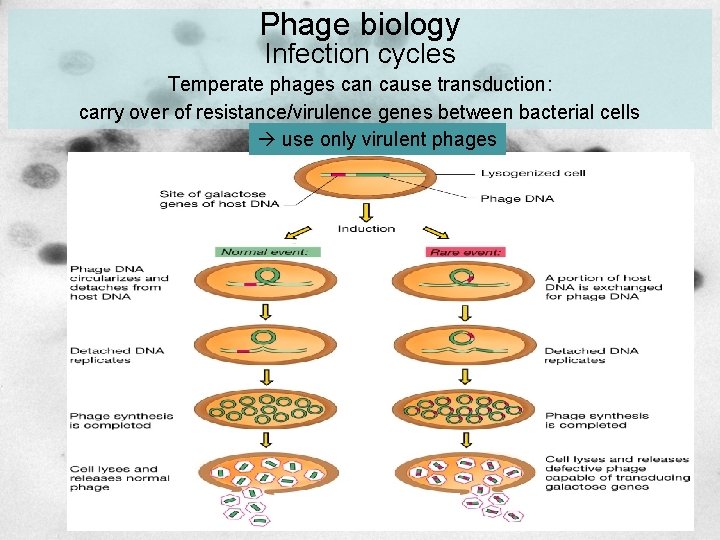
Phage biology Infection cycles Temperate phages can cause transduction: carry over of resistance/virulence genes between bacterial cells use only virulent phages
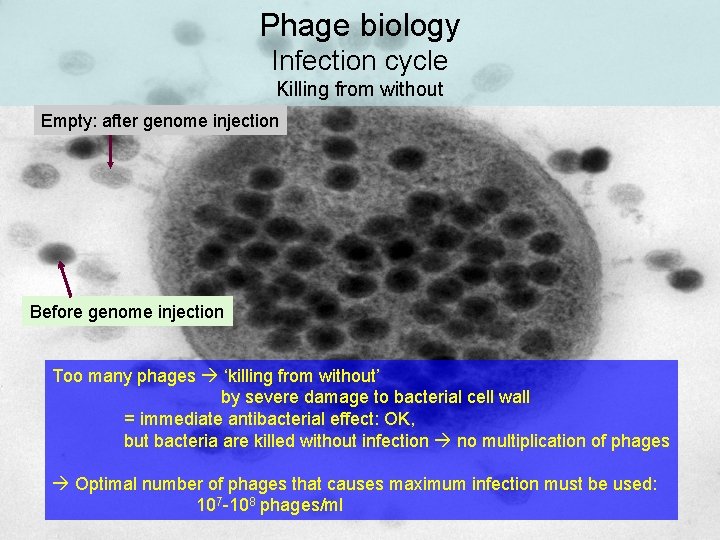
Phage biology Infection cycle Killing from without Empty: after genome injection Before genome injection Too many phages ‘killing from without’ by severe damage to bacterial cell wall = immediate antibacterial effect: OK, but bacteria are killed without infection no multiplication of phages Optimal number of phages that causes maximum infection must be used: 107 -108 phages/ml

Phage therapy: history Frederick Twort 1915 Félix d'Herelle 1917

Phage therapy: history E. H. Hankin in 1896: The first evidence for a viral-like agent with antibacterial properties Ganges river in India capable of passing through a porcelain filter smaller than a bacterial cell reduces titres of the bacterium Vibrio cholerae Hankin suggested that it might help to explain the low incidence of cholera in people using water from the Ganges = holy river with health-promoting properties Adhya & Merril. 2006. The road to phage therapy. Nature 443: 754 -755. https: //en. wikipedia. org/wiki/Ernest_Hanbury_Hankin EH. 1896. L‘action bactericide des Eaux de la Jumna et du Gange sur le vibrion du cholera. Ann Inst Pasteur 10: 511.

Phage therapy: history 1896: Ernest Hanbury Hankin 1915: Frederick Twort 1917: Felix d'Herelle: Sur un microbe invisible antagoniste des bacilles dysentériques. Acad Sci Ser D 165: 373. 1921: First phage therapy report published by Bruynoghe & Maisin. See later … 1923: George Eliava starts the Microbiology Institute in Tbilisi, Georgia: ������ � Sakar t ve l o 1925: d’Herelle F. Essai de traitement de la peste bubonique par le bactériophage. La Presse Med 33: 1393 -1394. 1934: Eliava learns about phage therapy from d’Herelle. 1936: d'Herelle is invited by (Georgian) Stalin and Eliava to the (Eliava) Institute. 1937: Eliava is executed (by Beria) – d’Herelle flees, back to France.

