Ph meter By Dr SHAILJA JOGI Govt Bilasa

Ph meter By Dr. SHAILJA JOGI Govt. Bilasa Girls P. G. College Bilaspur

p. H is a unit of measure which describes the degree of acidity or alkalinity (basic) of a solution. It is measured on a scale of 0 to 14. The formal definition of p. H is the negative logarithm of the hydrogen ion activity. p. H = -log[H+]

p. H value The p. H value of a substance is directly related to the ratio of the hydrogen ion and hydroxyl ion concentrations. If the H+ concentration is higher than OHthe material is acidic. If the OH- concentration is higher than H+ the material is basic. 7 is neutral, < is acidic, >7 is basic

The p. H scale corresponds to the concentration of hydrogen ions. If you take the exponent of the H 3 O+ concentrations and remove the negative sign you have the p. H of the solution. For example pure water H+ ion concentration is 1 x 10 -7 M, therefore the p. H would then be 7.

p. H The addition of acid to water increases the concentration of hydrogen ions and reduces the concentration of hydroxyl ions The addition of a base would increase the concentration of hydroxyl ions and decrease the concentration of hydrogen ions

Acids and Bases An acid can be defined as a proton donor, a chemical that increases the concentration of hydrogen ions in solution. A base can be defined as a proton acceptor, a chemical that reduces the concentration of hydrogen ions in solution.

p. H Measurement A p. H measurement system consists of three parts: a p. H measuring electrode, a reference electrode, and a high input meter. The p. H measuring electrode is a hydrogen ion sensitive glass bulb. The reference electrode output does not vary with the activity of the hydrogen ion.

Digital p. H Meter
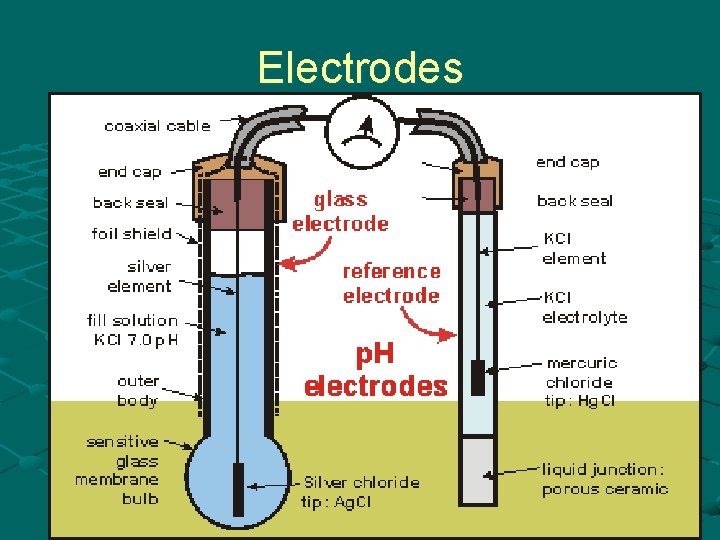
Electrodes
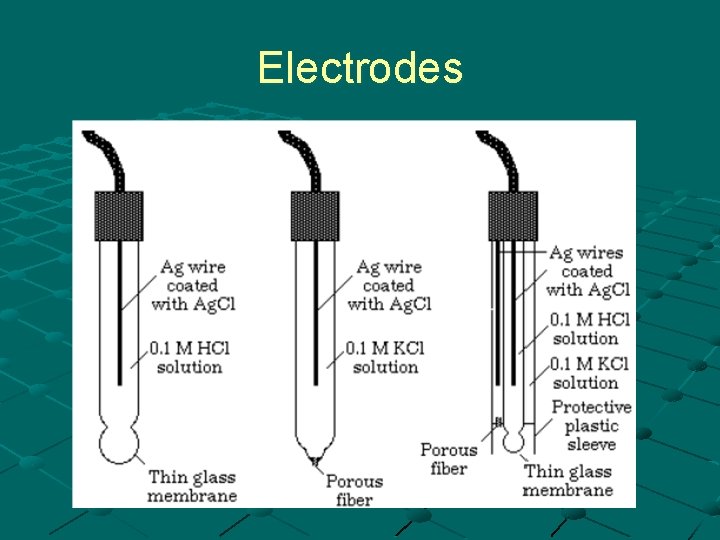
Electrodes

Glass electrode The key feature of the p. Hsensitive electrode is a thin glass membrane whose outside surface contacts the solution to be tested. The inside surface of the glass membrane is exposed to a constant concentration of hydrogen ions (0. 1 M HCl).
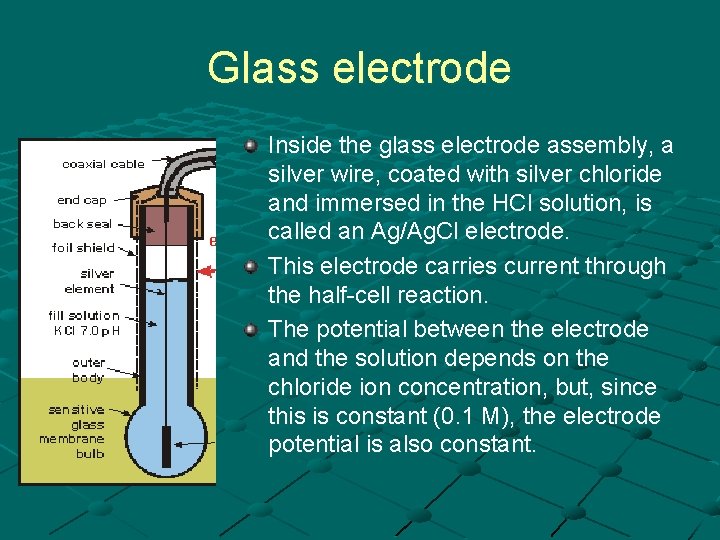
Glass electrode Inside the glass electrode assembly, a silver wire, coated with silver chloride and immersed in the HCl solution, is called an Ag/Ag. Cl electrode. This electrode carries current through the half-cell reaction. The potential between the electrode and the solution depends on the chloride ion concentration, but, since this is constant (0. 1 M), the electrode potential is also constant.

The potential difference between the inside of the glass electrode and the outside is caused by the oxides of silicon in side the glass: Si. O- + H 3. O+ = Si. O. H+ + H 2. O

Reference Electrode A reference electrode is needed to complete the electrical circuit. The calomel reference electrode consists of a glass tube with a potassium chloride (KCl) electrolyte which is in intimate contact with a mercuric chloride element at the end of a KCL element.

p. H Meter A sample is placed in a cup and the glass probe at the end of the retractable arm is placed in it. The probe is connected to the main box. There are two electrodes inside the probe that measure voltage. One is contained in liquid with fixed p. H. The other measures the acidity of the sample through the amount of H+ ions.

p. H Meter A voltmeter in the probe measures the difference between the voltages of the two electrodes. The meter then translates the voltage difference into p. H and displays it on the screen. Before taking a p. H measurement the meter must be calibrated using a solution of known p. H.

Calibration The p. H meter should be standardized each time it is used with a buffer of known p. H To calibrate the p. H meter, rinse the electrode with deionized water Place the electrode in a standard solution, e. g. , p. H 7. Turn the selector to "p. H". Adjust the p. H meter to the appropriate p. H.

Calibration Rinse the electrode with second standard, standard p. H buffers used should be 7 and 4 or 10, depends on the final p. H, . Turn the selector to "p. H". Adjust the temperature knob to the second standard p. H. Rinse the electrode with deionized water, and return the electrode to the soaking solution.

Precautions The glass membrane must be thoroughly hydrated to work properly. Do not allow the electrode to remain out of water any longer than necessary. When the electrode is not in use, keep it immersed in the p. H 4. 00 buffer. Do not put the electrode down on the desk.

Precautions When rinsing the electrode, never wipe the end, but blot gently since wiping can create a static electric charge, which can cause erroneous readings, Make sure the solution you are measuring is at room temperature since the p. H can change with a change in temperature.

Precautions The p. H-sensitive glass membrane is very thin and very easily broken. Do not touch the membrane with anything harder than a Kim-Wipe and do that very gently. Do not drop the electrode or bump it on the bottom of the beaker when immersing it in a solution.

Temperature and Buffers Temperature compensation is contained within the instrument because p. H electrodes are temperature sensitive. Temperature compensation only corrects for the change in the output of the electrode, not for the change in the actual solution. Buffers are solutions that have constant p. H values and the ability to resist changes in p. H. They are used to calibrate the p. H meter.
- Slides: 22