Phage therapy: history Phages: discovered during WW I Antibiotics: applied during WW II The interbellum: first golden age for phage therapy in the West (are we entering the second golden age now? ) Commercialization of phages in France and USA in 1930’s L’Oréal: Bacté-intesti-phage, Bacté-pyo-phage, Bacté-staphylo-phage Eli Lilly: Colo-lysate, Entero-lysate, Staphylo-lysate Phage therapy was abandoned in the West, because of lack of understanding of the high specificity and of the exact mode of action of phages exaggerated claims of effectiveness: urticaria, herpes, eczema the rise of broad-spectrum antibiotics: penicillin in WW II but phage therapy research continued in Georgia, Russia and Poland

Phage therapy: Weaknesses 1. Narrow spectrum Disadvantage: species and clone need to be identified before application not sufficient time in case of acute (life-threatening) infection Is at the same time an advantage: commensal microflora not affected “Elk nadeel hep sijn foordeel” (Johan Cruyff) Solutions: 1. Use of phage mixtures (cocktails) 2. Application in chronic infections: time to select appropriate phages 3. Broad spectrum phages (e. g. ISP, most S. aureus, incl. MRSA!) exist. 4. Add phages to antibiotics broader spectrum less chance of development of antibiotic resistance less chance of development of phage resistance ethically easier to apply phages on top of standard care

Phage therapy: Weaknesses 2. Bacterial resistance Major strategies of bacteria for developing phage resistance: 1. Mutation of cell wall receptors which are used by phages to adhere. 2. DNA restriction/modification systems: nonmodified (phage) DNA is restricted. 3. CRISPR/Cas: adaptive immunity in bacteria! 4. Lysogenic phages can induce resistance to other phages CONSIDERATIONS & SOLUTIONS: Mutant bacteria can become susceptible for other phages. Mutant bacteria can loose virulence. E. coli K 1 -phages induce phage-resistant E. coli but these are K 1 negative: reduced virulence (1). Other solutions: see next slides 1. Smith WH and Huggins MB. 1982. Succesful treatment of experimental Escherichia coli infections in mice using phage: its general superiority over antibiotics. J. Gen. Microbiol. 128: 307 -318.

Phage therapy: Strengths Current problems in infectious diseases 1. Intrinsic antibiotic resistance is increasing. 2. Limited number of antibiotics in the pipeline. Companies less interested because application of new antibiotics will be restrained to last resort application: to avoid development of resistance. 3. Antibiotics not efficient against biofilm: chronic, recurrent infections due to poor penetration through biofilm matrix lowered metabolism: dormant bacteria (also problem for phages? ) Chronic biofilm-related infections: infection of airways in CF-patients: Pseudomonas aeruginosa chronic otitis media: Haemophilus influenzae recurrent urinary tract infection: uropathogenic Escherichia coli burn wounds: Pseudomonas aeruginosa, Staphylococcus aureus foreign object infections: catheters, protheses: Staphylococcus spp. , Propionibacterium acnes !!! chronic infection: time to select the best phages. Some phages can penetrate the biofilm matrix!

Phage therapy: Strengths 4. Phages have a narrow spectrum: infect only one species/few strains. * No effect on commensal microflora * No cross-resistance effects: resistance to phage 1 <> resistance to phage 2 * Treatment can be customized/personalized 5. Phage kinetics differ from kinetics of inert antibiotics. * Antibiotics are being cleared from the body, while phages multiply by factor 10 -50 (burst size): (in theory) one single dose can be sufficient to treat an infection * Less dependent on blood stream: phages pass also BBB (1) * Phages are cleared when they have cleared their bacterial host 6. No relation to antibiotic resistance: MDR bacteria can be treated. e. g. , ISP: Intravenous Staphylococcal Phage: active against MRSA 7. Phages can be combined with antibiotics: different killing mechanisms. 1. Dabrowska et al. 2005. Bacteriophage penetration in vertebrates. J Appl Microbiol 98: 7 -13.
Phage therapy: Strengths 8. Some phages can escape resistance: ISP, active against 95% of S. aureus strains including MRSA is used since decades, with almost no resistance development 9. In case of bacterial resistance, phages can evolve: Phages can be ‘trained’ against resistant bacteria One can ‘fish’ for new phages

Phage therapy: Strengths 10. Phages are safe: Theoretical considerations 1 Phages are safe by definition: viruses which infect only bacteria 10. 1. Bacteriophages infect specifically bacteria since they need to recognize bacterial cell wall structures: peptidoglycane, LPS. Even among bacteria: very narrow spectrum of bacterial strains that can be infected. 10. 2. Bacteriophages that were manipulated genetically to infect mammalian cells were not able to multiply inside the mammalian cells after infection (1). 10. 3. No bacteriophage genes can be found in the human genome retro-viruses have left hundreds of genes integrated into the human genome. In summary, bacteriophages have no tropism towards mammalian (eukaryote) cells and cannot multiply in them. Di Giovine et al. 2001. Binding properties, cell delivery, and gene transfer of adenoviral penton based displaying bacteriophage. Virology 282: 102 -112.

Phage therapy: Strengths 10. Phages are safe: Theoretical considerations 2 10. 4. Bacteriophages are numerous and ubiquitous: but no phage infections have been reported. Numerous (1) Estimate of total number of tailed phage particles on Earth: 4 -6 x 1031 = 10 -fold of number of bacterial cells. Compare: Number of stars in Universe: 1020 - 1024 Ubiquitous (2) Up to 109 phages per ml of surface waters In animal sera, in vaccines, in food Escherichia coli phages in 11% of faeces of healthy persons Bacteroides fragilis phages in 68% of faeces of healthy persons "We live in a sea of phages" still no infections with phages have been reported 1. Bergh. 1989. Nature 340: 467 -468 2. Whitamn et al. 1998. PNAS 95: 6578 -6583

Phage therapy: Strengths 10. Phages are safe: Empirical evidence: Humans 1 10. 5. During the long history of using bacteriophages as therapeutic agents bacteriophages have been administered to thousands/millons of humans (1) (i) orally, in tablet or liquid formulations (105 to 1011 bacteriophages/dose) (ii) rectally (iii) locally: skin, eye, ear, nasal mucosa, burn wounds, rinses and creams (iv) as aerosols or intrapleural injections (v) intravenously: ISP 1. Sulakvelidze et al. 2001. Bacteriophage therapy. Antimicrob Agents Chemother 45: 649 -659. 2. Weber-Dabrowska et al. 2000. Bacteriophage therapy of bacterial infections: an update of our institute’s experience. Arch Immunol Therap Experiment 48: 547 -551.

Phage therapy: Strengths 10. Phages are safe: Empirical evidence: Humans 2 10. 5. "Vaccination" study in Tbilisi, Georgia (1965) 17, 044 children ingested bacteriophages against Shigella dysenteriae No adverse effects were reported. Babalova et al. 1968. Preventive value of dried dysentery bacteriophage. Zh. Mikrobiol. Epidemiol. Immunobiol. 2: 143 -145.

Phage therapy: Strengths 10. Phages are safe: Experimental evidence: Humans 3 10. 5. E. coli bacteriophage ΦX 174 is IV injected since decades to test the influence on the immune response of different medicines. E. g. IV injection of 109 ΦX 174 bacteriophages/kg body weight, twice, in 18 patients with chronic renal failure (1). This research group uses this approach since the early 70 s without reporting any adverse effects (2, 3). 1. Bearden et al. 2005. Rituximab inhibits the in vivo primary and secondary antibody response to a neoantigen, bacteriophage ΦX 174. Am. J. Transplant. 5: 50 -57. 2. Ochs et al. 1971. Immunologic responses to bacteriophage ΦX 174 in immunodeficiency diseases. J. Clin. Investigation 50: 2550 -2558. 3. Wedgwood et al. 1975. The recognition and classification of immunodeficiency diseases with bacteriophage ΦX 174. Birth Defects Orig. Artic. Ser. 11: 331 -338.
Phage therapy: Strengths 11. Phages are efficient: Experimental evidence: Animals 1 11. 1. Intramuscular injection (single) in one leg with bacteriophage MW to treat intramuscular E. coli infection in the other leg in mice is more effective than multiple IM administration of antibiotics + phages can ‘migrate/travel’ through the body to other sites of infection Smith WH, Huggins MB. 1982. Succesful treatment of experimental Escherichia coli infections in mice using phage: its general superiority over antibiotics. J. Gen. Microbiol. 128: 307 -318.

Phage therapy: Strengths 11. Phages are efficient: Empirical evidence: Humans 1 11. 2. "Vaccination" study in Tbilisi, Georgia (1965) 30. 769 children aging 6 months to 7 years old 17. 044 children ingested bacteriophages against Shigella dysenteriae 13. 725 children, living at the opposite side of the streets, served as a control group. Dysentery incidence in control group is 2. 6 fold higher than phage treated group. Babalova et al. 1968. Preventive value of dried dysentery bacteriophage. Zh Mikrobiol Epidemiol Immunobiol 2: 143 -145.

Phage therapy: Strengths 11. Phages are efficient: Empirical evidence: Humans 2 11. 3. Reported experience from Poland Georgia 1307 patients with suppurative bacterial infections caused by multidrug-resistant bacteria of different species were treated with specific bacteriophages (BP). full recovery in 1123 cases (85. 9%) transient improvement in 134 cases (10. 9%) ineffective only in 50 cases (3. 8%) high efficacy of phage therapy in combating difficult bacterial infections which do not respond to treatment with the available antibiotics. Chanishvili N. 2012. Phage therapy--history from Twort and d'Herelle through Soviet experience to current approaches. Adv Virus Res 83: 3 -40. Weber-Dabrowska B. et al. 2000. Bacteriophage therapy of bacterial infections: an update of our institute’s experience. Arch Immunol Therap Experiment 48: 547 -551.
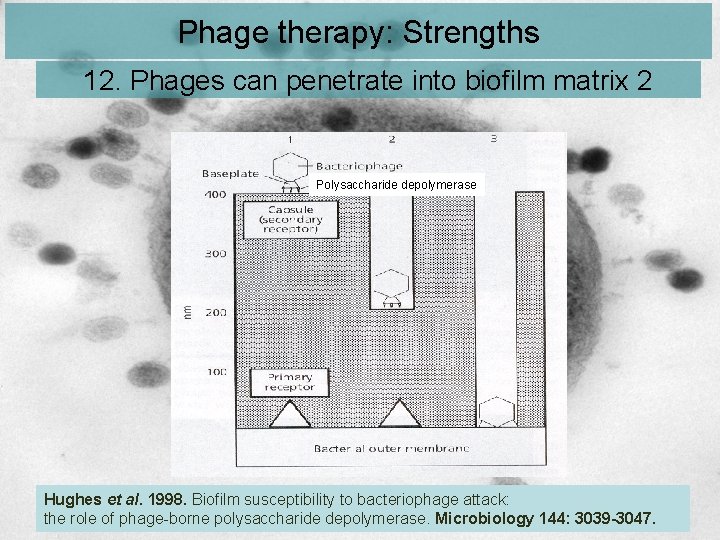
Phage therapy: Strengths 12. Phages can penetrate into biofilm matrix 2 Polysaccharide depolymerase Hughes et al. 1998. Biofilm susceptibility to bacteriophage attack: the role of phage-borne polysaccharide depolymerase. Microbiology 144: 3039 -3047.
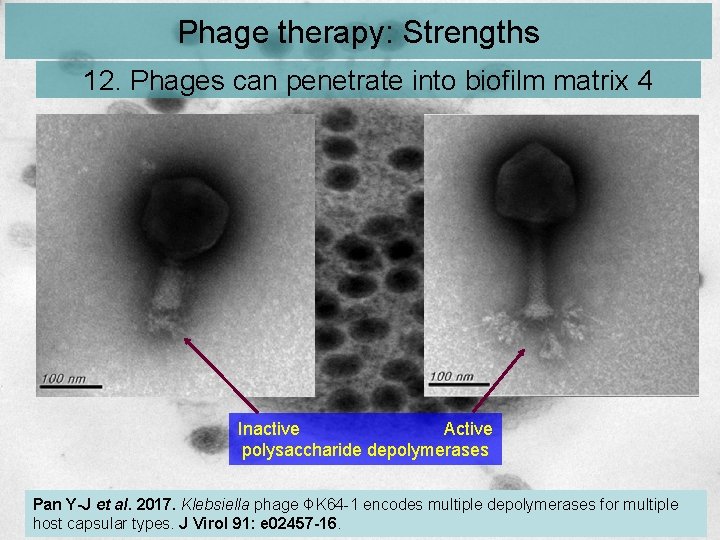
Phage therapy: Strengths 12. Phages can penetrate into biofilm matrix 4 Inactive Active polysaccharide depolymerases Pan Y-J et al. 2017. Klebsiella phage ΦK 64 -1 encodes multiple depolymerases for multiple host capsular types. J Virol 91: e 02457 -16.
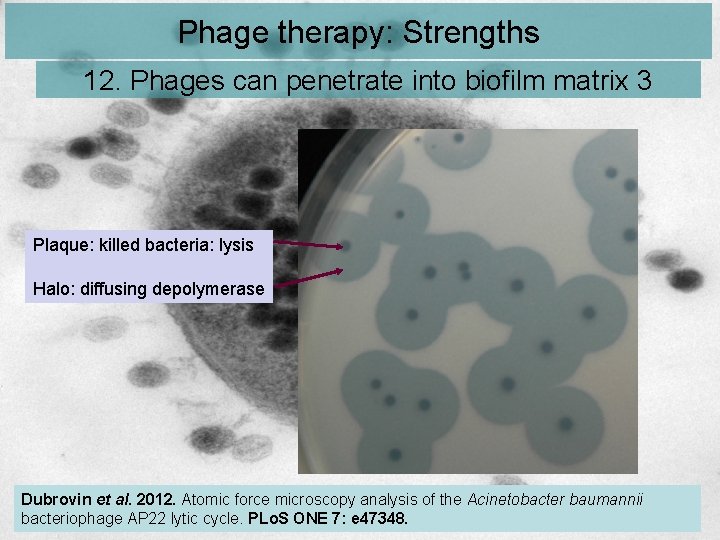
Phage therapy: Strengths 12. Phages can penetrate into biofilm matrix 3 Plaque: killed bacteria: lysis Halo: diffusing depolymerase Dubrovin et al. 2012. Atomic force microscopy analysis of the Acinetobacter baumannii bacteriophage AP 22 lytic cycle. PLo. S ONE 7: e 47348.

Phage therapy: history & Belgium First published report: BELGIUM Bruynoghe R & Maisin J. 1921. Essais de thérapeutique au moyen du Bactériophage du Staphyloccoque. J Compt Rend Soc Biol 85: 1120 -1121.

Phage therapy: history & Belgium 1934 -1936: phages from France to Georgia Félix D’Herelle & George Eliava

Phage therapy: history & Belgium Phages from Sakartvelo to Belgium Maya goes fishing for phages from the Ghent University Hospital sewage Vaneechoutte M, Chanishvili N (Eliava Insitute, Tbilisi, Georgia). *2002 - 2004. Application of bacteriophages in the prevention of secondary bacterial infections in cystic fibrosis patients. INTAS Project 03 -51 -6610. * 2002 -2004. Susceptibility of Pseudomonas aeruginosa strains obtained from cystic fibrosis patients to phages. NATO grant Sa-LST. CLG. 978597.

Phage therapy: history Jean-Paul Pirnay & Maia Merabishvili 12 October 2009: Ramelot Award

Phage therapy: regulatory framework Crossing the Rubicon In Belgium, since last year, magistral phages can be used to treat patients Gilbert Verbeken, Daniel De Vos & Jean-Paul Pirnay (QAMH) Alain Fauconnier (FAGG)

Phage therapy: regulatory framework Magistral phages can be used to treat patients in Belgium

Phage biology & phage therapy Summary Phages are everywhere: The world is a phage. We live in a sea of phages. Phages are safe Phages are efficient for many patients/cases also against antibiotic resistant bacteria (and against bacteria in biofilm: chronic infection) Phages are not always efficient! Phages can be applied as magistral preparations: (only) in Belgium (in EC)! Phage ISP is highly active against MRSA!

Many, many thanks to Maya Merabishvili Nina Chanishvili Lab. MCT/LBR/Eliava Gilbert Verbeken Daniel De Vos Jean-Paul Pirnay Lab. MCT, QAMH Christine Rohde DSMZ, Braunschweig Leen Van Simaey, Jonas Van Belleghem Hans Duyvejonck Els Van Mechelen, Stefan Vermeulen LBR/Ho. Gent
- Slides: 42